IJCRR - 12(14), July, 2020
Pages: 49-55
Date of Publication: 22-Jul-2020
Print Article
Download XML Download PDF
Effect of Partial Sleep Deprivation on Neurocognitive Functions in Female Nursing Students: An observational study in India
Author: Manisha Uppal, Sujata Gautam, Mary Vibha Lakra, Anupama V Betigeri, Asha Gandhi, Sunita Mondal
Category: Healthcare
Abstract:Aim: The objective of this study was to evaluate the effects of cumulative partial sleep deprivation (i.e. restricted sleep < 6 hours a night for 3-4 consecutive nights) on various aspects of Neurocognitive functions in female nursing students.
Methodology: Pittsburgh Quality of Sleep Index (PQSI) scoring and neurocognitive tests viz. Letter cancellation test (LCT), Trail making tests A and B and Forward and Backward digit span tests were performed in 30 apparently healthy female nursing students. The students, who slept for a minimum of 7 hours / day for at least three consecutive nights, were selected for baseline assessment during their non-sleep deprived phase. Repeat evaluation was performed when the sleep duration was < 6 hours / day for at least three consecutive nights in their sleep-deprived phase.
Result: The study demonstrated a significant increase in PQSI scores (p=0.0.0001***), Letter cancellation test time duration (p=0.0022**), LCT errors (p=0.0109*), Trail making tests A and B time duration (p=0.0048**, p=0.0001*** respectively) and a significant decrease in Backward digit span test score (p=0.0135*) during sleep-deprived phase as compared to non-sleep deprived phase. No significant change was observed in the Forward digit span test (p=0.3097). No statistically significant association was obtained between the PSQI score and the various neurocognitive tests.
Conclusion: Chronic Partial sleep deprivation deteriorates neurocognitive function viz; visual scanning, response speed, sustained attention, executive function, and working memory as demonstrated by significant changes in various neurocognitive tests.
Keywords: Partial sleep deprivation, Neurocognitive function tests, Pittsburgh Quality of Sleep Index
Full Text:
INTRODUCTION
Sleep is a complex and dynamic physiological process essential for human survival. Although the exact sleep requirements may differ for individuals based on age, genetics, social and environmental factors1, it is recommended that adults should sleep seven or more hours per night on a regular basis to promote optimal health2. Unfortunately, compromising on one’s sleep to meet one’s daily responsibilities has become an increasingly common practice amongst people nowadays, in particular among young adults3.
Good quality of sleep is a must for nursing students learning and acquiring clinical skills at a professional level. Poor quality of sleep in students can have an impact on various areas such as efficacy, mental health, stability, physical health, and activity4. While total sleep deprivation is a useful model for studying the neurocognitive effects of sleep deprivation, it is a much less representative form of sleep loss than chronic partial sleep deprivation5. Chronic partial sleep deprivation(sleep restriction to <6hr /24 hours for multiple consecutive days) is much more common nowadays, owing to a wide range of factors including medical conditions, sleep disorders, work demands, and social and domestic responsibilities6.
The prefrontal cortex of the brain, the area responsible for complex cognitive processes, is thought to be especially vulnerable to the effects of sleep loss when planning, coordinating, and self-regulating behaviors are required 7. Partial sleep deprivation has been observed to impair attention, working memory, executive function, and verbal creativity in some studies 8, 9, whereas others have not found any significant decrement in attention10-12, executive function12, or speed of processing 8-10. Though many early studies of chronic partial sleep restriction reported conflicting effects on cognitive performance 5, 6, few experiments in the past few years have also reported that chronic sleep restriction results in rapid cumulative increases in cognitive performance errors and the effects are in proportion to the dose of sleep and chronicity of restriction5, 6, 13-15.
Sleep deprivation and irregular sleeping habits have long been recognized as phenomena prevalent among college students. Nursing students are among those college students that face high levels of stress and time restraints right from the beginning of their course. This makes them especially vulnerable to sleep deprivation. Further, they are expected to learn and perform several clinical tasks. Poor quality of sleep can impact their efficacy. Till date, several studies have been done to evaluate the impact of total sleep deprivation on various parameters. Despite chronic sleep restrictions being more prevalent, not many studies have been done to evaluate its effects, especially in college students. This study aimed to determine whether restricted sleep (<6 hours a night for 3-4 consecutive nights), would have a negative impact on the cognitive performance of nursing students, specifically focusing on visual scanning, response speed and sustained attention, executive function and working memory.
OBJECTIVES
-
To study the effect of cumulative partial sleep deprivation (i.e. restricted sleep <6 hours a night for 3-4 consecutive nights) on neurocognitive functions and sleep quality (PSQI score) in nursing students.
-
To find out the relationship between PSQI score and neurocognitive functions.
MATERIALS AND METHODS
An observational cross-sectional study was carried out in the Department of Physiology, Lady Hardinge Medical College and Associated Hospitals, New Delhi. The study was initiated after taking prior approval from the institutional ethics committee (IEC) for human research. It was conducted according to the principles of the Declaration of Helsinki. We are presenting here only a part of the Short-Term Student ICMR Project (Reference ID: 2014-01641).
Inclusion criteria: The study group comprised of 30 apparently healthy, female nursing students in the age group of 18- 21years. All the study participants served as their own controls.
Exclusion criteria: The study participants with a history of medical illness viz; cardiorespiratory diseases, psychiatric illness, sleep disorders, endocrine disorders, those with history of any drug intake that may affect CNS, those who smoke or consume alcohol were excluded from the study.
Selection and Grouping: The study participants were asked to maintain a daily sleep diary (sleep log book) to record the details of sleep quality and quantity. Participants who had slept for a minimum of 7 hours every night for at least three consecutive days were selected for baseline recordings during their non-sleep-deprived phase (NSD). Regular assessment of their sleep diary was done for about a period of one week to one month, to identify the sleep deprivation period among them. Repeat recordings of all the tests were done in students when the sleep duration was <6 hours per night for at least three consecutive days i.e. in their sleep-deprived phase (SD), to see the effect of chronic partial sleep deprivation in them.
Methodology:
All the subjects were informed about the study and explained the procedure. A signed written informed consent was obtained from each subject. The study participants were called in the morning to the Department of Physiology for the recordings. They were instructed to abstain from coffee/nicotine for at least 12 hours before the recording. A detailed history was taken and a general physical examination was carried out.
All the tests were performed in a quiet room and the temperature was maintained around 23- 25°C. The subjects were allowed to adapt to the experimental conditions and ample time was given to relax. On the day of the recording, all the tests were carried out in each participant in the following sequence during the NSD phase and SD phase.
Pittsburg quality of sleep index (PQSI):
Subjective sleep quality was measured by using the Pittsburgh Sleep Quality Index. This index consists of 19 items that yield scores on 7 subscales (sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleep medications, and daytime dysfunction). The score for each item is from 0 to 3. Additionally, the subscale scores were used to compute a global score (0 to 21), with higher scores indicative of poor sleep quality. Computed global scores > 5 have a diagnostic sensitivity of 89.6% and a specificity of 86.5% to differentiate between good and poor sleepers16- 17.
Neuro-Cognitive Test Battery: In the Neurocognitive Battery18,19, the tests were presented to all the subjects in the following order:
-
Letter cancellation test (LCT): This test assesses visual scanning, response speed, and sustained attention. The task sheet consists of six 52 characters row in which the target character is randomly interspersed approximately 18 times in each row. The subject was instructed to cancel out all target letters. The score was the time taken by the subject to actually perform this task.
-
Trail making tests (TMT): It involves a series of skills related to attention including complex scanning, coordination, visuo-motor tracking, speed of information processing, and motivation. Both parts of the Trail Making Test consist of 25 circles distributed over a sheet of paper.
Part A: Assesses visuo-motor speed and attention. The circles are numbered 1 – 25. The subject is instructed to draw a line to connect the numbers in ascending order. The score is the time taken by the subject to complete the task.
Part B: Apart from testing visuo-motor speed and attention, this test requires the subject to shift strategy and hence, is a sensitive measure of executive function as well. In this subject was instructed to connect (1 – 13) numbered and (A – L) lettered circles by alternating between the two sequences (i.e., 1-A-2-B-3-C, etc.). The total time taken by the subject to complete the task gives the score.
-
Digit span:
Forward digit span (FDS): Assesses immediate verbal memory span. In the test, subjects were asked to verbally repeat sequences of digits of increasing length read out by the examiner. The score was the maximum number of digits that the subject could repeat in the correct order.
Backward digit span (BDS): Besides assessing auditory attention and short-term memory capacity, this test also assesses the ability to manipulate information in the verbal working memory (and hence is a sensitive measure of executive function). The subject had to repeat as sequence of numbers of increasing digit length backward i.e. in the reverse order to what was said by the examiner. The list begins with a 2-digit sequence and the last digit sequence is of 7 numbers. The score was the maximum number of digits that the subject was able to correctly repeat in reverse order.
-
analysis
The data was transferred on an Excel spreadsheet, and descriptive analysis was expressed as mean ± standard deviation (SD). The data was tested for normal Gaussian distribution and a comparison of neurocognitive functions was done using Wilcoxon matched-pairs signed-rank test for non-parametric data and paired t-test for parametric data during non-sleep deprived and sleep-deprived phases. The correlations were performed using Pearson’s correlation for parametric data and spearman’s correlation method for non-parametric data. The level of significance was set at probability value < 0.05, the power of the study was at 80% and α-error was at 5%. For statistical analysis, software GraphPad Prism (version 5.03) was used.
-
The mean age of the participants in the present study was 18.5 ± 0.14 (SD) years and the mean BMI was 20.5 ± 0.18 (SD) Kg/m².
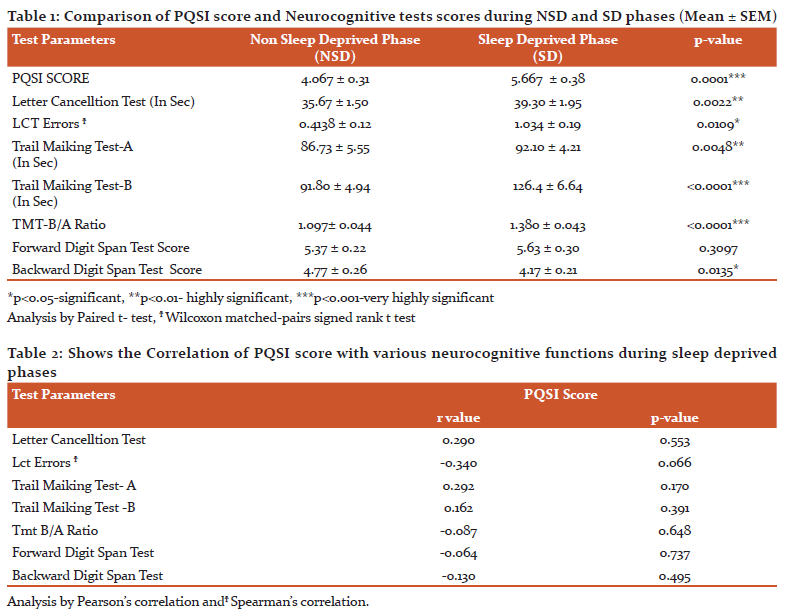
Comparisons of PQSI score and Neurocognitive tests during non-sleep deprived and sleep-deprived phase sis given in Table 1 and demonstrate the following significant observations in sleep deprived phase
-
There was a statistically significant increase (p=0.0001***) in the PQSI scores in the sleep deprived phase as compared to the non-sleep-deprived phase.
-
The time taken to perform the Letter cancellation task was significantly more in the sleep deprived phase (p=0.0022**) as compared to the non-sleep deprived phase. Errors in performing LCT were also more for in the sleep-deprived phase (0.0109*)
-
The mean score for Trail making test-A was significantly more (p=0.0048**) during sleep-deprived phase than during non-sleep-deprived phase. The Trail making test-B mean shows greater time to complete the trail by the participants in sleep deprived phase as compared to non-sleep deprived phase score (p<0.0001***). The ratio of Trail making test B score to Trail making test A score was also significantly increased during sleep deprived phase (p<0.0001***)
-
Significant decrease (p=0.0135*) in the Backward Digit span test score was seen in sleep deprived phase.
Table 2 Demonstrates the relationship between various neurocognitive functions and PQSI scores during sleep deprived phases. No significant association was obtained between the neurocognitive tests (LCT, LCT errors, TMT-AandB, TMT-B score /TMT-A score Digit span forward and backward) and PQSI scores.
DISCUSSION
The present study evaluated the effect of partial sleep deprivation on neuro-cognitive functions in nursing students. The sleep quality was assessed by computing PQSI scores and the cognitive functions were assessed by neurocognitive tests.
-
SUBJECTIVE SLEEP QUALITY: PQSI SCORE
Pittsburgh sleep quality index (PQSI) assesses the sleep quality and quantity in students. There was a statistically significant increase (p=0.0001***) in the PQSI scores in the sleep-deprived phase (5.667 ± 0.38) as compared to the non-sleep deprived phase (4.067 ± 0.31) in our study. This increase in PQSI scores indicates that the sleep quality was poor when the test subjects were partially sleep-deprived. The PSQI test has internal consistency and reliability coefficient (Cronbach’s alpha) of 0.8316- 17.
-
COGNITIVE FUNCTION ASSESSMENT: NEUROCOGNITIVE TESTS
Neurocognitive tests (NCT) are routinely administered to all the age groups both during health and disease to assess various cognitive domains18. These tests can be presented to the subjects using either traditional paper-and-pencil neurocognitive tests or computerized cognitive assessment tests19. In the present study, paper-and-pencil NCTs (LCT, TMT- A and TMT- B, FDS and BDS) were used for assessment of cognitive function and significant differences were found between tests performed during non-sleep-deprived and sleep-deprived phases. For the assessment of cognition, many Indian studies have used these neurocognitive tests20, 21.
The time taken to perform the Letter cancellation task by the participants was significantly more in the sleep-deprived phase (p=0.0022**) as compared to the non-sleep-deprived phase. Errors in performing Letter cancellation test were also more in the sleep deprived phase (0.0109*). The participants took greater time to perform the Letter cancellation task and made more errors and thus, shown a decline in the capacity for concentration, speed of visual scanning, selective attention, and rapid response activation and inhibition.
The participants in sleep-deprived phase also performed poorly and took more time in Trail making test A and B which involved a series of skills related to attention, including complex scanning, coordination, visuo-motor tracking, and speed of information processing. The mean time taken for Trail making test A and B during sleep-deprived phase was significantly more (p=0.0048**, p<0.0001*** respectively) than during non-sleep-deprived phase showing greater time taken to complete the trail by the participants after partial sleep deprivation. The ratio of Trail making test B score to Trail making test A score was also significantly increased during sleep-deprived phase (p<0.0001***). The derived scores provide the advantage of removing the individual variance in speed of response before set-shifting capacity is calculated.
Backward Digit span test assesses the ability to manipulate information in verbal working memory. The score is the maximum number of digits that the subject can repeat in the reverse order after they are read out to them. A significant decrease in the Backward Digit span test score was observed (p=0.0135*) in sleep-deprived phase. No significant change was observed on comparing the baseline mean value of the FDS test during non-sleep deprived phase with its mean value during the sleep-deprived phase. This decrease in backward digit span test score is suggestive of impairment is not just auditory attention and short-term memory, but also in executive function and it appears to be more sensitive test to sleep deprivation. No statically significant correlation was obtained between the various neurocognitive tests and PQSI scores.
There is substantial evidence that the working memory and executive function both heavily rely on the prefrontal cortex, anterior cingulate cortex, and salience network7. The prefrontal cortex is particularly sensitive to sleep loss. Electroencephalographic (EEG) studies have demonstrated that increases in EEG theta activity associated with sleep deprivation are most evident in frontal brain regions22. PET neuroimaging studies corroborate these findings, with results indicating decreased metabolism in the pre-frontal cortex with sleep deprivation23.
The vulnerability of the pre-frontal cortex to sleep loss has led to the hypothesis that tasks such as executive functions should be highly sensitive to sleep loss24. Though results from many studies have supported this notion24-27, many others have failed to demonstrate an influence of sleep loss on executive function28-30. The tasks used to measure cognitive function have been varied and it is likely that elements of these tasks are sub-served by different neural systems, which may be differently affected by sleep loss. In the present study, the decline in neurocognitive function was observed even with 3-4 consecutive nights of cumulative partial sleep deprivation, validating our hypothesis and confirming the effects on sleep physiology and waking functions. The methodology used in the present study was chosen to maximize feasibility, participant familiarity, and minimize testing time. Also, the participants in our study served as their own controls, which took care of inter-subject variability as a confounding factor.
The results obtained in our study are in concurrence with the results of previous studies24-27 and also in line with the result of a recent study in which the authors reported that partial sleep deprivation leads to a statistically significant decrease in cognitive function31. Another recent study showed that cumulative mild partial sleep restriction (1 hour less than what the subjects considered optimal) over 6 nights negatively impacted working memory capacity but had no effect on sustained attention, response inhibition, or decision making32.
Taking more time in performing in Letter cancellation test, Trail making test A and B and making more errors in Letter cancellation task along with an increase in the ratio of Trail making test B score to Trail making test A score and decrease in Backward digit span test score by the study participants during sleep-deprived phase indicates an overall reduction in cognitive abilities. These results obtained thus show that chronic partial sleep deprivation affects attention, visual search and scanning, sequencing and shifting, psychomotor speed, abstraction, flexibility, execution, modification of a plan of action, and additionally, the ability to maintain two trains of thought simultaneously33,34. These abilities are essential for optimum performance in environments that require sustained multi-tasking, organization, and problem-solving. Even at the more basic level of every-day quality of life, executive functions are critical.
Detriments due to sleep loss compromise the health and well-being of students and negatively impact the outcome of the patients they serve. Tasks of sustained visual attention are sensitive to sleepiness35,36. Sustained visual attention is critical for performing cognitive tasks such as driving, maintaining security watch, participating in business meetings, concentrating in lectures, monitoring sophisticated equipment (e.g., medical equipment, air traffic, and space mission controls), assisting in performing the critical procedure and surgeries, etc. In occupations such as medicine, such a decrease in performance can be detrimental, can lead to undesired outcomes, and is a cause of concern with respect to patient safety.
If the demands of nursing students’ training result in sleep deprivation among students, this will impair students’ cognitive processing for two independent reasons: detriments and loss of benefits. The detriments resulting from sleep loss include impaired cognitive and behavioral performance, partly from diminished attention and arousal. The loss of benefits due to the lack of sleep includes missing sleep-dependent cognitive processing such as memory consolidation and insight formation: the building blocks of learning, creativity, and scientific discovery37. These losses are also ethically problematic, incompatible with our health providing goals, and inconsistent with the educational aspirations of training
CONCLUSIONS
There was a significant decline in neurocognitive functions during sleep-deprived phase even with 3-4 consecutive nights of cumulative partial sleep deprivation in nursing students, validating our hypothesis and objectives. This suggests that cumulative partial sleep deprivation indeed affects visual scanning, response speed and sustained attention (Letter Cancellation Test); visuo-motor speed, attention, executive function (Trail Making Test A and B), auditory attention, and short-term memory (Backward Digit Span).
LIMITATIONS
The results of the study may be viewed with caution due to the small sample size (n=30). Also, our study group consisted of only female nursing students. The coping mechanisms and therefore, neurocognitive scores, might differ in their male counterparts. Participants carried out their nights of sleep deprivation in an environment of their choice rather than a supervised environment. Therefore, the study design was reliant on self-reported sleep deprivation and form completion. Objective methods of assessment and a larger test selection would have given a wider, more holistic perspective on the impact of sleep deprivation in the students.
RECOMMENDATIONS
Further studies need to be carried out to explore the importance and impact of a recovery night sleep on the neurocognitive scores after a period of partial sleep deprivation with a larger and more diverse sample group, in terms of sex and age.
ACKNOWLEDGEMENT
The authors are thankful to the Indian Council of Medical Research (ICMR) for accepting their proposal under the Short-term Studentship (STS) program.
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this articlehas been reviewed and discussed.
SOURCES OF FINANCIAL SUPPORT- None
COMPLIANCE WITH ETHICAL STANDARDS
Informed consent: All participants gave written informed consent, and the study was approved by Institutional Ethics Committee (Lady Hardinge Medical College, Delhi, India, ECHR/PR/2014 /36).
Conflict of interest: On behalf of all authors, the corresponding author states that there is no conflict of interest.
References:
-
Chaput JP, Dutil C, Sampasa-Kanyinga H. Sleeping hours: what is the ideal number and how does age impact this. Nat Sci Sleep. 2018;10:421–430.
-
Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38(6):843–844
-
Zitting KM, Münch MY, Cain SW, et al. Young adults are more vulnerable to chronic sleep deficiency and recurrent circadian disruption than older adults. Sci Rep. 2018;8(1):11052.
-
Ryu SH. Quality of life and quality of sleep in medical college students. Korean Soc Biol TherPsychiatr 2009; 15:29-36.
-
Goel N, Rao H, Durmer JS, Dinges DF. Neurocognitive consequences of sleep deprivation. Semin Neurol. 2009;29(4):320–339. doi:10.1055/s-0029-1237117
-
Banks S, Dinges DF. Behavioral and physiological con- sequences of sleep restriction. J Clin Sleep Med 2007;3(5): 519–528
-
Killgore WD. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105-129.
-
Randazzo AC, Muehlbach MJ, Schweitzer PK, Walsh JK. Cognitive function following acute sleep restriction in children ages 10-14. Sleep 1998;21:861–8.
-
Sadeh A, Gruber R, Raviv A. The effects of sleep restriction and extension on school-age children: what a difference an hour makes. Child Dev 2003;74:444–55.
-
Carskadon MA, Harvey K, Dement WC. Acute restriction of nocturnal sleep in children. Percept Mot Skills 1981;53:103–12.
-
Fallone G, Acebo C, Arnedt JT, Seifer R, Carskadon MA. Effects of acute sleep restriction on behavior, sustained attention, and response inhibition in children. Percept Mot Skills 2001;93:213–29.
-
Kopasz M, Loessl B, Valerius G, et al. No persisting effect of partial sleep curtailment on cognitive performance and declarative memory recall in adolescents. J Sleep Res 2010;19:71–9.
-
Van Dongen HPA, Maislin G, Mullington JM, Dinges DF. The cumulative cost of additional wakefulness: dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep 2003;26(2):117–126
-
Dinges DF, Pack F, Williams K, et al. Cumulative sleepiness, mood disturbance, and psychomotor vigilance performance decrements during a week of sleep restricted to 4-5 hours per night. Sleep 1997;20(4):267–277
-
Belenky G, Wesensten NJ, Thorne DR, et al. Patterns of performance degradation and restoration during sleep restriction and subsequent recovery: a sleep dose-response study. J Sleep Res 2003;12(1):1–12
-
Carpenter JS, Andrykowski MA. Psychometric evaluation of the Pittsburgh Sleep Quality Index. J Psychosom Res1998;45(1 Spec No):5-13.
-
Buysse DJ, Reynolds CF, III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiat Res. 1989;28:193–213
-
Maura M, Boone KB, Razani J, DElia LF. Handbook of Normative data for Neuropsychological Assessment. 2nd edition. USA: Oxford University Press;2005.
-
Lezak MD, Howieson DB, Loring DW. Neuropsychological assessment. 4th ed. New York: Oxford University Press; 2004. pp. 337–74.
-
Jaiswal A, Bhavsar V, Jaykaran, Kantharia ND. Effect of antihypertensive therapy on cognitive functions of patients with hypertension. Annals of Indian Academy of Neurology 2010; 13(3): 180–183.
-
Sharma VK, Das S, Mondal S, Goswami U, Gandhi A. Effect of Sahaj Yoga on neuro-cognitive functions in patients suffering from major depression. Indian J Physiol Pharmacol. 2006 Oct-Dec;50(4):375-83.
-
Finelli LA, Baumann H, Borbély AA, Achermann P. Dual electroencephalogram markers of human sleep homeostasis: correlation between theta activity in waking and slow-wave activity in sleep. Neuroscience. 2000; 101:523–529.
-
Thomas M, Sing H, Belenky G, Holcomb H, Mayberg H, Dannals R, Wagner H, Thorne D, Popp K, Rowland L, Welsh A, Balwinski S, Redmond D. Neural basis of alertness and cognitive performance impairments during sleepiness. I. Effects of 24 h of sleep deprivation on waking human regional brain activity. Journal of Sleep Research. 2000; 9:335–352.
-
Harrison Y, Horne JA, Rothwell A. Prefrontal neuropsychological effects of sleep deprivation in young adults- a model for healthy aging? Sleep. 2000; 23:1067–1073.
-
Harrison Y, Horne JA. One Night of Sleep Loss Impairs Innovative Thinking and Flexible Decision Making. Organ Behav. Hum. Decis. Process. 1999; 78:128–145.
-
Harrison Y, Horne JA. Sleep loss impairs short and novel language tasks having a prefrontal focus. Journal of Sleep Research. 1998; 7:95–100.
-
Horne JA. Sleep loss and "divergent" thinking ability. Sleep. 1988; 11:528–536.
-
Tucker AM, Whitney P, Belenky G, Hinson JM, Van Dongen HPA. Effects of sleep deprivation on dissociated components of executive functioning. Sleep. 2010; 33:47–57.
-
Sagaspe P, Sanchez-Ortuno M, Charles A, Taillard J, Valtat C, Bioulac B, Philip P. Effects of sleep deprivation on Color-Word, Emotional, and Specific Stroop interference and on self-reported anxiety. Brain Cogn. 2006; 60:76–87.
-
Fallone G, Acebo C, Arnedt JT, Seifer R, Carskadon MA. Effects of acute sleep restriction on behavior, sustained attention, and response inhibition in children. Perceptual Mot Skills. 2001; 93:213–229.
-
Samatra, Dpg and Kesanda, I Made andAdnyana, Oka and Widyadharma, Eka. (2017). The Effect of Partial Sleep Deprivation in Decrease of Cognitive Function in Resident Doctors of Udayana University/Sanglah General Hospital. International Journal of Science and Research (IJSR). 6. 215-218. 10.21275/ART20172233.
-
Santisteban Lopez, Jose and Brown, Thomas and Ouimet, Marie and Gruber, Reut. (2018). Cumulative mild partial sleep deprivation negatively impacts working memory capacity but not sustained attention, response inhibition, or decision making: a randomized controlled trial. Sleep Health. 10.1016/j.sleh.2018.09.007.
-
Glynda Kinsella and John R. Crawford: Chapter -7: Executive function and its assessment. Neurology and Clinical Neuroscience, 2007, Pages 83-95. Mosby.
-
James A. Holdnack. Chapter -4: The development, expansion, and future of the WAIS-IV as a cornerstone in comprehensive cognitive assessments. Handbook of Psychological Assessment (Fourth Edition)2019, Pages 103-139. Academic Press.
-
Glenville M, Broughton R, Wing AM, Wilkinson RT. Effects of sleep deprivation on short duration performance measures compared to the Wilkinson auditory vigilance task. Sleep. 1978; 1:169– 176.
-
Jewett ME, Dijk D-J, Kronauer RE, Dinges DF. Dose-response relationship between sleep duration and human psychomotor vigilance and subjective alertness. Sleep. 1999; 22:171–179.
-
Jeffrey M. E. Cognitive benefits of sleep and their loss due to sleep deprivation. Neurology. April 12, 2005; 64 (7) E25-E27
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License