IJCRR - 13(5), March, 2021
Pages: 11-19
Date of Publication: 03-Mar-2021
Print Article
Download XML Download PDF
Application of Newly High-performance ActiveLysine Modified Silica Monolith for Non-steroidal Anti-inflammatory Drugs Extraction
Author: Owidah Z, Jilani AL
Category: Healthcare
Abstract:Introduction: The drug analyses processes include a number of steps starting from samples collection to the final reporting of the results. The process of the drug extraction is usually, time consuming, labour-intensive, complex and prone to contamination. Therefore, the drug extraction can be considered to be one of the most important stages because it is a possible source of inaccuracy in the overall analysis. Objective: The aim of this work is fabricating a new material with the ability to pre-concentrate drugs of abuse in biological fluids with high extraction efficiency. This was achieved by the fabrication of silica-based monolithic materials, followed by chemical on column modifications of the silica surface with active lysine groups. Method: The columns modifications were characterised by using different analytical techniques, such as Brunauer EmmettTeller (BET) analysis and scanning electron microscopy (SEM) coupled with energy dispersive X-ray (EDAX) analysis. Result: In this study, the isolation of non-steroidal anti-inflammatory drugs (NSAIDs), namely acetylsalicylic acid and ibuprofen (IBP), was achieved successfully by using an active lysine silica monolith in which the drugs were adsorbed on solid support while any contaminants were removed by washing the monolithic materials; finally the purified drugs were eluted from the monolithic material. Conclusion: The promising results indicated that the extraction efficiencies of the two NSAIDs were both more than 85%. Linearity was obtained in the range of 10-100 ng ml-1. The intra- and inter-monolithic columns indicated good monolith reproducibility, with relative standard deviations (RSDs) of less than 4.4% and 6.1%, respectively.
Keywords: Quality of Life (QOL), Rectal Cancer, High Anterior Resection (HAR), Low Anterior Resection (LAR), Abdominoperineal Resection (APR)
Full Text:
Introduction
Worldwide today, the utilisation of over counter medicine such as non-steroidal anti-inflammatory drugs (NSAIDs) are the most commonly used as analgesics and an antipyretic. Mainly it used to treat arthritis due to its analgesic and antipyretic properties also, utilised for the treatment of pain, inflammation, vascular headaches and fever.1,2 However, excessive NSAIDs consumption in cases of overdose or chronic abuse can lead to toxic effects include: 1) increased risk of developing renal or hepatic liver tumours; 2) reduced platelet function; 3) changes in kidney function, and 4) prolongation of pregnancy or childbirth on their own.3 To improve NSAID therapy management for long-term, the monitoring of NSAIDs is considered the importance management factor.4 Therefore, it is important to develop a suitable specific and sensitive analytical method to monitor the levels of NSAID compounds in biological samples.5-8
Sample preparation is an essential step in the most analytical instruments to pre-concentrate the analytes of interest or to remove any interfering materials. So far, different sample preparation methods including liquid-liquid extraction (LLE)5-7 and solid-phase extraction (SPE)8-11 have been used for determination and pre-concentration of low levels of NSAIDs from diverse sample matrices. However, LLE is labour-intensive, time-consuming, complicated, non-selective and emulsion formation and carried out depend on hazardous organic solvents.12-14
To overcome these drawbacks the SPE technique has been considered due to its ease of use, being relatively rapid, and being easy-to-automate, portable, and solvent-free.15,16 Many materials can be used as SPE-sorbents including inorganic monolithic materials and organic monolithic materials. In fields of inorganic polymer monolithic material, many publications focused on the extraction of different types of drugs17-19 and Though, there are some drawbacks with organic monolithic materials, such as low mechanical stability, poor hydrophobicity and small surface area, therefore, the widespread application for drug extraction is limited by using inorganic monolithic materials.1,5,6
The major goal of the present work is fabricating an inorganic high performance modified monolithic material which followed by surface modifications with active lysine groups to form ion-exchange materials.7 The silica surface was chemically modified with lysine using a 3-glycidoxy propyltriethoxy silane linkage to get a stable ion-exchange stationary phase.20 The silica was fabricated in a plastic syringe to get rode shape, to use for aspirin and ibuprofen extraction from the biological matrix.
A new anion-exchange/hydrophobic monolith as a stationary phase for nano liquid chromatography of small organic molecules and inorganic anions. Characterisation of the lysine modified monolithic materials was carried out using different techniques to study the morphological, chemical, and physical properties. Also, the chromatographic performance (retention time) or extraction efficiency (recovery of target analytes) was evaluated.
MATERIALS AND METHODS
Chemicals and Materials
Tetraethyl ortho-silicate (TEOS), tetra-methyl-ortho-silicate (TMOS), anhydrous toluene, ammonia, nitric acid, HPLC-grade methanol and acetonitrile (ACN) were purchased from Fisher Scientific ( Loughborough, UK). Polyethene oxide (PEO) with an average relative molecular mass MW of 10,000 Da, acetic acid, 3-Glycidyloxypropyl) tri-methoxy-silane (GPTMS), sodium phosphate dibasic, sodium phosphate monobasic, acetylsalicylic acid, ibuprofen and lysine were purchased from Sigma-Aldrich (Poole, UK). The plastic disposable syringes were purchased from Scientific Laboratory Supplies and Falcon™ conical centrifuge tubes were purchased from Scientific Laboratory Supplies (Nottingham, UK).
Instrumentation
The scanning electron microscope (SEM) was a Cambridge S360 instrument (Cambridge, UK) and the energy-dispersive X-ray spectroscopy (EDAX) system was an INCA 350 EDX system, Oxford Instruments (Abingdon, UK). The samples for SEM analysis were coated with a thin layer of gold-platinum (thickness approximately 2 nm) using a SEMPREP 2 Sputter Coater [Nanotech Ltd., Sandy, UK]. The scanning electron images of silica monoliths were obtained using an accelerating voltage of 20 kV and a probe current of 100 pA in high vacuum mode. A hot-plate stirrer from international LLC (West Chester, PA, USA) was used. The Brunauer Emmett-Teller (BET) form Micromeritics Ltd. (Dunstable, UK) model was used for a surface area and porosity analysis. High-temperature muffle furnace from Laboratory Instruments Ltd (Wertheim, Germany) was used. High-performance liquid chromatography (HPLC) system with a 785A UV/Visible Detector from PerkinElmer (California, USA) was used. The symmetry C18 column, 150 x 4.6 mm packed with5 μm silica particles, was purchased from Phenomenex (Torrance, CA, USA).
Fabrication of the lysine silica monolith column
The fabrication of the silica-based monoliths was carried by the hydrolysis and poly-condensation of precursors using the procedure reported by Nakanishi21 with some modifications to the composition and experimental conditions. The reagents of the silica-based monolith were mixed inside a 50 ml polyethene centrifuge tube which was located in an ice bath to promote the sol-gel reaction. The desired amount of polyethene oxide 0.282 g with an average relative molecular mass (MW=100,000) was mixed with 2.537 ml of 1 M nitric acid and 0.291 ml of distilled water or 4 ml of (0.02M) acetic acid. The solution was then mixed using a magnetic stirrer for 30 min while the polymer fully dissolved. Then, 2.256 ml of tetraethyl ortho-silicate (TEOS) or tetra-methyl-ortho-silicate (TMOS) was added to the transparent solution and mixed for 30 min. until the two-phase mixture gradually became the homogeneous solution.5-8
The disposable plastic syringe (1 ml, internal diameter 4.5 mm) was used as the mould to obtain bar or column-shaped monoliths for the homogeneous mixture. the syringe was sealed at the outlet end with polytetrafluoroethylene (PTFE) tape and then 0.8 ml of the resulting homogeneous mixture was poured slowly down the inside of the syringe which was then shaken carefully to remove any air bubbles. The inlet end of the syringe was closed with a small lid and sealed tightly with PTFE tape as well.
Finally, the syringe was placed upright in a glass beaker and left in the oven at 40°C 24 hours. After that, the monolithic column was removed from the plastic and then placed in a purified water bath to remove any possible residue. Following washing, the resulting monolithic column was treated with 30 ml of 1 M aqueous ammonia solution in a100 mL of the conical flask at 85 ºC for 24 hours. Then, the resulting monolithic column was rinsed with distilled water for 8 hours and the water changed every 2 hours until a neutral pH was obtained. The monolithic columns were then placed in the oven for 6 hours at 40°C, followed by a further 3 hours at 100°C. Finally, the columns were placed in the oven at 500°C for 3 hours.20-22
Modification of the silica monolith surface with lysine groups
The surfaces of the monolithic silica column were covalently modified with lysine to obtain ion exchange phase. This was achieved by generating the desired epoxy, diol, and lysine bonded phases in three main steps as reported by.22
The monolithic column was continuously fed with a mixture of 1 ml of 3-glycidoxy propyl trimethoxysilane (GPTMS) in 10 ml of anhydrous toluene was pumped for three hours. Next, the monolithic column was placed in an oven at 110°C for 1 hour and the epoxy monolith was then thoroughly washed with 1 ml of toluene and then with 1 ml of methanol to clean the column of any residue. To convert the epoxy groups to diols, a 1 ml aliquot of 0.1M hydrochloric acid was passed through the column for 2 hours and then placed in the oven at 60 oC for 1 hour and then washed with 1 ml of purified water and 1 ml of methanol. Finally, the lysine phase was achieved by continuously flowing a mixture of 1 M lysine solution through the monolithic column 2 hours. Subsequently, the monolithic column was placed in the oven at 75oC for 1 hour. The 1 M lysine solution was prepared by dissolving 4.38g of lysine in 50 ml of 50 mM of phosphate buffer at pH 8. Finally, the resulting lysine-bonded phase was rinsed with 1 ml of purified water and 1 ml of methanol at a flow rate of 20 μl min-1 at room temperature.9,10
Characterisation of the fabricated materials
The morphology of the prepared silica monolith was characterised using scanning electron microscopy (SEM). To determine changes of the chemical composition of the internal surface of the silica-based monolith before and after modification with lysine, the energy dispersive analysis of X-ray spectroscopy (EDAX) was performed. Besides, a Brunauer-Emmett-Teller (BET) instrument was used to measure the surface area, pore size, and pore volume within the monoliths using nitrogen adsorption and desorption isotherms at 77 K. The pore volume and pore size distribution within the monoliths were also determined from isotherms using the Barrett-Joyner-Halenda (BJH) model. A small piece of the monolithic column was weighed and placed into the BET instrument for analysis.23-26
Drug Extraction
Acetylsalicylic acid and ibuprofen were prepared separately by dissolving an accurately weighed amount of each drug in 20 mM acetate buffer at pH 4.7 to achieve a concentration of 100 ng ml-1. All stock solutions were stored at 4 °C. Concentrations of the working of 80, 60, 40, 20 and 10 ng ml-1 were freshly prepared by diluting the stock solution in the appropriate buffer and stored at 4 °C.
The lysine monolithic column was cut (1 cm) and placed inside the plastic syringe, which was used as a mould. The steps commonly involved in the solid-phase extraction (SPE) procedure are used as follows: conditioning and equilibration of the adsorbent, loading of the sample, removing of impurities (washing) and eluting of the target analyte. All steps in the extraction procedure used a flow rate of 20 μl min-1. Cleaning and conditioning of the column were carried out with 1 ml of water and then 1 ml of the 20 mM acetate buffer at pH 4 which was used for preparing the working solution. The standard solution (500 µL) was loaded into the column and then washed three times with the buffer. Finally, the elution step was carried out using 20 mM phosphate buffer pH 7 and the elute was collected into an Eppendorf tube for further analysis using the HPLC system.11-15
The chromatographic analysis was performed using HPLC with a UV detector to determine extraction efficiency by comparing the peak areas of the analyte (drug) extraction with the peak areas of the non-processed analyte standard solutions. The mobile phase used is acetonitrile (ACN) and 50 mM of phosphate buffer at pH 5.0 with isocratic conditions (50:50) at ambient temperature (around 23°C). The sample injection volume was 20 μl, the flow rate was set to 1 ml min1, and the detection wavelength was adjusted to 230 nm. The extraction recovery (ER) was calculated by the following equation as reported by Miyazaki et al.23
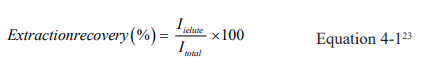
Where Ielute is the amount of analyte eluted from the sorbent, and Itotal is the amount of analyte introduced into the sorbent.
Result and discussion
Formation of a Sol-gel Silica Monolith
In this study, it was decided that silica monolith would be fabricated and followed by modification of the surface with lysine as a sorbent for the extraction of NSAIDs. The formation of the sol is based on hydrolysis and condensation reactions of the metal alkoxides precursors, either TMOS or TEOS.24,25 The composition of the starting mixture was a metal alkoxide, either TMOS or TEOS. The fabrication procedures using a sol-gel process involving two kinds of reactions: hydrolysis of the sol-gel precursor and polycondensation of the hydrolysed products.24 25 The initial solution also contains a water-soluble polymer such as polyethene oxide (PEO) acting as a porogen to form the macropores and micropores in the silica gel.26,27
Also, a catalyst is used which can be an acid catalyst (such as acetic acid or nitric acid), It is known that TMOS is undergoing more rapid hydrolysis in the sol-gel process than TEOS. A weak acid such as acetic acid was therefore used for hydrolysis of TMOS, while a strong acid such as nitric acid was chosen as a catalyst for TEOS, as reported in the literature.28,29
At the mixing step, the partially polymerised solution is then poured into a mould before its viscosity becomes too high. This casting process can be influenced by several factors such as the shape of the mould which determines the final shape of the monolithic column product. In this work, the aplastic 1 ml syringe was used as the mould for the fabrication of silica monoliths. After the polymerisation and drying process, shrinkage of the monolithic structure enabled the silica rods to be released easily from the plastic syringe.
The monolithic rods were then treated with an aqueous 1 M ammonia solution at 80oC for 24 hours to produce the mesoporous. The formation of the mesopores on the monolithic silica surface is vital to obtain a high surface area.Besides increasing surface area, the ammonia hydrothermal treatment increases the mechanical strength of the monolithic structure.30-34 The wet gel monolith is then dried in an air-circulating oven to remove the majority of the solvents used at 40 ºC. After drying, subsequent heat treatment is carried out at a high temperature at 500 oC for 3 hours to decompose the organic residues without serious deformation of the monolithic structure. Besides, the mechanical stability the monolith increases as a result of calculations and a bright white and crack-free monolith was formed.27,35
To identify the effect of each step of the sol-gel process on the internal structure of the silica monoliths, SEM was obtained. Figure 1and Figure 4 shows the internal structure of the TEOS silica monolithic rods during the fabrication process. Figure 1(A) the SEM image of TEOS after the gelation step and before treatment with ammonia. The through pores which give the macroporous structure can be seen and the surfaces can be seen to be smooth. Figure 1 (B) shows the SEM image of TEOS after hydrothermal treatment by ammonia 1M NH4OH, and Figure 1 (C) shows the SEM image of TEOS after the calcination step at 500°C for 3 hours.

Figure 1: The internal structure is analysed for the fabrication steps using SEM images for silica monolithic rods consisting of TEOS + PEO (100K) + 1 M nitric acid (A) before 1 M NH4OH treatment (B) after 1 M NH4OH treatment (C) after 1 M NH4OH treatment and calcination. The scale bar is 1 µm
It can be seen that the surface of the silica monolithic rods changes after hydrothermal treatment, becoming rougher. This rough surface is an indication that mesopores have been tailored inside the surface skeleton by a dissolution re-precipitation process, where the dissolution of the silica occurs on the convex surface and reprecipitation occurs on the concave surfaces. Also, it can be seen that there is no significant difference in general morphology comparing the SEM images of the monolith before and after calcination step.30,31
Modification with lysine groups
Once the silica monolithic column was prepared, it was then modified with lysine. Surface modification of the silica-based monolith was carried out on-column by a continuous flow of the reagents through the porous monolithic silica column inside the heat-shrinkable tube. A system was designed to allow a solid-phase extraction process to occur within a monolithic rod. Figure 2 shows how the monolith rod was sealed within a heat-shrinkable Polytetrafluruoroethylene (PTFE) tube and connected to the borosilicate tube.

Figure 2: The monolith rod sealed within a PTFE tube and connected to the borosilicate tube.
The resulting silica monolithic rod within the heat-shrinkable tube was then ready to be prepared for either surface modification or the solid phase extraction process. This method was very effective and there was no leakage during the continuous flow chemical reaction. The syringe pump was connected to the tubing using a two-piece finger-tight fitting.
In this study, the monolithic silica rod was chemically modified with lysine. The three main steps involved are shown in Figure 3. Scheme (A) in Figure 3 shows the first step where 3- glycidoxypropyltrimethoxysilane (GPTMS) was covalently bonded to the surface of a silica monolith in order to form an epoxy surface monolith. The trimethoxysilane group reacted with the silanol groups (Si-OH) on the surface of thesilica-based monolith, while the glycidyl functional groups offered the necessary active sites for other components to be further immobilised. In the second steps (B) of Figure 3, a diol was generated by treatment with hydrochloric acid. Finally the GPTMS-modified silica was reacted with lysine and the primary amino groups in the lysine form a secondary amino group, as can see in Figure 3 scheme (C)
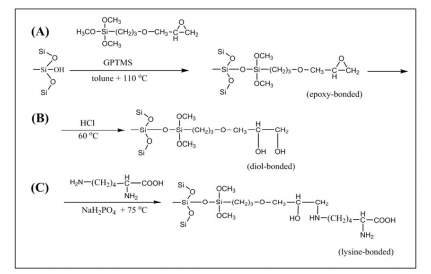
Figure 3: The schematic reaction pathways for the generation of the lysine bonded monolith in three steps
Characteristics of the fabricated materials:
The structural morphology of the lysine modified monolithic rods was examined by SEM analysis to investigate the effect of the surface modification. The SEM image was acquired for the tetraethyl orthosilicate (TEOS) monolithic silica prepared with polyethylene glycol (PEO) 200 K, and Figure 4 shows the internal structure of the TEOS monolithic silica during the modification process steps. Figure 4- (A) shows the SEM image of the TEOS monolithic silica before modification. Figure 4 (B) shows the image after the sanitation process with GPTMS. Figure 4 (C) shows the monolithic silica after the complete lysine modification process.

Figure 4. SEM images for the TEOS monolith column during the lysine modification process. (A) The monolithic column before modification. (B) The monolithic column after silanisation by GPTMS. (C) The monolithic column after lysine modification
Figure 4 shows a homogeneous morphology during the modification process and there are no significant differences in comparing the SEM images of the monoliths before and after modification with lysine. It was expected that the size of the pores of the amino-modified silica monolith would decrease due to the attaching of the lysine onto the inner surface of the pores; however, it was observed that the structural morphology of both samples was similar and there were no significant differences in the through pores for the monolith before and after the modification process.32,33 Further examination of the physical properties of the lysine modified monolithic column was carried out by comparing the surface area and pore volumes before and after modification. Table 1 illustrates the calculated specific surface area (BET method) for TEOS prepared with PEO 200 K after the modification process.
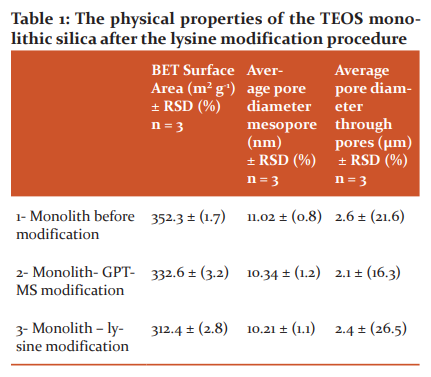
The results in Table 1 show that the surface area decreased as a result of the lysine modifications process from 352 to 312 m² g-1, a reduction of approximately 11%. Whilst there is no significant change in the size of the mesopore and the through pores diameter of modified monolithic column comparing with the result of the non-modified monolithic column in as seen Table 1. The reason for the decrease in surface area after modification could be due to blocking of the micropore access (at scales less than 2 nm) in the silica-based monolith by the bonded phase (lysine chains attached to the silica surface), thus confirming the modification of the surface with lysine groups.36, 37
EDAX or EDX analysis for elemental characterisations was carried out to confirm the modification process by comparing the chemical composition of the monolithic silica column during the modification process. Figure 5 (A) shows the EDX spectra of the bae monolithic silica with the percentage of elements detected being 7, 49, and 45 for C, O, and Si, respectively. Figure 5 (B) silica-based monolith after silanisation with GPTMS with the percentage of elements detected being 14, 46, and 40 for Si, C, and O, respectively. While Figure 5 (C)of the lysine silica-based monolith shows a peak for nitrogen (N) on the monolithic surface after lysine modification, this peak was not present in the other spectra.
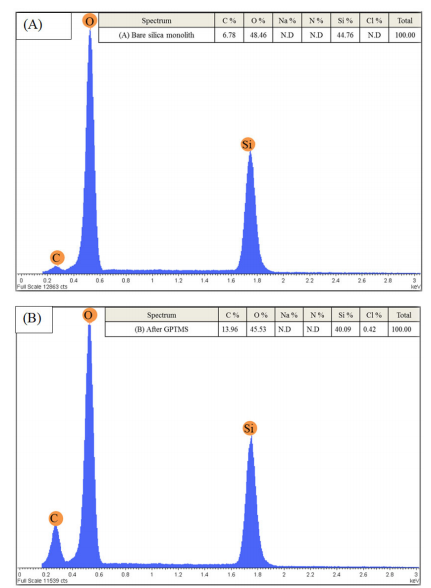
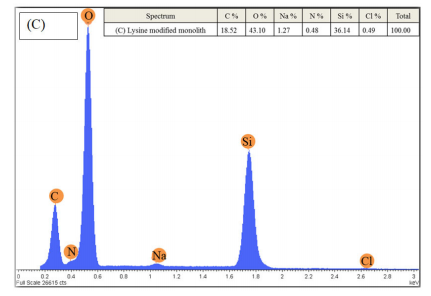
Figure 5 (A) EDX spectra of the 1-non-modified silica-based monolith, (B) silica-based monolith after silanisation with GPTMS and (C) silica-based monolith after lysine modification steps
The corresponding EDX analysis as shown in figure 5 indicates that the monolithic silica is mainly composed of silicon (Si), carbon (C), and oxygen (O). During the modification process, the percentage of carbon increased from 6.78 to 13.96% after glycidyloxpropyl trimethoxysilane (GPTMS) silanisation, indicating that the silanisation process was successful. Further modification of the silica surface with lysine leads to the percentage of carbon increasing to 18.5%. Figure 5 shows the lysine silica-based monolith peak for nitrogen (N) which was discovered on the monolithic surface after lysine modification.34,35 This peak was not present in the other spectra. Na and Cl spectra were also observed with a very low percentage after the lysine modification process and this could have come from the lysine solution, or the H2O used during washing. This indicates that the monolithic surface was successfully modified by the lysine groups.
Extraction of drugs
The fabricated of lysine-TEOS columns was also investigated for the extraction of NSAIDs. In this work, acetylsalicylic acid and ibuprofen were chosen as a model which are acidic compounds due to their pKa values of 3.5 and 4.4 respectively. At a pH greater than 4, most of the drug will be in the ionised form with a negative charge. This value is compatible with the pH range of lysine in zwitterionic state at between 4 and 8 where the lysine can have a positive and negative charge. The mechanism of acidic drug capture with an anion-exchanger depends on the pH of the solution. At pH 4, the amino group in the lysine is positively charged and electrostatic interaction with the negatively charged acidic drug can occur.
Recoveries of acetylsalicylic acid and ibuprofen as acidic drugs were determined by using High performance liquid chromatography (HPLC-Uv) system. A 400μl aliquot of a mixed 100 ng ml-1 acetylsalicylic acid and ibuprofen standard was passed through the lysine modified column using the syringe pump at a flow rate of 20μl min-1. The recovery of the target analytes was calculated by comparing the chromatographic peak areas after direct injection of standard samples with those obtained by the same samples after extraction. The percentage recovery of the target analytes was plotted on a bar chart as shown in Figure 6.
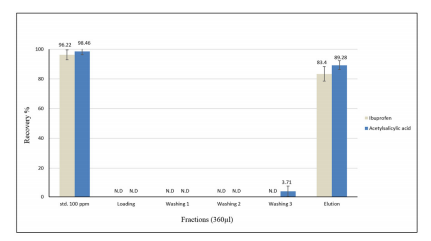
Figure 6: The extraction profile for 100 ng ml-1 of ibuprofen and acetylsalicylic acid using lysine modified column and the average recovery percentage was derived from three consecutive experiments and the error bars indicate one standard deviation
The extraction profile data present in Figure 6 demonstrates the efficient recovery of acetylsalicylic acid and ibuprofen at 89% and 83%, respectively using the TEOS lysine modified column. The remaining small amount of analyte was retained in the column. In general, the TEOS modified lysine column showed good selectivity towards a mixture of acidic drugs and the column enabled efficient extraction.36-38
The method for extraction recovery was evaluated in terms of linearity, LOD, LLOQ, and precision to verify the reliability and applicability of lysine-TEOS columns in extracting acidic drugs when analysed using HPLC with UV detection. Standard solutions of acetylsalicylic acid and ibuprofen at concentrations of 100, 80, 60, 40, 20 and 10 ng ml-1 were analysed by HPLC three consecutive times and the peak areas were plotted against concentration as presented in Figure 7.
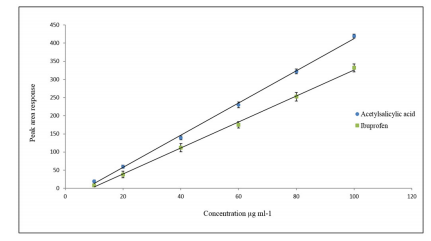
Figure 7: Calibration curve of acetylsalicylic acid and ibuprofen standard solutions with peak areas plotted versus the concentration in μg ml-1
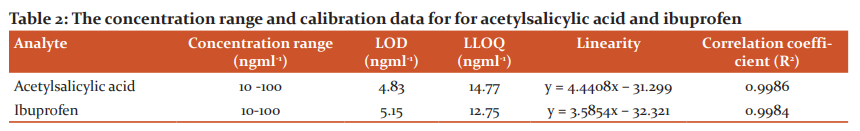
Calibration curves for the target analytes showed a good linearity over the range used and a correlation coefficient (R2) more than 0.9984. LOD and LLOQ were calculated and summarised in Table 2 and 3.
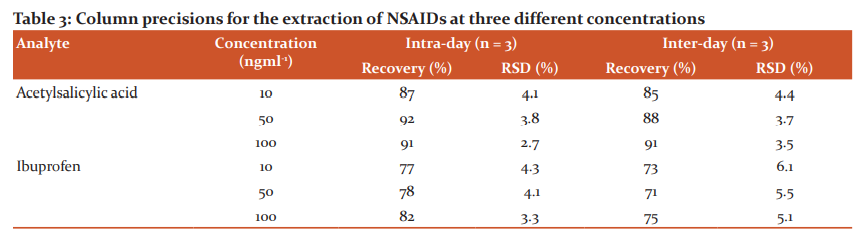
The precision performance of the lysine-TEOS column for extraction was evaluated in terms of repeatability (within-day) and reproducibility (between-day) from the results of the peak areas of each reference standard. In this study, different concentrations were used: low (10ng ml-1), medium (50ng ml-1), and high (100ng ml-1). Each analysis was carried out in triplicate on the same day to determine within-day precision. The same procedure was repeated over three consecutive days in triplicate using the same lysine modified column to obtain the inter-day precision as shown in Table 3. The intra-day %RSD was in the range from 2.7 to 4.3% and the inter-day %RSD in the range from 3.7 to 6.1% for the examined concentrations. These results indicate that using lysine-TEOS columns for extraction could provide good reliability and applicability NSAIDs, as the %RSD values were less than the acceptable limit of 15%.38
Conclusions
In this study, the fabricated lysine silica monolithic column was successfully synthesized by the on-column reaction of lysine with GPTMS-modified silica TEOS monolith which, successfully trap and extract of NSAIDs. The fabricated materials were also studied by using different analytical techniques. Furthermore, the synthesized lysine silica monolith exhibited structure quality and reproducibility with good extraction stability.
References:
1. Luo YB, Zheng H, Wang J, Gao G, Yu Q, Feng Y. An anionic exchange stir rod sorptive extraction based on monolithic material for the extraction of non-steroidal anti-inflammatory drugs in environmental aqueous samples. Talanta 2011;86:103-108.
2. Bempong DK, Bhattacharyya L. Development and validation of a stability-indicating high-performance liquid chromatographic assay for ketoprofen topical penetrating gel. J Chromatogr A 2005;1073(1-2):341-346.
3. Hu T, Ting H, Tao P, Xiao J, Dong C, Han H, et al. Simultaneous determination of thirty non-steroidal anti-inflammatory drug residues in swine muscle by ultra-high-performance liquid chromatography with tandem mass spectrometry. J Chromatogr A 2012; 12(19):104-113.
4. Sarafraz-Yazdi A, Amirhassan A, Gholamhossein R, Hossein EH. Determination of non-steroidal anti-inflammatory drugs in urine by hollow-fibre liquid membrane-protected solid-phase microextraction based on sol-gel fibre coating. J Chromatogr B 2012;908:67-75.
5. Noche GG, Eric M. In situ aqueous derivatization and determination of non-steroidal anti-inflammatory drugs by salting-out-assisted liquid-liquid extraction and gas chromatography-mass spectrometry. J Chromatogr A 2011;1218(37):6240-6247.
6. Roškar R, and Kmetec V. Liq chromatographic determination of diclofenac in human synovial fluid. J Chromatogr B 2003;788(1):57-64.
7. Lo D, Mike V. Acidic and neutral drugs screen in blood with quantitation using microbore high-performance liquid chromatography–diode array detection and capillary gas chromatography–flame ionization detection. Foren Sci Int 1997;90(3):205-214.
8. Migowska N, Chen W, Young C. Simultaneous analysis of non-steroidal anti-inflammatory drugs and estrogenic hormones in water and wastewater samples using gas chromatography-mass spectrometry and gas chromatography with electron capture detection. Sci Total Envt 2012;441:77-88.
9. Rezaei F, Moradi M. Solid-phase extraction as a cleanup step before microextraction of diclofenac and mefenamic acid using nanostructured solvent. Talanta 2013;105:173-178.
10. Madrakian T, Mazaher A, Abbas A, Mohammad S. Selective solid-phase extraction of naproxen drug from human urine samples using molecularly imprinted polymer-coated magnetic multi-walled carbon nanotubes prior to its spectrofluorometric determination. Analyst 2013;138(16):4542-4549.
11. Lyu DY, Yang CX, Yan XP. Fabrication of aluminium terephthalate metal-organic framework incorporated polymer monolith for the microextraction of non-steroidal anti-inflammatory drugs in water and urine samples. J Chromatogr A 2015;1393:1-7.
12. Vas G, Vokey K. Solid?phase microextraction: a powerful sample preparation tool before mass spectrometric analysis. J Mass Spectr 2004;39(3): 233-254.
13. Fan Y, Yu-Qi F, Shi LD, Zhong HW. In-tube solid-phase microextraction using a β-cyclodextrin coated capillary coupled to high-performance liquid chromatography for determination of non-steroidal anti-inflammatory drugs in urine samples. Talanta 2005; 65(1):111-117.
14. Nováková L, Vl?ková H. A review of current trends and advances in modern bio-analytical methods: Chromatography and sample preparation. Analy Chim Act. 2009; 656(1–2): 8-35.
15. Zare F, Ghaedi M, Daneshfar A. The headspace solid-phase microextraction of polycyclic aromatic hydrocarbons in environmental water samples using silica fibre modified by self-assembled gold nanoparticles. Analyst Methods 2015;7(19): 8086-8093.
16. Campestre C, Enric Z. Analysis of imidazoles and triazoles in biological samples after MicroExtraction by the packed sorbent. J Enz Inh Med Chem 2017;32(1):1053-1063.
17. Ma Q, Hu M, Chen F. Preparation of a poly (N?isopropyl acrylamide?co?ethylene dimethacrylate) monolithic capillary and its application for in?tube solid?phase microextraction coupled to high?performance liquid chromatography. J Separ Sci 2009 32(15?16):2592-2600.
18. Svec F. Porous polymer monoliths: The amazingly wide variety of techniques enabling their preparation. J Chromatogr A 2010;1217(6):902-924.
19. Yu QW, Xin W, Qiao M, Bi-Feng Y, Hai-Bo H, Yu-Qi F. Automated analysis of non-steroidal anti-inflammatory drugs in human plasma and water samples by in-tube solid-phase microextraction coupled to liquid chromatography-mass spectrometry-based on poly (4-vinyl pyridine-co-ethylene dimethacrylate) monolith. Analy Meth 2011;4(6): 1538-1545.
20. Sugrue EP, Nesterenko N, Paull B. Fast ion chromatography of inorganic anions and cations on a lysine bonded porous silica monolith. J Chromatogr A 2005;1075(1-2):167-175.
21. Nakanishi K, Soga N. Phase separation in silica sol-gel system containing polyacrylic acid I. Gel formaation behavior and effect of solvent composition. J Non-Cryst Sol 1992; 139:1-13.
22. Huang G, Chen S, Lu C. Preparation and evaluation of a lysine-bonded silica monolith as polar stationary phase for hydrophilic interaction pressurized capillary electrochromatography. Electrophoresis 2008;29(18):3896-3904.
23. Causo TJ, Nischang I, Critical differences in chromatographic properties of silica- and polymer-based monoliths. J Chromatogr A 2014;1358(0):165-171.
24. Kumar A, Dhananjay KT. A review on development of solid phase microextraction fibres by sol-gel methods and their applications. Analyst Chimica Acta 2008;610(1):1-14.
25. Pierre AC. Introduction to sol-gel processing. 1. 2013: Springer Science & Business Media.
26. Nakanishi K, Soga N. Phase Separation in Silica Sol-Gel System Containing Poly (Ethylene oxide) II. Effects of Molecular Weight and Temperature. Bull Chem Soci Jap 1997;70(3):587-592.
27. Nunez O, Nakanishi K, Tanaka N. Preparation of monolithic silica columns for high-performance liquid chromatography. J Chromatogr A 2008;1191(1-2):231-252.
28. Siouffi AM. Silica gel-based monoliths prepared by the sol-gel method: facts and figures. J Chrom A 2003;1000(1-2):801-818.
29. Minakuchi H, Zeho M. Octadecylsilylated porous silica rods as separation media for reversed-phase liquid chromatography. Analyt Chem 1996;68(19):3498-3501.
30. Takahashi R, Nakanishi K, Soga N. Small-angle X-Ray scattering study of nanopore evolution of macroporous silica gel by solvent exchange. Far Discuss 1995;101:249-263.
31. Nakanishi K, Chen H, Feng L, Formation of hierarchical pore structure in silica gel. J Sol-Gel Sci Techn 2000;17(3):191-210.
32. Nakanishi K. Double pore silica gel monolith applied to liquid chromatography. J Sol-Gel Sci Tech 1997;8(1-3):547-552.
33. Ishizuka N, Hiroyoshi M, Kazuki N, Kazuyuki H, Nobuo T. Chromatographic characterization of macroporous monolithic silica prepared via sol-gel process. Colloids and Surfaces A: Physic Engg Asps 2001;187-188:273-279.
34. Puy G. Influence of the hydrothermal treatment on the chromatographic properties of monolithic silica capillaries for nano-liquid chromatography or capillary electrochromatography. J Chromatogr A 2007;1160(1–2):150-159.
35. Fletcher P, Ping H, Stephen MK, Andrew M, Permeability of silica monoliths containing micro-and nano-pores. J Por Mater 2010;3:1-8.
36. Simpson NJ, Solid-phase extraction: principles, techniques, and applications. 2000, New York: Marcel Dekker. xi, 514.
37. Zhao XS, Lu GQ, Hu X. Chemical modification and characterization of MCM-41 and adsorption study. Adsor Sci Tech 2000;12:386-390.
38. Peters FT, Drummer OH, Musshoff F. Validation of new methods. For Sci Int 2007; 165(2–3):216-224.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License