IJCRR - 12(13), July, 2020
Pages: 06-12
Date of Publication: 06-Jul-2020
Print Article
Download XML Download PDF
Influence of Basic Fibroblast Growth Factor on Biological and Phenotypic Properties of Human Bone Marrow Mesenchymal Stem Cells
Author: Narendra Nitilapura, Shama Rao, Siddharth M. Shetty, Meenakshi Arumugam, Veena Shetty A., Mohana Kumar B.
Category: Healthcare
Abstract:Background: Bone marrow-derived mesenchymal stem cells (BMSCs) are ideal candidates for cell-based therapy due to their self-renewal and multilineage differentiation ability. During in vitro expansion, BMSCs tend to lose their proliferation rate and multipotency limiting their clinical use. Hence, supplementation of basic fibroblast growth factor (bFGF) during in vitro culture might positively influence the biological and phenotypic properties of expanded BMSCs.
Aim: This study was aimed to evaluate the supplementation of bFGF on selected biological and phenotypic characteristics of BMSCs.
Methods: Plastic-adherent BMSCs were cultured without (0 ng/mL) and with (5 ng/mL and 10 ng/mL) bFGF up to five passages and analyzed for their morphology, viability, proliferation rate, population doubling time (PDT), colony forming unit-fibroblast (CFU-F) assay, senescence activity, genetic stability and cell surface marker expression.
Results: BMSCSs exhibited a small, spindle-shape morphology in bFGF supplemented groups as compared to elongated fibroblast-like cells in control. No significant difference in viability and PDT was observed. However, noticeable differences were observed in proliferation rate and CFU-F ability between bFGF supplemented group and control. Further, the senescence activity of BMSCs was considerably decreased under the influence of bFGF. BMSCs had a normal karyotypein both bFGF supplemented and control groups. Lastly, addition of bFGF in expansion media slightly modified the phenotypic markers expression in BMSCs, corresponding to the concentrations used.
Conclusion: Supplementation of BFGFat10 ng/mL could be considered as an optimal and efficient concentration for expanding BMSCs in culture before their use in cell-based therapy.
Keywords: Basic fibroblast growth factor, Bone marrow mesenchymal stem cells, Human, In vitro, Markers expression
Full Text:
INTRODUCTION
Bone marrow is considered as the most commonly used tissue source of mesenchymal stem cells (MSCs)1. MSCs derived from bone marrow (BMSCs) are ideal candidates for cell therapy due to their ability of self-renewal and multilineage differentiation2. It is known that, occurrence of MSCs in the bone marrow is in low amount (0.001-0.01%), and ex vivo expansion is indispensable to obtain sufficient cell number for clinical applications and tissue engineering purposes3. It is observed that for cartilage tissue engineering, 8-10×107 cells/mL of tissue are needed initially4, and based on different nature of defects, a large number of cells are required for cell therapy purposes5. However, prolonged in vitro culture expansion of MSCs reduces proliferation potential and multipotency, and further leading to senescence which is undesirable for cell therapy6.Strategies such as modification of media composition, cell culture conditions, and the use of growth factors to enhance the proliferation of MSCs have been proposed to minimize the detrimental features.
Among the growth factors being employed, basic fibroblast growth factor (bFGF) has been shown to be involved in the promotion of self-renewal ability, maintenance of stemness, and suppression of senescence in vitroin MSCs derived from different tissue sources7-9. The supplementation of bFGF in culture medium enhanced the proliferation of BMSCs and periodontal ligament stem cells, and preservedtheir differentiation ability10-12.In addition, BMSCs grown under the influence of bFGF have displayed to maintain their stem cell features with enhanced proliferation rate and a greater propensity towards osteogenesis and chondrogenesis5, 13-14.
To further understand the influence of bFGF on BMSCs for cell-based therapeutic applications, we investigated its supplementation at 0, 5 and 10 ng/mL on selected biological and phenotypic characteristics, such as morphology, viability, proliferation rate and doubling time, colony-forming unit-fibroblast (CFU-F) ability, genetic stability, senescence activity and cell-surface markers expression. The results might offer additional support to the prospective utility of ex vivo expanded BMSCs supplemented with bFGF for cartilage tissue regeneration.
MATERIALS AND METHODS
Isolation and culture of BMSCs
Bone marrow sample collection and in-vitro procedures were approved by the Institutional Ethics Committee and Institutional Committee for Stem Cell Research (IC-SCR). Human bone marrow aspirates were collected and processed after obtaining informed consent from patients (n=3), who were undergoing knee arthroscopy procedure for ligament injury with cartilage damage. Under local anaesthesia, about 15-20 mL of bone marrow suspension was harvested from the posterior iliac crest and collected in a 50 mL tube containing the same volume of heparinized (10 U/mL) phosphate-buffered saline (PBS, Gibco, Life Technologies, Grand Island, NY, USA).
The establishment of cell cultures was performed by following previously published protocol with minor modifications15. Briefly, mononuclear cells from bone marrow aspirate were collected by density gradient solution (Ficoll-Paque PLUS; 1.077 g/mL, GE Healthcare Life Sciences, Uppsala, Sweden). The cells were washed twice in PBS and culturedin Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Life Technologies) with 10% fetal bovine serum (FBS, Gibco, Life Technologies), 100 U/mL penicillin, and 100 μg/mL streptomycin (Gibco, Life Technologies) at 37°C in a humidified atmosphere and 5% CO2 in air.For culturing the adherent cells, the basal medium consisting of DMEM, 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin without (Control) or with bFGF (5 ng/mL and 10 ng/mL, Biolegend, CA, USA) was changed twice a week until the cells reached 80-90% confluency.Once reached confluency, all BMSCs were dissociated using a 0.25% (w/v) trypsin-ethylene-diaminetetraacetic acid (EDTA)(Gibco, Life Technologies) and sub-passaged five times for subsequent analyses.
Morphology
The morphology of BMSCs was assessed using phase-contrast microscope (Olympus, Tokyo, Japan) and any changes in morphological features during different time points of culture and also at various passages were recorded.
Viability assay
Cell viability was performed by 0.4% trypan blue (Gibco, Life Technologies) exclusion assay with a hemocytometer. The viability was assessed at every sub-passage of BMSCs.
Proliferation rate and population doubling time (PDT)
BMSCswere plated at 2×103 cells/cm2 in 12-well plates in a basal medium and cultured for 12 days. The culture medium was refreshed every 3 days. Cells in every three wells were detached with 0.25% trypsin-EDTA on day 3, 6, 9 and 12, and counted using a hemocytometer. The average cell numbers for every three wells in every replicate were determined and the proliferation rate was calculated. PDT was calculated using a formula; PDT= t(log2) / (log Nt-log No), where t represents culture time, and No indicate the cell numbers before seeding and Ntrepresent the cell numbers after seeding.
Colony-forming unit-fibroblast (CFU-F) ability
CFU-F ability of BMSCs was determined by seeding at a cell density of 0.5×103cells per well in 6-well culture plate, and cultured for 2 weeks with regular change of a fresh medium at every 3 days intervals. The cells were subsequently fixed in ice-cold ethanol for 5 min and the assay was performed using Crystal violet (Sigma-Aldrich, St. Louis, MO, USA) staining. All stained colonies which were made up of more than 25 cells were recorded as CFUs.
Senescence activity
The senescence activity in BMSCs was analyzed using senescence associated (SA)-β-Gal staining kit (Cell Signaling Technology, MA, USA) by following the manufacturers’ instructions. Briefly, the cells were washed with PBS and incubated for 15 min with 3.7% formaldehyde (Sigma-Aldrich), and again washed twice with PBS. Color development was observed by incubating the cells for overnight at 37°C with the kit-supplied staining solution (40mM citric acid/sodium phosphate pH 6.0, 150 mMNaCl, 2 mM MgCl2, 5 nM potassium ferricyanide, and 1 mg/ml X-gal in dimethyl sulphoxide). Cell culture plates were then observed for the development of blue colour by microscopic examination.
Karyotype analysis
Genetic stability was assessed by GTG-banded karyotype on metaphase spreads from cultured BMSCs at passage 4. GTG banding was carried out using trypsin (0.05%) and 1% Giemsa stain. Well banded metaphase spreads were analyzed using a fluorescence microscope (Olympus, Tokyo, Japan).
Phenotypic marker analysis by flow cytometry
Standard flow cytometry analysis was employed for determining the cell surface marker profile of BMSCs. Cells were analyzed for the expression of MSC-specific markers (CD29, CD73, and CD90) and the absence of CD34 and CD45 markers using Fluorescence-Activated Cell Sorter (FACSCalibur, Becton Dickinson, NJ, USA). Briefly, BMSCs at ~80% confluence were washed twice in PBS and incubated with 0.1% bovine serum albumin (Sigma-Aldrich) for 30 min to block nonspecific binding. Then, the cells were incubated with unconjugated anti-human mouse antibodies, such asCD29 (eBioscience, CA, USA, 1:100), CD73 (Biolegend, 1:100), CD90 (eBioscience, 1:100), CD34 (Biolegend, 1:100) and CD45 (eBioscience, 1:100) for 2 hrs at 37°C. Cells were washed twice in cell staining buffer (Biolegend), and incubated with fluorescein isothiocyanate (FITC)-conjugated anti-mouse IgG (eBioscience, 1:100) for 1 hr at room temperature. A minimum of 10,000 FITC-labeled cells were acquired (Forward scatter/Side scatter)and analyzed with Cell Quest software (Becton Dickinson). The corresponding isotype-matched (negative) control (eBioscience) was also used. The experiment was performed in duplicates and average data were obtained.
Statistical analysis
All data were expressed as the mean ± SD from at least three independent experiments. SPSS software (SPSS Inc, Chicago, USA) was used for one way analysis of variance (ANOVA) with Tukey’s post-hoc test. P values less than 0.05 were considered significant.
RESULTS
Primary culture establishment and morphology
MSCs were successfully isolated from low-density mononuclear cell population of bone marrow based on their selective attachment to plastic culture dishes when compared to hematopoietic cells. BMSCs at primary culture in basal medium were supplemented with bFGF (5 and 10ng/mL) in comparison to control (0 ng/mL). Readily adhered BMSCs reached 80-90% confluency in 15 days and used for analyses until passage 5.
BMSCs showed slightly elongated and fibroblast-like morphology in control (0 ng/ml bFGF, Figure 1A). Whereas in bFGF supplemented groups, cells showed small, spindle-shaped morphology (5ng/mL bFGF, Figure 1B and 10 ng/mL bFGF, Figure 1C). BMSCs supplemented with 10ng/mL bFGF exhibited more homogeneous and denser cell population than 5 ng/mL bFGF, despite exhibiting similar morphological features.
Viability, proliferation rate and PDT
Viability of BMSCs from all the groups at different passages was determined and any changes in viability during culture expansion were also recorded. No significant differences in percentage viability between control and bFGF supplemented BMSCs were observed. Cell viability was found more than 95% in all the three groups (Figure 2A). The proliferation of BMSCs in the initial days of culture (Day 0 to Day 3) was comparatively slow but increased remarkably from Day 6 to Day 12 (Figure 2B). A significant(p=0.001)difference was observed in terms of proliferation rate between 10 ng/mL bFGF and control BMSCs, whereas no significant (p=0.08)difference was found between 5 ng/mL bFGF and control. Moreover, no significant (p=0.17) difference in PDT was observed among all BMSCs, although PDT was slightly lower in 10 ng/mL bFGF supplemented group (Figure 2C).
Colony forming unit-fibroblast (CFU-F) ability
CFU-F ability of BMSCs between control and bFGF supplemented groups were determined at passage 3. Crystal violet stained colonies were recorded in 6-well culture plates with an initial seeding density of 0.5×103 cells/well.CFU-F assay was performed on 14th day of culture, and a higher number of colonies (more than 25 cells) was visualized in bFGF supplemented groups (both in 5 ng/mL bFGF and 10 ng/mL bFGF) than in control (Figure 3A-C).
Senescence associated β-galactosidase (SA-β-Gal) staining
BMSCs at passage 5 were subjected for senescence activity by SA-β-Gal assay and the results were recorded microscopically. BMSCs cultures showed a significantly lower number of senescent cells in bFGF supplemented groups when compared to control (Figure 4). However, no significant (p=0.10) difference was observed between 5ng/mL and 10 ng/mL bFGF supplemented BMSCs.
Karyotyping
Genetic stability of BMSCs was analyzed by GTG-banding. BMSCs from control and bFGF supplemented groups showed normal karyotype without any chromosomal aberrations (Figure 5).
Phenotypic marker analysis
BMSCs were phenotypically characterized by flow cytometry analysis with a panel of five markers at passage 3 after culture with or without bFGF. Flow cytometry analysis showed that the BMSCs from all the groups had a positive expression of MSC-specific markers, such as CD29, CD73 and CD90 in contrast to hematopoietic cells markers, CD34 and CD45, which showed<1% expression (Figures 6 & 7). CD29 and CD73 showed significantly (p<0.05) higher expression in bFGF supplemented BMSCs than in control. Whereas, CD90 had lower expression in BMSCs cultured with bFGF (Figure 7). Data indicated that BMSCs supplemented with 10 ng/mL bFGF showed slightly higher expression of MSC-specific markers.
DISCUSSION
The application of effective cytokines and growth factors to maintain the self-renewal ability and induce specific cell lineage of MSCs is one of the important factors in cell-based therapy. It is known that bFGF is involved in the stimulation of proliferation, protection from senescence, apoptosis and genetic damages, and conservation of multilineage differentiation ability of BMSCs5. Moreover, production of large scale MSCs is required for clinical applications in regenerative medicine16. During this process, it is necessary to ensure that BMSCs preserve the biological properties, such as morphology, growth, self-renewal ability, genetic stability and marker expression specific to BMSCs. Hence, the present study evaluated the effect of bFGF on in vitro culture of BMSCs derived from patients with cartilage disorders as a promising source for autologous therapy.
In this study, BMSCs cultured under the influence of bFGF exhibited a small, spindle-shaped features, while cells grown without bFGF showed elongated fibroblast-like morphology. These observations are in agreement with previous studies5,13. Nawrocka et al.7 recorded similar results in adipose-derived MSCs, where bFGF supplemented at 5ng/mL and 10ng/mL showed thinner, spindle-shaped, reduced cytoplasm and achieved higher cell densities as compared to control cells. Another study on placenta-derived MSCs reported a smaller sized, stronger plastic-adhered morphology in bFGF supplemented group, while bigger sized, less defined, polygonal-shaped cells in non-supplemented group16. It is suggested that these results depend on the type and source of MSCs used in the study.
Cell death during in vitro culture is a common phenomenon which may be due to minor manipulation of cells by culture media, expansion conditions, trypsinization etc. and any reduction in viability leads to a detrimental effect on cell biology and therapeutic properties. In this study, cell viability was observed above 90% in bFGF supplemented and control groups with no significant difference in values. Similarly, a previous study demonstrated that bFGF had no significant effect on cellular viability of BMSCs grown in spheroid culture system17. Also, Zeng et al.18 reported that 10ng/mL bFGF supported the proliferation of MSCs derived from gingiva by increasing the cell viability17.
In general, in vitro expansion of BMSCs reduces the replicative potential. In the present study, utilization of bFGF (both 5 ng/mL and 10 ng/mL) supported the replication of BMSCs by amplifying the proliferation rate and lowering the time required for population doubling. During 12 days of culture under the influence of bFGF, slow proliferation was recorded in initial days, which increased drastically from day 6 onwards and proved its mitogenic action on BMSCs. Earlier studies have also reported the role of bFGF in augmenting the proliferation rate of BMSCs5,14,19.Although doubling time was slightly lowered by bFGF, it was not significant in this study as observed previously on lowering of PDT with higher proliferation12.
BMSCs are capable of forming colonies when seeded at low density and colony-forming ability is one of the salient features of MSCs. In this study, supplementation of bFGF increased the colony-forming efficiency of BMSCs as compared to control. It has been documented that bFGF has a role in enhancing the colony formation ability of MSCs along with increase in size of colonies7, 14-16, 20. Moreover, when BMSCs expand ex vivo for a longer period, they undergo reduction in proliferation potential due to replicative stress and reach the senescent state with growth arrest, but remain metabolically active 3,21.At present SA-β-Gal staining assay is one of the well-established methods to assess cell senescence activity. In the present study, we observed that senescence activity of BMSCs was considerably decreased under the influence of bFGFwhen compared to control. Supplementation of 5 ng/mL bFGF to the culture markedly lowered the number of senescent cells, and no difference was observed with the addition of 10ng/mL bFGF. Earlier results highlighted that bFGF suppresses the MSCs cellular senescence depending on the length of culture through down-regulation of transforming growth factor-beta2 expression8. It appears that BMSCs cultured with bFGF show minimal senescent cells, and as a safety measure, the expansion should be reduced as much as possible3.
Ex vivo expansion of MSCs in the laboratory for a longer period is more prone to genetic alterations, mutations, deletions and other chromosomal abnormalities3, 6. Cells with genetic defects can lead to transformation, which is highly undesirable for their use in regenerative and tissue engineering applications. Therefore, it was essential to assess the effect of bFGF supplementation on the cytogenetic stability of BMSCs. We observed that BMSCs had a normal karyotype and did not carry any chromosomal abnormalities in both bFGF supplemented and control groups. Thus, it was found safer to use bFGF in culture medium in-vitro as a growth factor to obtain the ideal characteristics of BMSCs.
MSCs are known to express specific surface markers, such as CD29, CD73, CD44, CD90, and CD105, but not hematopoietic cell markers, including CD14, CD34 and CD4520-21. In this study, the addition of bFGF in expansion media modified the immunophenotype of BMSCs, corresponding to the concentrations used. Amongst the analyzed markers CD29 and CD73 showed significantly higher expression in bFGF supplemented groups, but not for CD90. Both hematopoietic markers, CD34 and CD45, were not altered by the addition of bFGF. However, BMSCs displayed a typical feature of bearing positive markers and lacking hematopoietic cell markers. Previously, Zenget al18 showed an increase in STRO-1 marker expression in gingival tissue-derived MSCsby bFGF. A study on BMSCs showed that bFGF had no effect on the expression of cell surface markers, except for CD29 and HLA-DR14. On a similar note,no effect of bFGF on CD44 expression was observed in Wharton’s jelly-derived MSCs22. Supplementation of bFGF at 10 ng/mL could be considered an optimal and efficient concentration for expanding BMSCs in culture before their use in cell-based therapeutic applications.
CONCLUSION
Our study findings showed an important role of bFGF in in-vitro culture of BMSCs. Compared to the supplementation of 5 ng/mL bFGF, BMSCs cultured with 10ng/mL bFGF resulted in enhanced proliferation and colony-forming efficiency, prevention of cells from senescence, and maintenance of cytogenetic stability and higherexpression of selected surface markers. Further evaluation on preservation of multilineage differentiation ability especially towards chondrogenesis for the clinical use of BMSCs expanded by bFGF could be further investigated.
Acknowledgements
This work was supported by NUFR3 grant (2016-2019) from Nitte (Deemed to be university).Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Conflicts of interests
The authors declare that there are no conflicts of interest
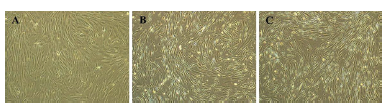
Figure 1. Morphology of BMSCs. Cells exhibited elongated fibroblast-like morphology prominently in control (0 ng/mL bFGF, A), whereas, a small, spindle-shaped morphology was observed in bFGF supplemented groups (5 ng/mL and 10 ng/mL, B and C, respectively). Images magnification: 4x.
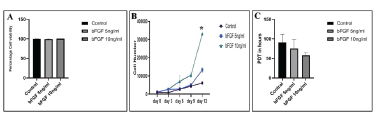
Figure 2. Viability, proliferation rate and PDT of BMSCs. A. Percentage cell viability between the control and bFGF supplemented BMSCs showed no significant differences (p=0.152). B. A significant difference was observed in terms of proliferation rate between 10 ng/mL bFGF supplementation and control (*, p=0.001). But, no significant difference was found between 5 ng/mL bFGF and control (p=0.084). C. In PDT, no significant difference was observed between the control and bFGF supplemented groups (p=0.174).
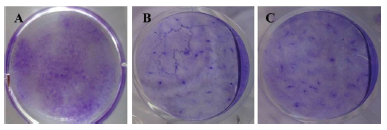
Figure 3. Colony-forming unit-fibroblast (CFU-F) analysis of BMSCs. Macroscopic images showing the ability of CFU-F which was higher in bFGF (5 ng/mL and 10 ng/mL) supplemented BMSCs (B & C) than in control (A) as assessed by 1% Crystal violet staining.

Figure 4.Senescence activity in BMSCs.A significant difference in senescence was observed in BMSCs of control (0 ng/mL) when compared to bFGF supplemented with 5 ng/mL (p=0.014) and also 10 ng/mL group (p=0.0012). However, no significant difference was observed between (5 ng/mL) and 10 ng/mL groups (p=0.104). Superscripts a and b indicate statistical difference.
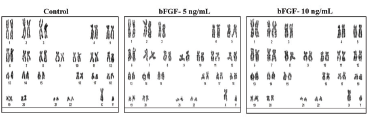
Figure 5. Karyotyping of BMSCs. Genetic stability was determined by GTG-banding karyotype and the chromosomal spreads exhibited a normal ploidy in BMSCs at passage 4 cultured without (control) or with bFGF supplementation.
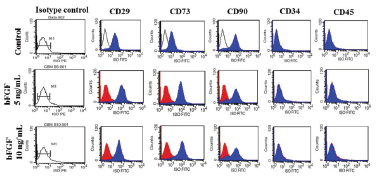
Figure 6. Cell surface marker analysis of BMSCs by flow cytometry. Blue histograms represent the extent of positivity of cells for the markers analysed as indicated. Dark lines or red histograms represent isotype control. All BMSCs showed the positivity for markers, such as CD29, CD73 and CD90 and negative for CD34 and CD45.
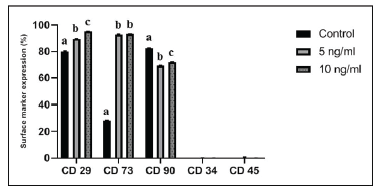
Figure 7.Percentage expression of cell surface markers in BMSCs by flow cytometry. Mean percentage value of each surface antigen showed the significant difference in the expression of markers as indicated by the superscripts a, b, and c (p<0.05). Expression of CD34 and CD45 markers was less than 1% in all BMSCs.
References:
-
Steens J, Klein D. Current strategies to generate human mesenchymal stem cells in vitro. Stem Cells Int 2018; 2018:6726185.
-
Kalomoiris S, Cicchetto AC, Lakatos K, Nolta JA, Fierro FA. Fibroblast growth factor 2 regulates high mobility group a2 expression in human bone marrow-derived mesenchymal stem cells. J Cell Biochem 2016; 9999:1–10.
-
Neri S. Genetic stability of mesenchymal stromal cells for regenerative medicine applications: A fundamental biosafety aspect. Int. J. Mol. Sci2019; 20:2406.
-
Solchaga LA, Tognana E, Penick K, Baskaran H., Goldberg VM, Caplan AI, et al. A rapid seeding technique for the assembly of large cell/scaffold composite constructs. Tissue Eng 2006; 12:1851.
-
Solchaga LA, Penick K, Goldberg VM, Caplan AI, Welter JF. Fibroblast growth factor-2 enhances proliferation and delays loss of chondrogenic potential in human adult bone-marrow-derived mesenchymal stem cells. Tissue Eng Part A2010;16:3:1009-19.
-
Behrens A, Van Deursen JM, Rudolph KL, Schumacher B. Impact of genomic damage and ageing on stem cell function. Nat Cell Biol 2014; 16:201–207.
-
Nawrocka D, Kornicka K, Szydlarska J, Marycz K. Basic fibroblast growth factor inhibits apoptosis and promotes proliferation of adipose-derived mesenchymal stromal cells isolated from patients with type 2 diabetes by reducing cellular oxidative stress.Oxid Med Cell Longev 2017; 2017: 3027109.
-
Ito T, Sawada R, Fujiwara Y, Seyama Y, Tsuchiya T. FGF-2 suppresses cellular senescence of human mesenchymal stem cells by down-regulation of TGF-β2. BiochemBiophys Res Commun 2007; 359:108-14.
-
Wang R, Liu W, Du M, Yang C, Li X, Yang P. The differential effect of basic fibroblast growth factor and stromal cell-derived factor-1 pretreatment on bone morrow mesenchymal stem cells osteogenic differentiation potency. Mol Med Rep 2018 Mar; 17(3):3715-3721.
-
Tasso R, Gaetani M, Molino E, Cattaneo A, Monticone M, Bachi A, et al. The role of bFGF on the ability of MSC to activate endogenous regenerative mechanisms in an ectopic bone formation model. Biomaterials 2012; 33(7):2086–96.
-
Zhang C, Guo H, Yang, Chen, Huang, Liu, et al. The biological behaviour optimization of human periodontal ligament stem cells via preconditioning by the combined application of fibroblast growth factor-2 and A83-01 in in vitro culture expansion. J Transl Med 2019; 28:17(1):66.
-
Solchaga LA, Penick K, Porter JD, Goldberg VM, Caplan AI, Welter JF. FGF-2 enhances the mitotic and chondrogenic potentials of human adult bone marrow-derived mesenchymal stem cells. J. Cell. Physiol 2005; 203(2):398-409.
-
Song, G, Ju Y, Soyama H. Growth and proliferation of bone marrow mesenchymal stem cells affected by type I collagen, fibronectin and bFGF.MaterSciEng C2008; 1467–1471.
-
Lee JS, Kim SK, Jung BJ, Choi SB, Choi EY, Kim CS. Enhancing proliferation and optimizing the culture condition for human bone marrow stromal cells using hypoxia and fibroblast growth factor-2. Stem Cell Res 2018 Apr; 28:87-95.
-
Park BW, Kang EJ, Byun JH, Son GM, Kim JH, Hah YS, et al. In vitro and in vivo osteogenesis of human mesenchymal stem cells derived from skin, bone marrow and dental follicle tissues. Differentiation 2012; 83(5):249?259.
-
Vellasamy S, Vidyadaran S, George E, Ramasamy R. Basic fibroblast growth factor enhances the expansion and secretory profile of human placenta-derived mesenchymal stem cells. Malaysian J Med Health Sci 2016; 12: 49-59.
-
Tae JY, Ko Y, Park JB. Evaluation of fibroblast growth factor-2 on the proliferation of osteogenic potential and protein expression of stem cell spheroids composed of stem cells derived from bone marrow. Exp Ther Med 2019; 18(1):326-331.
-
Zeng S, Fang Y, Zhang Q, Peng B, Zhang Z, Zhao W, et al. Effect of basic fibroblast growth factor on the gingiva-derived mesenchymal stem cells.Int J Clin Exp Med 2019; 12(5):6347-6356.
-
Karlsen TA, Brinchmann JE. Expression of inflammatory cytokines in mesenchymal stromal cells is sensitive to culture conditions and simple cell manipulations. Exp Cell Res 2019; 1; 374(1):122-127.
-
Wu J, Huang GT, He W, Wang P, Tong Z, Jia Q, et al. Basic fibroblast growth factor enhances stemness of human stem cells from the apical papilla.J Endod 2012; 38(5):614-22.
-
Park J, Lee J, Yoon BS, Jun KE, Lee G, Kim YI, et al. Additive effect of bFGF and selenium on expansion and paracrine action of human amniotic fluid-derived mesenchymal stem cells. Stem Cell Res Ther 2018; 9:293.
-
Nekanti U, Mohanty L, Venugopal P, Balasubramanian S, Totey S, Ta M. Optimization and scale-up of Wharton’s jelly-derived mesenchymal stem cells for clinical applications. Stem Cell Res 2010; 5(3):244-54.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License