IJCRR - 13(5), March, 2021
Pages: 54-57
Date of Publication: 03-Mar-2021
Print Article
Download XML Download PDF
Antibacterial Activity of Fused Zinc Oxide Nanoparticles with Bacteriocin Sub Class II B [Lactacin F] Against Shigella Species \? A Synergistic Study
Author: S. Kaviya Sri, Malini Evangeline Rose, Monica, Kamala, Muthumariyammal, Shelina Namperipakkam, B. Manipriya
Category: Healthcare
Abstract:Introduction: Antibacterial effect of zinc oxide nanoparticles (ZnO-NPs) has remarked a huge interest overall especially by the usage of nanotechnology to synthesis particles in the nanometer size. ROS has been a central point for a few systems including cell wall destruction because of ZnO confined communication, improved film porousness, the disguise of NPs because of loss of proton intention power and take-up of poisonous broke up zinc particles. Objective: The antibacterial impact of zinc oxide (ZnO) nanoparticle and bacteriocin on Shigella species. Methods: Preliminary tests have been done for the identification of Lactobacillus acidophilus strains producing bacteriocin. Chloroform\?Methanol (2:1 v/v) was used for crude Bacteriocin extraction. However produced precipitate at Solvent-Aqueous interphase was collected aseptically, the solvent was evaporated and the precipitate was kept in a buffer which was used for the antimicrobial study. Sep-pack C18 cartridge and reverse-phase High-Performance Liquid Chromatography on a Nucleosil column were used for purification of lactation F. In this study Agar well diffusion and Broth dilution techniques were used to obtain antimicrobial activity of Lactation F. Synergistic antibacterial activity was done with 1: 1 combination of lactation f and ZnO nanoparticle against. Results: Lactacin F was purified and the antimicrobial testing was done with and without zinc oxide nanoparticle, which shows that Lactation F with zinc oxide nanoparticle had expanded zone of restraint to Shigella species when contrasted with Lactacin F alone. Conclusion: Invitro antibacterial activity of the synergistic compound was more effective than the crude extract of bacteriocin against Shigella species.
Keywords: Bacteriocin, Lactacin F, Shigella species, Synergy, Zinc oxide
Full Text:
INTRODUCTION
Bacteriocins have been grouped into three classes based on the different criteria such as producer organisms, molecular sizes, physical properties, chemical structures and mode of actions. In this present study, we isolated subclass II [b] bacteriocins.1,2 This subclass includes heterodimeric bacteriocins which consist of two peptides. Members of this subclass of the bacteriocins meet three criteria: (i) full antimicrobial activity needs both peptides and the individual peptides show little or no activity, (ii) one immunity protein is sufficient to get immunity, and (iii) the genetic organization of the bacteriocin system includes two sequential bacteriocin structural genes encoding the individual peptides and a single immunity gene. Lactococcin G is the first discovered bacteriocin of this group and its antimicrobial activity depends on both α- and β-peptides.3 Plantaricin and lactacin F are also other important representative examples for two-peptide bacteriocins. Their mechanism of action involves the dissipation of membrane potential and a decrease in the intracellular Adenosine Tri Phosphate concentration. Although the presence of both peptides is required to obtain full antimicrobial activity, the individual peptides can act as residual antimicrobials with a modest effect in some cases.
Having a satisfactory measure of Zn in the body is essential for the protein and DNA combination, ideal working of insusceptible reaction like immune response and metalloenzyme work. Antibacterial effect of zinc oxide nanoparticles (ZnO-NPs) has gotten the huge interest of nanotechnology to integrate particles in the nanometer locale. Specific underline was given to bactericidal and bacteriostatic systems with centre around the age of receptive oxygen species (ROS) including hydrogen peroxide (H2O2), OH-(hydroxyl extremists), and O2-2 (peroxide).4 ROS has been a main consideration for a few components including cell divider harm because of ZnO restricted connection, improved film penetrability, the disguise of NPs because of loss of proton intention power and take-up of poisonous disintegrated zinc particles. These have prompted mitochondria shortcoming, intracellular outpouring, and delivery in the quality articulation of oxidative pressure which caused possible cell development restraint and cell death.5
Thus, in synergy, zinc oxide nanoparticles and lactacin D were used against Shigella species in this study.
MATERIALS AND METHODS
This is the Cross-sectional study done for a period of one year from June 2018- May 2019 at Saveetha Medical College and Hospital, Thandalam.
Isolation and Identification of different probiotics strains6
Two grams of curd sample was transferred in a flask containing MRS Broth as enrichment media and added distilled water to 100 ml and incubated in 37°C. After 24 h, 100 μl of enriched samples were spread on MRS agar. Then, they have anaerobically incubated in [Candle jar] incubator at 37 degree Celsius which is the optimum temperature for lactobacillus species for 24 hours. After the period of incubation 8, isolated colonies were grown. In that, 1 colony shows 100% resemblance with lactobacillus acidophilus. For long term storage Figure 1 shows cultural characteristics of Lactobacillus species. Figure 2 shows Gram staining under the compound microscope with a magnification 100x. Preliminary identification tests like catalase, oxidase, indole and motility were performed. Through antagonist activity of Lactobacillus acidophilus against common pathogens, bacteriocin producers were selected and used for further studies.7 Chloroform – Methanol (2:1 v/v) was used for crude Bacteriocin extraction
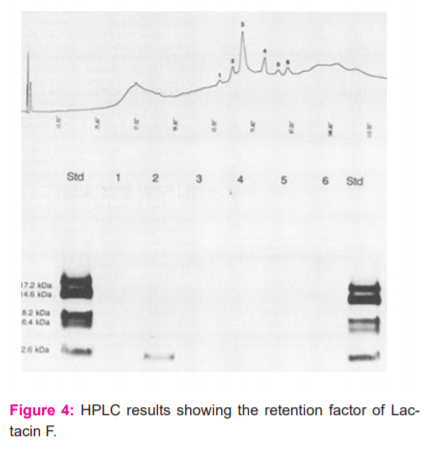
Purification of crude bacteriocin by HPLC method7
Figure 3 shows the Ammonium sulfate precipitation method. Bacteriocin cleansing Method of Precipitation with Ammonium sulfate, Sep-pack C18 cartridge and converse stage HPLC on a C18 Nucleosil section. A 24-h-old culture (200 ml) of L. acidophilus ST31 was centrifuged for 15 min at 20,000 x g, 4ºC. The dynamic supernatant was treated for 10 min at 80ºC to forestall bacteriocin proteolysis. Ammonium sulfate (Kimax) was delicately added to the cell supernatant (kept up at 4ºC) to acquire 60% immersion (1, 6), and blended for 4h. After centrifugation (1h at 20,000 x g, 4ºC), the pellet was resuspended in 25 mM ammonium acetic acid derivation (pH 6.5) and stacked on a Sep-Pack C18 cartridge (Waters Millipore, MA, USA). The cartridge was washed with 20% I-propanol in 25 mM ammonium acetic acid derivation (pH 6.5) and the bacteriocin was eluted with 40% I-propanol in 25 mM ammonium acetic acid derivation (pH 6.5).
In the wake of drying under diminished tension (Speed-Vac; Savant, France), the divisions were halfway disintegrated in 0.1% trifluoroacetic corrosive (TFA) and tried for antimicrobial action. This dynamic portion was additionally cleaned by invert stage HPLC on a C18 Nucleosil segment (250 x 4.6 mm). Elution was performed by applying a straight slope from 0.1% TFA (dissolvable A) to 90% acetonitrile in 0.1% TFA (dissolvable B) in 65 min. Polypeptides, recognized by A220, were gathered physically. After drying under diminished tension and resuspension in 1 ml of de-ionized water, the watery polypeptide arrangements were put away at - 20ºC.
Isolation and Identification of Shigella species
The in-house strain of Shigella dysenteriae and Shigella flexneri were used for antimicrobial testing of Lactacin F with zinc oxide. To check the synergistic activity of Zinc oxide nanoparticles with lactacin F agar cup diffusion method was adapted. 8
Agar cup Diffusion Method
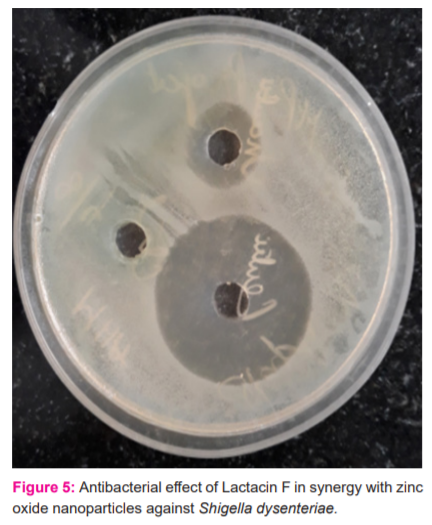
Lactacin F and Zinc oxide nanoparticles of 20- 25nm size were added in 1:1 proportion of microgram per millilitre and tested against lawn culture of Shigella species by making wells in the medium. 9
RESULTS AND DISCUSSION
In this study out of 25 isolates of Lactobacillus species, 18 were Lactobacillus acidophilus. All these strains had antagonistic activity against Escherichia coli ATCC 25922. American Type Culture Collection 10mL of Bacteriocin were extracted and purified from these isolates. Lactacin F was purified and the antimicrobial testing was done with and without zinc oxide nanoparticle, which shows that Lactacin F with zinc oxide nanoparticle had increased (>20mm) zone of inhibition to Shigella species when compared to Lactacin F alone and the positive control with Gentamicin Disc.10 Figure 5 shows the agar cup diffusion testing of the synergy compounds.
Different approaches can be employed which includes herbal-based, 11 in silico based,12 nanomaterial-based,13 and combination therapy.14 These approaches are tried recently against A. baumannii also and some of them shown promising results. ZnO NPs have also shown antimicrobial activity on skin-specific bacteria15 like Streptococcus mutans, Streptococcus pyogenes. This study showed effective antibacterial efficacy of zinc oxide even in presence of Lactacin F against Shigella species.
The activity of lactocidin against Salmonella enteritidis, Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus led to speculate that L. acidophilus producer strains were essential in controlling undesirable flora in the intestinal tract. However, antibacterial activity at pH 7.0 was lost with further purification as described by Vincent in his study.15 Reports also says that Bacteriocin produced by L. acidophilus has a broad-spectrum antagonistic property.16
CONCLUSION
Thus, along with probiotics, nanoparticles can be incorporated and it can play a key role in easy absorption and highly efficient elimination of pathogens in the intestine. Instead of antibiotics, Lactacin F combined with zinc oxide nanoparticles can be effective against enteric pathogens. In future, it helps to reduce the intake of antibiotics and creating community-acquired drug resistance in microbes. This combination may even help in the skin and a soft tissue infection which needs future research work.
Authors acknowledgement: This research was supported/partially supported by Saveetha medical college and hospital. We are thankful to our colleagues who provided expertise that greatly assisted the research.
FUNDING: No funding sources
CONFLICT OF INTEREST: None declared
ETHICAL APPROVAL: The study was approved by the Institutional Ethics Committee, No. SU216/2018
AUTHORS CONTRIBUTION:
Kavyasree and Malini Evangeline rose made substantial contributions to conception, acquisition of data, took part in drafting the article or revising it critically for important intellectual content, Monica, Nithya, Shelina namperipakkam and Muthumariyammal made HPLC analysis [Graphical data] and final approval of the version to be published and agreed to be accountable for all aspects of the work.


References:
-
Field D, Begley M, O'Connor PM, Daly KM, Hugenholtz F, Cotter PD, et al. Bioengineered nisin A derivatives with enhanced activity against both Gram-positive and Gram-negative pathogens. PLoS One 2012;7(2):468-484.
-
Wirawan RE, Klesse NA, Jack RW, Tagg JR. Molecular and genetic characterization of a novel nisin variant produced by Streptococcus uberis. Appl Environ Microbiol 2006;72(5):1148–1156.
-
Oppegard C, Rogne P, Emanuelsen L, Kristiansen PE, Fiml G, Nissen-Meyer J. The two-peptide class II bacteriocins: structure, production, and mode of action. J Mol Microbiol Biotechnol 2007;13(4):210-219.
-
Umamageswari, B. Manipriya, M. Kalyani. Evaluation of Antibacterial activity of Zinc oxide Nanoparticles against Biofilm Producing Methicillin-Resistant Staphylococcus aureus (MRSA). Res J Pharm Tech 2018;11(5):1884-1888.
-
Sirelkhatim A, Mahmud S, Seeni A. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Letters 2015; 7(3):219–242.
-
Saavedra L, Taranto MP, Sesma F, de Valdez GF. Homemade traditional cheeses for the isolation of probiotic Enterococcus faecium strains. Int J Food Microbiol 2003;88:241–245
-
Burianek LL. Yousef AE. Solvent extraction of bacteriocin from liquid cultures. Lett Appl Microbiol 2000;31:193-197.
-
Alabi OS, Sanusi EA. Efficacy of three disinfectant formulations against multidrug-resistant nosocomial agents. Afri J Clin Expe Microbiol 2012; 13(3):178-182.
-
Tiwari V, Tiwari M, Biswas D. Rationale and design of an inhibitor of RecA protein as an inhibitor of Acinetobacter baumannii. J Antibiot 2018;71:522–534.
-
Bhuvaneshwari G, Shameem banuAS and Kalyani M. Nonfermentative gram-negative bacilli: Phenotypic identification and a correlation between biofilm formation and antibiotic susceptibility testing. Int J Res Pharma Sci 2018; 9(4):1229-1234.
-
Roy R, Tiwari M, Donelli G, Tiwari V. Strategies for combating bacterial biofilms: a focus on anti-biofilm agents and their mechanisms of action. Virulence 2018;9(2):522–554.
-
Verma P, Tiwari M, Tiwari V. In-silico high throughput virtual screening and molecular dynamics simulation study to identify inhibitor for AdeABC efflux pump of Acinetobacter baumannii. J Biomol Struct Dyn 2018;36:1182–1194.
-
Wan G, Ruan L, Yin Y, Yang T, Ge M, Cheng X. Effects of silver nanoparticles in combination with antibiotics on the resistant bacteria Acinetobacter baumannii. Int J Nanomed 2016;11:3789–3800.
-
Jain J, Arora S, Rajwade JM, Omray P, Khandelwal S, Paknikar KM. Silver nanoparticles in therapeutics: development of an antimicrobial gel formulation for topical use. Mol Pharm 2009;6:1388–1401.
-
Vincent JG, Veomett RC, Riley RF. Antibacterial activity associated with Lactobacillus acidophilus. J Bacteriol 1959;78:477–484.
Klaenhammer TR. Bacteriocins of lactic acid bacteria. Biochimie 1998;70:337–349.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License