IJCRR - 8(2), January, 2016
Pages: 29-32
Print Article
Download XML Download PDF
EVALUATION OF BLOOD COUNT AMONG BLOOD DONORS ATTENDING KOSTI TEACHING HOSPITAL BLOOD BANK, WHITE NILE STATE, SUDAN
Author: Ahmed M. Elnour, Nada Y. Ali, Moataz M. Ali, Elsharif A. Bazie, Algily A. Edries, Hadeel A. Ali, Mohiera O. Ataalmnan, Abozer Y. Elderdery, Nawal E. Omer
Category: Healthcare
Abstract:Objectives: Current study aimed to evaluate blood counts among blood donors attending Kosti hospital blood bank. Methodology: This cross sectional descriptive study was performed at the blood bank of Kosti teaching hospital during December 2014-Febrauray 2015 and aimed to determine the haematological parameters of blood donors. One hundred donors were chosen randomly to participate in this study. Venous blood (2.5ml) was obtained from each individual into an EDTA container. The full blood count (FBC) was done for each donor, using fully automated haematology analyzer (Sysmex). Peripheral blood film for morphology was done using Giemsa stain. Results: Results showed that haemoglobin concentration was normal in 55% of the donors, less than the normal in 43% and above the normal in 2% of studied group. RBCs were 70% normal, 4% lower than normal and 26% higher than normal. 78% of the donor showed WBC within normal range and the remaining 18% were lower than normal. Platelets counts in donors were 90% normal, 8% low and 2% high. The Haematocrit values were 87% normal, 7% low and 6% high. The MCV in donors was 74% normal and 26% low. MCH value was 2% normal, 97%low and 1%high. The MCHC was 3%normal and 93% low. Conclusion: There is considerable number of donors with haemoglobin concentrations lower than normal as well as lower red blood cells indices. Thus full blood count should be incorporated in evaluating blood donors to insure both good quality of blood and safety of the donors.
Keywords: Blood donors, Red blood cells, Red blood cell indices, Platelets, White blood cells
Full Text:
INTRODUCTION
Blood transfusion is an essential part of modern medicine. While blood transfusion can be life-saving it is not without risk(1). Specific pathologies associated with transfusion include transfusion-related acute lung injury (TRALI), transfusion related immunomodulation (TRIM), circulatory overload, bacterial infections and haemolytic transfusion reactions are complications that can lead to deleterious outcomes(2). Additionally, there is a growing body of evidence indicating an association between transfusion, especially of aged/stored blood products, and increased levels of morbidity and mortality in patients(3). The primary goal of any blood transfusion is to provide the patient with donor red blood cells that optimally survive after transfusion and serve their function and to ensure that the patient actually benefits from the transfusion.
To achieve this goal, donor red cells that are compatible with those of the patient’s blood are selected for transfusion(4). Blood safety is a major concern all over the world. One of the most important steps used to ensure blood safety is blood donor selection(5-8). Blood donor eligibility is determined by medical interview, based on national guidelines for donor selection(9). Donor screening criteria are established to protect both donors and recipients(10). To ensure blood safety, safe donors need to be recruited and high-risk donors should be discouraged from donation(11). For people who are considering donating blood, a local blood bank can describe the criteria for being a blood donor(11). Criteria for selection of donor including: The donor age , Weight of the person, temperature and pulse could be normal, The systolic and diastolic blood pressures are within normal limits without medication, Hb is normal, free from acute respiratory diseases, cardiovascular disease, epilepsy and other Central Nerves System (CNS) disorders(12).
The blood donors must be free from any blood transfusion diseases that, can be determined by history and examination, pregnant and lactating women excluded because of high iron requirements(13). Flowing our visiting Kosti teaching hospital blood bank we observed that, blood donors haematological parameters not measure, ABO blood group and virus screening only laboratory investigation done. So, our current study aimed at to know if the donors haematological parameters within the normal range or not.
MATERIALS AND METHODS One hundred fit blood donors without any history of longterm medication use, illness, and accepted by the physician for donate blood were selected randomly from donors attending Kosti teaching hospital blood bank in White Nile State, Sudan to participate in the current study. Third of the blood donors were from Kosti town, the most blood donors donate before periods 6 month -2 years. Data collected by structural interviewing questionnaire. All participants were completed an individual informed consent form. 2.5 ml of blood was taken from each individual in a sterile and clean EDTA container. The full blood count (FBC) was done, using fully automated haematology analyzer (Sysmex). Peripheral blood film for morphology was done using Giemsa stain. The results were recorded in the master sheet and the information fed to the statistical software program SPSS (version 21), frequencies were calculated as descriptive in all the results.
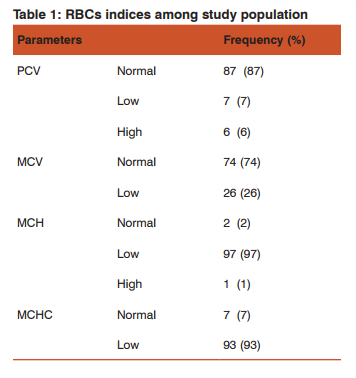
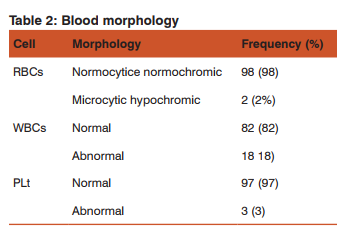
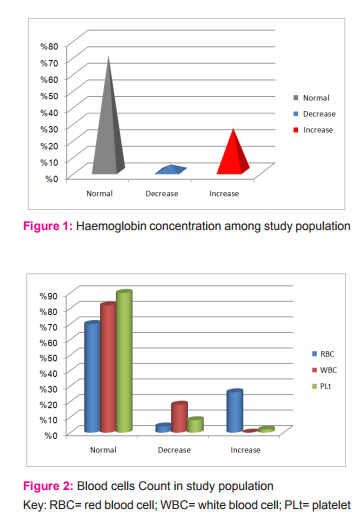
RESULTS Our results showed that all participants were males because females not normally donated blood in Sudan(14). Age ranged from 20 to 54 years. The results were processed statistically by using SPSS (version-21). The results include the following: of the Hb concentrations measured in donors 55% were normal, (43%) less than normal and2% more than normal, as shown in Figure 1.Corresponding results for RBCs were 70% normal, 4% low and 26% high. WBCs in donors were 78% normal and 18%low. Platelets in donors were 90% normal, 8%lowand 2%high (See Figure 2). The RBCs indices were set out in Table 1 as following: the Haematocrit values were 87% normal, 7%lowand 6% high. The MCV in donors was 74% normal and 26% low. MCH value was 2% normal, 97% low and 1% high. The MCHC was 3% normal and 93% low.
DISCUSSION Blood safety is a major concern all over the world. One of the most important steps used to ensure blood safety is blood donor selection. The transfusion of blood and its components is therapeutic and always associated with some level of risk, which if not well-screened, could lead to several complications. Laboratory tests such as a complete blood count are performed to find out if the patient’s symptoms are likely to be relieved. The present study aims to know the haematological profile of screened blood donors attending Kosti teaching hospital, White Nile State, Sudan. Hundred blood donors who accepted by physician were randomly selected for the investigation of haematological parameters. After obtaining an informed consent, 2.5 ml venous blood was collected from the subjects and dispensed into EDTA anticoagulant bottles and mixed by gentle inversion. Complete blood count was determined by haematology autoanalyzer-Symex-Kx-21N, while a thin blood film was prepared for examination of blood cell morphology.
From the results of this study, the majority of blood donors (69%) were between 25 and 39 years of age and all donors were men because usually women do not donate blood in Sudan, this findings consistent with study in Khartoum blood bank, Sudan in 2011stated that the majority of blood donors (74%) were between 25 and 39 years of age and all of them were men(14). The present results revealed that, more than half (55%),(Figure 1) of the participants were in normal Hb concentration, this results were inconsistent with study done in Benin City hospital in Nigeria in 2012(15), reported that, the majority blood donation Hb concentration (Hb) of 91.2% (146/160) of the paid donor blood were ≥12 g/dl while 8.8% (14/160) were >12 g/dl. Concerning RBCs counts the present study showed that, the majority of the participants (70%) within the normal RBCs counts, while quarter of them (26%) showed increased in RBCs count, and only (4%) showed decreased RBCs counts (Figure 2).
On the other hand the majority (82%) of the TWBCs within the normal range and the remaining (18%) showed decreased in TWBCs (Figure 2). 90% of blood donors in this study showed normal PLTs count, 8% increased PLTs count and only 2% showed decreased in PLTs count (Figure 2). Regarding PCV the current study results showed that the majority participants (87%) within the normal reference, 7% of them the PCV was decreased and the rest 6% increased (Table2).In contrast study done in Kenya blood donors haemoglobin (Hb), red cell parameters (red cell count, haematocrit, MCV, MCH and MCHC), total and differential white blood cell (WBC) counts, and platelet counts in two donor populations concluded that, a significant number of Kenyan blood donors showed abnormal haematology profiles(16).
The present study showed that the three quarter (74%) of the blood donors within the normal MCV values and the remaining (26%) bellower the normal value (Table2), while the only of (2%) of the participants found within the normal range of MCH, (1%) higher than the normal range, and the majority(97%) were below than the normal value of MCH (Table 2), these findings were not in agreement with the study done in Suadia Arabia male donors regarding MCV values while consistent with the results concerning MCHthat, haematological parameters(MCV, MCH) were generally lower than reference values amongSuadia Arabia male donors(17).
The status of MCHC showed the almost same values of MCH the majority (93%) were lower than the normal range and only remaining 7% within the normal value (Table1). The present study disagree with the results reported in the previous study done in Nigeria stated that, the MCV of 43.1% (69/160) of paid donors were ≥75 fl while the MCH of 86.9% (139/160) of the paid donors was ≥27 pg and the majority of the test group had MCHC ≥30 g/dl(15). The current study showed that 98% of the RBCs morphology are normocytic normochromic picture (Table2) which not in agreement with the previous study done in which stated, a significant number of the paid donor population in Benin are anaemic (predominantly hypochromic microcytic anaemia)(15).Several studies also have reported anaemia in blood donors(18, 19),with higher frequencies in Africa than in Western countries(19, 20).
CONCLUSION There is considerable number of donors with haemoglobin concentrations lower than normal as well as lower red blood cells indices. Thus full blood count should be incorporated in evaluating blood donors to insure both good quality of blood and safety of the donors. Further studies are needed to strength this recommendation.
References:
1. Vamvakas EC BM. Transfusion-related mortality: the ongoing risks of allogeneic blood transfusion and the vailable strategies for their prevention. Blood. 2009;113(15):3406-17.
2. Tung JP, Fung YL, Nataatmadja M, Colebourne KI, Esmaeel HM, Wilson K, et al. A novel in vivo ovine model of transfusion-related acute lung injury (TRALI). Vox Sang. 2011 Feb;100(2):219-30.
3. Bhaskar B, Dulhunty J, Mullany DV, Fraser JF. Impact of blood product transfusion on short and long-term survival after cardiac surgery: more evidence. Ann Thorac Surg. 2012 Aug;94(2):460- 7.
4. Chapman JF, Elliott C, Knowles SM, Milkins CE, Poole GD. Guidelines for compatibility procedures in blood transfusion laboratories. Transfus Med. 2004 Feb;14(1):59-73.
5. Newman B. Blood donor suitability and allogeneic whole blood donation. Transfus Med Rev. 2001 Jul;15(3):234-44.
6. Eder A BC. Screening blood donors: Science, reason, and the donor history questionnaire. American Association of Blood Banks. 2007.
7. Cable R, Musavi F, Notari E, Zou S. Limited effectiveness of donor deferral registries for transfusion-transmitted disease markers. Transfusion. 2008 Jan;48(1):34-42.
8. Zou S, Musavi F, Notari EP, Rios JA, Trouern-Trend J, Fang CT. Donor deferral and resulting donor loss at the American Red Cross Blood Services, 2001 through 2006. Transfusion. 2008 Dec;48(12):2531-9.
9. Katz L, Strong DM, Tegtmeier G, Stramer S. Performance of an algorithm for the reentry of volunteer blood donors deferred due to false-positive test results for antibody to hepatitis B core antigen. Transfusion. 2008 Nov;48(11):2315-22.
10. Wu Y, Glynn SA, Schreiber GB, Wright DJ, Lo A, Murphy EL, et al. First-time blood donors: demographic trends. Transfusion. 2001 Mar;41(3):360-4.
11. Arslan O. Whole blood donor deferral rate and characteristics of the Turkish population. Transfus Med. 2007 Oct;17(5):379-83.
12. Rysgaard CD MC, Drees D. Positive hepatitis B surface antigen tests due to recent vaccination: a persistent problem. BMC ClinPathol. 2012:12-5.
13. Hoffbrand AV MP, Pettit LE, editor. Essential haematology. Fifth ed. London, UK: Blackwell; 2006.
14. Mahmoud OA GA, Metwally Del S, Elnour AM, Yousif GE Detection of occult hepatitis B virus infection among blood donors in Sudan. J Egypt Public Health Assoc. 2013;88(1):14-8.
15. Benedict N, Augustina AO, Nosakhare BG. Blood donation in Nigeria: standard of the donated blood. J Lab Physicians. 2012 Jul;4(2):94-7.
16. Rajab JA, Muchina WP, Orinda DA, Scott CS. Blood donor haematology parameters in two regions of Kenya. East Afr Med J. 2005 Mar;82(3):123-7.
17. Abdullah SM. The effect of repeated blood donations on the iron status of male Saudi blood donors. Blood Transfus. 2011 Apr;9(2):167-71.
18. Adediran IA, Fesogun RB, Oyekunle AA. Haematological parameters in prospective nigerian blood donors rejected on account of anaemia and/or microfilaria infestation. Niger J Med. 2005 Jan-Mar;14(1):45-50.
19. Tayou Tagny C, Monny Lobe M, Mbanya D. [Evaluation of two methods for haemoglobin measurement in Cameroonian blood donors]. Transfus Clin Biol. 2006 Dec;13(6):331-4.
20. Erhabor O, Ok O, Awah I, Uko KE, Charles AT. The prevalence of Plasmodia parasitaemia among donors in the Niger delta of Nigeria. Trop Doct. 2007 Jan;37(1):32-4.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License