IJCRR - 12(13), July, 2020
Pages: 13-18
Date of Publication: 06-Jul-2020
Print Article
Download XML Download PDF
Cardiac and pulmonary late effects in Hodgkin's lymphoma survivors in The Republic of North Macedonia
Author: Gazmend Amzai, Milce Cvetanoski, Oliver Karanfilski, Sonja Genadieva Stavric, Aleksandar Stojanovic
Category: Healthcare
Abstract:Background: Survivors with HL express higher morbidity and mortality rates than the general population of comparable age, mainly because of the late cardiological and pulmonary complications associated with the duration and character of previously administered therapy. The objectives of this study are to determine changes in cardiac and pulmonary functions in patients with Hodgkin's lymphoma treated with chemotherapy with or without mediastinal radiation therapy.
Patients and Methods: The study includes 287 patients with Hodgkin's lymphoma. An analysis of the ECG, echocardiography and spirometric examinations were utilized for the detection of potential cardiac and pulmonary late complications, resulting from the treatment of patients, following long-term remissions.
Results: Late adverse effects are a factor that further increases morbidity and mortality in our surviving HL patients. In 20.8% of the patients, heart disease was detected. It is confirmed that the total dose of doxorubicin influences the onset of late cardiac complications with statistical significance. Late cardiac adverse events occurred in 17.3% of our patients, who received a cumulative dose of doxorubicin greater than 200 mg/sq.m. Patients who received additional mediastinal radiotherapy, had an incidence of heart disease twice higher than patients who received chemotherapy only. In our analyzed series of patients, 24.7% manifested pulmonary toxicity and in 12.7%, a severe degree of pulmonary ventilatory failure was detected. With regard to the latter findings, we also confirm that patients who are receiving bleomycin develop significantly more pulmonary disorders, compared with patients who are receiving chemotherapy regimens without bleomycin.
Conclusion: Improvements have been introduced in formulating certain drugs, so that cardiotoxic effects are reduced or with diminished intensity, or even completely omitting them and replacing them with a less or non-toxic drug. With regard to radiotherapy, size and shape of radiation fields can be precisely customized and dose reduction became acceptable. Despite the progress achieved regarding treatment outcomes in HL patients, due to the optimization of therapeutic protocols, the issue of potential side effects, arising as a treatment consequence, remains a concern.
Keywords: Hodgkin’s lymphoma, Late complications, Cardiac complications, Pulmonary complications
Full Text:
Introduction
HL is very sensitive to chemotherapy and radiation, but treatment-related toxicity raises serious concerns and has a negative impact on the quality of life and the long term overall survival1. Late complications due to toxicity attributable to therapy are a major cause of late morbidity and mortality. Survival is expected to be long enough for patients to carry a lifetime risk of late treatment-related complications. Generally, more serious late complications of treatment are secondary malignancies, cardiac, pulmonary complications and infertility2-5.
A major challenge today is the reduction in mortality from cardio-pulmonary complications arising following treatment. The goal of high quality care is to minimize the adverse effects of treatment, while simultaneously maximizing the disease cure rate.
Many published reports and many relevant studies analyzing large groups of patients, have proven that the toxicity of otherwise successful treatment options ranks cardiovascular disorders as the most common non-malignant cause of death in these long-term survivor patients with Hodgkin's lymphoma.
Patients with Hodgkin's lymphoma, whose overall survival is longer than 5 years, have a 3 to 5 times higher incidence of several cardiovascular disorders, compared to the general population. The 25-year cumulative incidence of heart disease is 7.9% after combined chemotherapy and radiotherapy6.
Anthracyclines are one of the most widely prescribed, as well as among the most effective anticancer agents for the treatment of multiple solid tumors and hematological malignancies. Doxorubicin, as a chemotherapeutic, belongs to the family of anthracycline-type drugs and is a key component of chemotherapy regimens used in the treatment of Hodgkin's lymphoma. Although it is the most active substance and an essential component in the treatment of Hodgkin's lymphoma, unfortunately, life-threatening (lethal) cardiotoxicity continues to compromise its overall value. Despite many years of research, the exact mechanism of cardiotoxicity has not been fully elucidated. It is widely considered that doxorubicin is binding to iron, forming an intracellular complex, which catalyzes the production of highly reactive radicals that cause oxidative damage to myocytes.
The cardiotoxicity of doxorubicin is exponentially proportional to the dose administered. Heart failure will develop in approximately 30% of patients who receive a cumulative dose higher than 550 mg/sq.m. doxorubicin, and only in 4% of patients who receive a lower cumulative dose7-9. Cumulative doses of doxorubicin in the two most widely used chemotherapy regimens for Hodgkin's lymphoma, over 6 cycles of ABVD and 6 cycles of escalated BEACOPP regimen are 300 mg/sq.m. and 210 mg/sq.m. However, there is no safe dose for doxorubicin, because asymptomatic ventricular damage may occur even in patients receiving lower doses of doxorubicin, as a result of myocyte loss and interstitial fibrosis. This is confirmed by a study in which endomyocardial biopsy revealed typical histopathological changes at doses of doxorubicin <300 mg/sq.m. as well10.
Mediastinal radiation plays an important role in the high cure rate of patients with Hodgkin's lymphoma, but carries a high prevalence of symptomatic and preclinical or asymptomatic heart disease, increasing the risk of late heart disease by 2-7 times. The complications after radiation therapy are thought to be due to inflammation and fibrosis caused by radiation over certain cardiac structures, with signs and symptoms that become evident several years after the treatment is completed. Radiotherapy and anthracyclines have shown additive cardiotoxicity11.
In the literature, the frequency of pulmonary toxicity is about 10-25% and is manifested as pneumonitis or progressive pulmonary fibrosis. The incidence is related to the total cumulative dose of bleomycin, advanced patient age and a previous history of pulmonary disease. The immediate bleomycin toxicity-related mortality is about 1%. Patients with lung disease caused by bleomycin have a 5-year overall survival of 63%, versus 90% in patients who did not develop bleomycin-induced lung damage12-16.
Methods
The study incorporates data on 287 patients with Hodgkin’s lymphoma, all diagnosed and treated at the University Clinic of Hematology in Skopje from 2005 to 2015, with further follow up until the end of 2018. A majority of the patients (91.6%) received ABVD and 27.5% of patients initially received adjuvant mediastinal radiotherapy. Patients participating in the study should meet the following criteria to be included: age >14 years, documented histopathological diagnosis i.e., initial diagnosis of Hodgkin’s lymphoma, valid medical documentation with clinical and laboratory data on the primary disease, administered therapy as well as the clinical follow-up data of the patient. Patients who experienced long-term remissions underwent the following procedures:
Cardiologists and pulmologists analyzed echocardiograms and spirograms. All patients had cardiac and pulmonary evaluation prior to the start of chemotherapy and showed no sign of disease. Patients who had active heart or lung disease were excluded from the study prior to treatment. Assessment, toxicity analysis, distribution of late types of complications and non-neoplastic complications (cardiac, pulmonary complications), following both chemotherapy and radiotherapy, which became notable in our patient population during the follow-up and observation period, were the points of interest of this study. Data was compared with relevant literature reports, in order to assess the rate of complications in our population, versus data reported in respectable journals.
The statistical program SPSS 17.0 was used for performing general and specific statistical analyses. Databases were created in standard and generally available software programs. Standard descriptive and analytical methods were used for data presentation and preparation for analyses. The Shapiro-Wilk`s test was used for determination of statistical significance for p less than 0.05. The results are presented in tables and graphics.
Results
Post-therapy echocardiography, ECG and cardiac examination were performed in 168 patients with HL. In 79.2%, or 133 patients, a regular cardiac finding was recorded and in 20.8%, or 35 patients, a cardiac disorder was detected, mainly cardiomyopathy (40%), shown in Table 1.
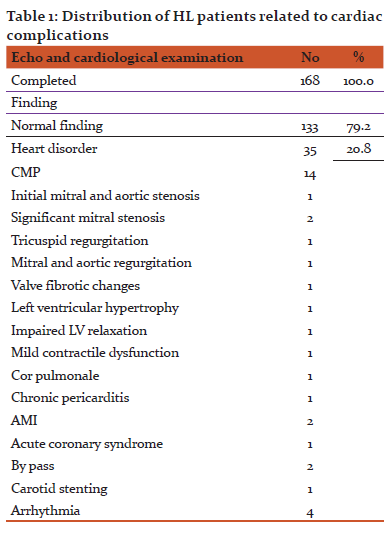
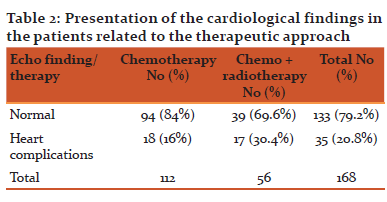
In principle, we have monitored the effect of radiation and assessed the long-term cardiac adverse effects. There is a definite relationship between the treatment modality, respectively patients receiving chemotherapy alone and patients receiving combined therapy (chemo + radiotherapy) and the echocardiographic abnormalities, delineating the more intensely treated population as the one at higher risk, manifesting significant statistical difference with a p-value <0.05 (Pearson Chi-square = 9.28892, p = 0.002305). In our population, radiotherapy doubled the chances of cardiac complications.
Cardiac disorders occurred in 16% of the patients receiving chemotherapy alone and in 30% of patients who received combined modality treatment (Table 2).
TABLE 2. Presentation of the cardiological findings in the patients related to the therapeutic approach
Doxorubicin is a major incriminated cytotoxic drug regarding post-chemotherapy heart disorders. Out of 35 patients with cardiac late adverse events reported, 29 received a total dose of doxorubicin >200 mg/sq.m. and 6 patients received a total dose of doxorubicin ≤200 mg/sq.m., the percentage difference being statistically significant with a p value <0.05 (Difference test p = 0.0000)
Spirometry, for evaluation of pulmonary function after treatment (chemotherapy alone or combined chemoradiotherapy), was performed in 166 patients with HL.
Out of the HL patients who underwent spirometry, 75.3% had regular pulmonary function and 24.7% had impaired pulmonary function. In 10.2% a mild degree of pulmonary ventilatory failure was reported. Severe degree of pulmonary ventilatory failure was reported in 12.7% of patients, while 1.8% reported a moderate degree of pulmonary ventilatory failure. Fibro-deformant bronchitis was registered in one patient (Fig.1).
FIG. 1. Distribution of patients with HL related to spirometry
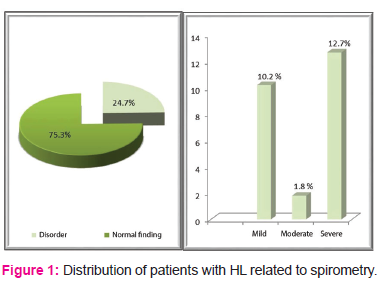
No association was observed between the treatment modality (patients receiving chemo and chemo + radiotherapy) and the spirometry findings (Pearson Chi-square = 3.12810, p = 0.372294).
Positive spirometric findings were reported in 41 patients, all receiving treatment with bleomycin. Of all the cases tested by spirometry, only 9 patients received AVD, i.e., bleomycin-free chemotherapy and in those patients all spirometric tests were normal. According to the Fisher exact test, this finding carries statistical significance (p = 0.0494) (Table 3).
TABLE 3. Presentation of spirometric findings in patients treated with and without bleomycin

Discussion
HL survivors make up one of the largest groups of cancer surviving patients in the World. However, surviving HL patients manifest a higher morbidity and mortality rate compared to the general age-matched population, due to the long-term therapy-related side effects. Improvements in the treatment (aggressively optimized treatment protocols) leave the question of possible long-term adverse effects open, mainly regarding the risk of developing a second malignancy, the risk of non-neoplastic complications, cardiac and pulmonary, as well as the issue very important to patients, such as the risk of developing secondary infertility.
In this study, we evaluated the incidence of cardiotoxicity in a total of 168 patients with Hodgkin's lymphoma, treated with chemotherapy, with or without adjuvant radiotherapy. We defined the characteristics of cardiac disorders and analyzed the risk factors contributing to the development of late adverse effects of treatment affecting the cardiac function. The utilized diagnostic procedures for evaluation of cardiac function, data regarding disease history, findings on physical examination, ECG and echocardiography results, revealed that 35 patients, or 20.8% of the total number, had developed verified valvular, myocardial and pericardial abnormalities. Of the total, 29 patients (17.3%) with documented late cardiac adverse events had received a cumulative dose of doxorubicin greater than 200 mg/sq.m. According to the Haddy study, 14% of the patients that received more than 200 mg/sq.m. doxorubicin had developed left ventricular dysfunction17. Therapy-related adverse events in the long-term follow-up validated the argument that doxorubicin is the primary responsible agent for acquiring heart disorders. There is no defined safe dose for doxorubicin, because of the fact that a subclinical form of cardiac toxicity also occurs in patients that had received lower doses of doxorubicin.
Reducing the risk of cardiotoxicity also means reducing the need for monitoring and specific cardiological treatment, which also means lowering the costs to the healthcare system and most importantly, improving the quality of life for the patient. The strategy of minimizing the toxic effects of doxorubicin and maximizing the therapeutic benefits of this agent, incorporates identifying high-risk populations, limiting the cumulative dose, individualizing the dosing approach, controlling the administered dose (reduction of maximal plasma levels of doxorubicin with continuous infusion reduces cardiotoxicity), use of liposomal formulations or newer analogue anthracyclines, as well as the use of cardioprotectors during the course of treatment18. Cardiac dysfunction associated with the administration of doxorubicin is treated much like the other forms of heart failure. Additional cardiotoxic drugs should be avoided, risk factors for cardiovascular disease should be reduced, blood sugar levels should be under control, hypertension also, lifestyle should be appropriately adjusted and cardiological medications should be used regularly.
To analyze the correlation between the type of therapy and the risk of heart failure, patients were categorized into non-radiotherapy and radiotherapy groups. A consequential connection between supradiafragmatic radiation and late cardiac complications has been confirmed. The risk of cardiac complications after radiation, in patients who have previously received chemotherapy that contains doxorubicin, is twice as high. Significant adverse effects of radiotherapy have been confirmed in many other studies in patients with Hodgkin's lymphoma who also received chemotherapy11. The duration of follow up in our study is comparable to other relevant studies, and is sufficient to estimate the impact of treatment on late cardiac disorders. However, in order to fully identify the magnitude of late cardiac adverse effects, an additional, longer follow up is required. The increased risk of heart failure persists longer after the completed treatment. According to a study, surviving patients with Hodgkin's lymphoma are exposed to a high risk of developing various cardiovascular disorders, even 35 and more years following treatment. They have a 4 to 6 times higher risk of developing cardiovascular disorders compared to the standard incidence of heart failure in the general population19. Doctors and patients need to be aware and critical of controlling this progressively increasing risk.
Bleomycin, as part of the ABVD regimen, can cause serious acute and chronic pulmonary toxicity. Although the incidence of bleomycin-induced clinically relevant pulmonary toxicity is small, the doctors are concerned since the risk of developing it cannot be predicted. A total of 166 patients with HL were analyzed for pulmonary complications after chemotherapy only and combined modality (chemo + radiotherapy) treatment. A pulmonary function reduction has been detected in almost 24.7% of the cases, and we emphasize the fact that in 12.7% of patients, the disorder is defined as a severe degree of pulmonary ventilation insufficiency. In this study, we found no significant effect of radiotherapy on the development of functional pulmonary diseases.
There is a significant difference in the risk for patients that received bleomycin, compared to those who had not received bleomycin (the group where bleomycin was discontinued due to drug hypersensitivity). Bleomycin plays an influential role in the occurrence of secondary pulmonary adverse effects. In practice, bleomycin toxicity can be reduced only if continuous measurements of diffusing capacity of the lungs for carbon monoxide (DLCO) are performed during treatment: if the capacity is reduced by 10% or more, bleomycin should be omitted in the further cycles of chemotherapy20,21. However, CO diffusion capacity is rarely monitored in patients undergoing ABVD chemotherapy. Under such circumstances, spirometry (pulmonary function tests) is a useful method for identification of patients who are developing bleomycin-induced toxicity and reduction in certain parameters may correlate with radiological changes and development of symptoms12,22. No matter what the real value of bleomycin in ABVD is, the replacement of bleomycin with brentuximab (Brentuximab + AVD) has been proven justifiable, since improved efficacy of this chemotherapy approach has been well documented23. The US Food and Drug Administration (FDA) approved brentuximab vedotin in March 2018 in combination with chemotherapy, for previously untreated patients with stage III or IV HL, which is an improvement of the initial standard treatment strategy (ABVD), introduced into clinical practice over 40 years ago.
The distribution of late cardiac and pulmonary complications and the comparison with relevant literature data, have shown comparable levels of occurrence as in the rest of the World population.
There is a strong tendency in recent years to adopt a general standpoint that improving the level of antitumor efficacy should be adequately compensated by minimizing the possibilities of developing immediate and late cardiac and pulmonary adverse events, as well as with minimizing fatal outcomes.
Conclusions
With the exception of some centers, contemporary medicine recommends two chemotherapy regimens for induction of remission in HL patients, and the scale of implementation depends on the region where one practices medicine. Having the experience of using both regimens widely and for a substantial period of time, today we are in the position to reflect on their positive effects and benefits for the patients, but also on their adverse effects. Knowing both aspects of their possibilities, contemporary tendencies are aimed at optimization of chemotherapy regimens, by means of dose reduction or absolute exclusion of toxic components and substitution with non-toxic and maybe even more potent drugs, based on completely different principles of action. A positive example is the elimination of bleomycin from both regimens and the introduction of AVD and the BrECADD regimens, as well as the trends towards incorporating novel agents, such as checkpoint inhibitors in treatment protocols.
Acknowledgements
The authors express their gratitude to Prof. Dr. Rosalinda Isijanovska for her statistical programming support in preparing this manuscript and all the patients who participated in this study.
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Abbreviations
HL - Hodgkin's lymphoma
HD - Hodgkin's disease
ECG - electrocardiogram
ABVD - doxorubicin, bleomycin, vinblastine, dacarbazine
BEACOPP - bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone
CMP - cardiomyopathy
LV - left ventricle
AMI - acute myocardial infarction
AVD - doxorubicin, vinblastine, dacarbazine
CO - carbon monoxide
BrECADD - brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin, dacarbazine, dexamethasone
Statement of Ethics
The Ethics Committee of the University “Sts. Cyril and Methodius”, Medical Faculty, Skopje, approved this study.
Disclosure Statement
The authors do not have any conflicts of interest to report regarding this study.
Funding Sources
There was no source of funding assigned to this study.
Author Contributions
All authors analyzed, reviewed and interpreted the patient data. G. Amzai is the major contributor in composing and writing the manuscript. O. Karanfilski and A. Stojanovic critically reviewed the paper, contributing with minor modifications. All authors reviewed and approved the final manuscript and are responsible for the accuracy and integrity of all aspects of the manuscript.
References:
-
Andrea K Ng. Current survivorship recommendations for patients with Hodgkin lymphoma: Focus on late effects. Blood. 2014 Nov;124(23):3373–79.
-
Hodgson D. Late Effects in Era of Modern Therapy for Hodgkin Lymphoma. Hematology Am Soc Hematol Educ Program. 2011 Dec;2011(1):323-9.
-
Master M, et al. Late Morbidity and Mortality in Patients With Hodgkin’s Lymphoma Treated During Adulthood. J Natl Cancer Inst. 2015 Feb;107(4).
-
Bessell EM, et al. Long-term survival after treatment for Hodgkin’s disease (1973-2002): improved survival with successive 10-year cohorts. Br J Cancer. 2012 Jul;107(3):531-6.
-
Castellino SM, et al. Morbidity and mortality in long-term survivors of Hodgkin lymphoma: a report from the Childhood Cancer Survivor Study. Blood. 2011 Feb;117(6):1806-16.
-
Aleman BM, van den Belt-Dusebout AW, De Bruin ML, et al. Late cardiotoxicity after treatment for Hodgkin lymphoma. Blood. 2007 Mar;109(5):1878-86.
-
Swain SM, Whaley FS, Ewer MS. Congestive heart failure in patients treated with doxorubicin: A retrospective analysis of three trials. Cancer. 2003 Jun;97(11):2869–79.
-
Lefrak EA, Pitha J, Rosenheim S, et al. A clinicopathologic analysis of Adriamycin cardiotoxicity. Cancer. 1973 Aug;32(2):302-14.
-
Steinherz LJ, Steinherz PG, Tan CTC: Cardiac toxicity 4 to 20 years after completing anthracycline therapy. JAMA. 1991 Sep;266(12):1672-7.
-
Friedman MA, Bozdech MJ, Billingham ME, et al. Doxorubicin cardiotoxicity: Serial endomyocardial biopsies and systolic time intervals. JAMA. 1978 Oct;240(15):1603-6.
-
van Nimwegen FA, Ntentas G, Darby SC, Schaapveld M, et al. Risk of heart failure in survivors of Hodgkin lymphoma: effects of cardiac exposure to radiation and anthracyclines. Blood. 2017 Apr;129(16):2257-65.
-
Sleijfer S. Bleomycin-induced pneumonitis. Chest. 2001 Aug;120(2):617-24.
-
Brice P, Tredaniel J, Monsuez JJ, et al. Cardiopulmonary toxicity after three courses of ABVD and mediastinal irradiation in favorable Hodgkin's disease. Ann Oncol. 1991 Feb;2(2):73-6.
-
Jóna Á, Illés Á, Szemes K, Miltényi Z1. Pulmonary alterations in Hodgkin lymphoma. Orv Hetil. 2016 Jan;157(5):163-73.
-
Horning SJ, Adhikari A, Rizk N, et al. Effect of treatment for Hodgkin's disease on pulmonary function: results of a prospective study. J Clin Oncol. 1994 Feb;12(2):297-305.
-
Hirsch A, Vander Els N, Straus DJ, et al. Effect of ABVD chemotherapy with and without mantle or mediastinal irradiation on pulmonary function and symptoms in early-stage Hodgkin's disease. J Clin Oncol. 1996 Apr;14(4):1297-305.
-
Haddy TB, Adde MA, McCalla J, Domanski MJ, Datiles M 3rd, Meehan SC, Pikus A, Shad AT, Valdez I, Lopez Vivino L, Magrath IT. Late effects in long-term survivors of high-grade non-Hodgkin's lymphomas. J Clin Oncol. 1998 Jun;16(6):2070-9.
-
Legha SS, Benjamin RS, Mackay B, et al. Reduction of doxorubicin cardiotoxicity by prolonged continuous intravenous infusion. Ann Intern Med. 1982 Feb;96(2):133-9.
-
van Nimwegen FA, Schaapveld M, Janus CPM, et al. Cardiovascular disease after Hodgkin lymphoma treatment: 40-year disease risk. JAMA Intern Med. 2015 Jun;175(6):1007-17.
20. Luursema PB, Star-Kroesen MA, van der Mark TW et al. Bleomycin-induced changes in the carbon monoxide transfer factor of the lungs and its components. Am Rev Respir Dis. 1983 Nov;128(5):880–3.
-
Comis RL, Kuppinger MS, Ginsberg SJ et al. Role of single-breath carbon monoxide-diffusing capacity in monitoring the pulmonary effects of bleomycin in germ cell tumor patients. Cancer Res. 1979 Dec;39(12):5076–80.
22. Wolkowicz.J, Sturgeon J, Rawji M, Chan CK. Bleomycin-induced pulmonary function abnormalities. Chest. 1992 Jan;101(1):97–101.
23. Connors JM, Jurczak W, Straus DJ, et al. Brentuximab vedotin with chemotherapy for stage III or IV Hodgkin's lymphoma. N Engl J Med. 2018 Jan;378(4):331-44.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License