IJCRR - 10(10), May, 2018
Pages: 35-38
Date of Publication: 30-May-2018
Print Article
Download XML Download PDF
Phylloides Tumor - A Study of Spectrum of its Histomorphological Parameters along with Ki-67 Expression
Author: Manish Gupta, Mustafa Ali
Category: Healthcare
Abstract: Objective: Aim of the study was to evaluate different parameters that are used to categorise phylloides tumor into benign, borderline and malignant type and also to know frequency of Ki-67 expression in different category.
Method: It was a prospective study of two year duration carried out in the department of pathology, King George's medical university and included total 30 cases.
Results: Most of patients in the entire category were more than 35 years of age and also severity of all the parameters increased as tumor spectrum shift from benign to malignant side.
Conclusion: Patient age and tumor size increases as the lesions progress from benign to malignant phyllodes tumor so is the ki-67 expression. Also Ki-67 antigen is a useful adjunct in the diagnosis and prognosis of phylloides tumor.
Keywords: Phylloides, Histomorphology, Ki-67 antigen
Full Text:
INTRODUCTION
Phyllodes tumor which derives from the greek word “phullon” or leaf are group of biphasic fibroepithelial tumors of breast of varying malignant potential ranging from benign tumor to fully malignant sarcomas. They were first fully characterized by Johannes Muller in 18381 and constitute 0.3% to 1% of all breast neoplasm. The term cystosarcoma phyllodes was initially described by Muller based on the tumor's “leaflike” projections in to the cystic spaces, sarcomatous stroma and “fleshy” gross appearance. This term has since been discouraged because greater than 70% of these lesion follow a benign course and only rarely exhibit cystic degeneration; phyllodes tumor, coined by the world health organization (WHO) in 1981, is now the preferred term.2 In 2003, WHO divided breast phyllodes tumor in three groups as benign, borderline and malignant, based up on the following criteria3 1. Stromal overgrowth 2. Stromal cellularity 3. Nuclear pleomorphism 4. Mitotic activity 5. Microscopic tumor border (circumscribed or infiltrating). Depending upon the above criteria benign tumors were classified when there was moderate stromal cellularity, mitotic figures were few if any per 10 high power field, well circumscribed border and absence of or mild nuclear pleomorphism. On the other hand malignant phyllodes tumor were diagnosed when they showed extensive stromal overgrowth, marked stromal cellularity, more than 10 mitotic figures per 10 high power field, infiltrating border and marked nuclear pleomorphism. Borderline phyllodes tumors were more atypical than benign but did not fulfil all the criteria of malignancy. All grades of phyllodes tumor have the potential of local recurrence, but the metastasis is mostly identified in borderline and malignant grades. Although a grading system has been set up, the criteria of this system are not rigidly defined and vary widely among the pathologist and it is often difficult to predict the clinical behaviour from the histology alone. In addition to the routine histomorphology, the study of Ki-67 antigen expression may be useful to differentiate benign, borderline and malignant phyllodes tumor and are also helpful in predicting the clinical behaviour.
MATERIAL AND METHODS
It is a prospective study of two year duration that included all the cases diagnosed as phylloides in Department of pathology and then were assessed and compared by histomorphological parameters used in the diagnosis. Finally Immunohistochemistry using Ki-67 was performed and its expression was recorded. Positive tissue control (Lymphoma for Ki-67) and negative control (Omission of primary antibody) were taken.
Hematoxylin and Eosin staining procedure: (Standard protocols were used)
Immunohistochemistry: (Standard protocols were used)
Primary Antibody Dako, Monoclonal Mouse Antibody Anti-Human Ki-67 Antigen, product code: IS626; Ready-to-use and Anti-Human CD10, Product code: IS648; Ready-to-use)
Interpretation of the Ki-67 immunostaining:
Presence of brown colored end product at the site of target antigen was indicative of positive reactivity. KI-67 antigen is expressed in the nucleus and Ki-67 labelling index is defined as the percentage of cells that showed a positive nuclear stain. At least 1000 stromal cells were counted for this analysis, and all Ki-67 indices were determined by one observer.
Statistical Analysis
The results are presented in Mean±SD (standard deviation), Median and Percentages. The dichotomous / categorical variables are compared by using Chi-square test. The one way analysis of variance (ANOVA) test is used to compare the continuous variables among the final diagnosis. The Tukey's multiple comparison test is used for pair wise multiple comparison between two groups, if found significant in ANOVA test analysis. The p-value <0.05 is considered as significant. All the analysis is carried out by using SPSS (statistical package for the social sciences) 16.0 version.
RESULTS
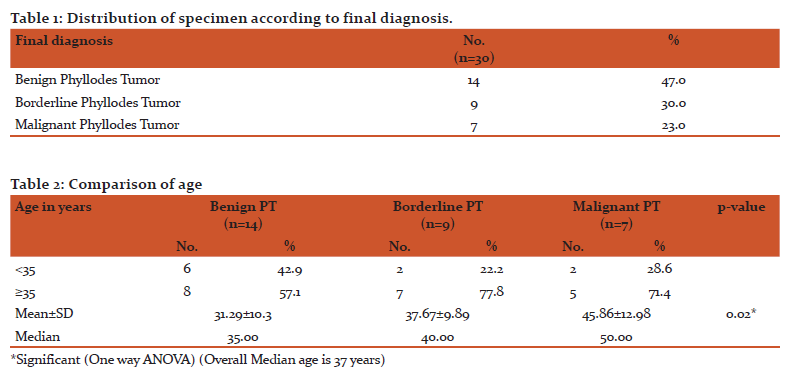
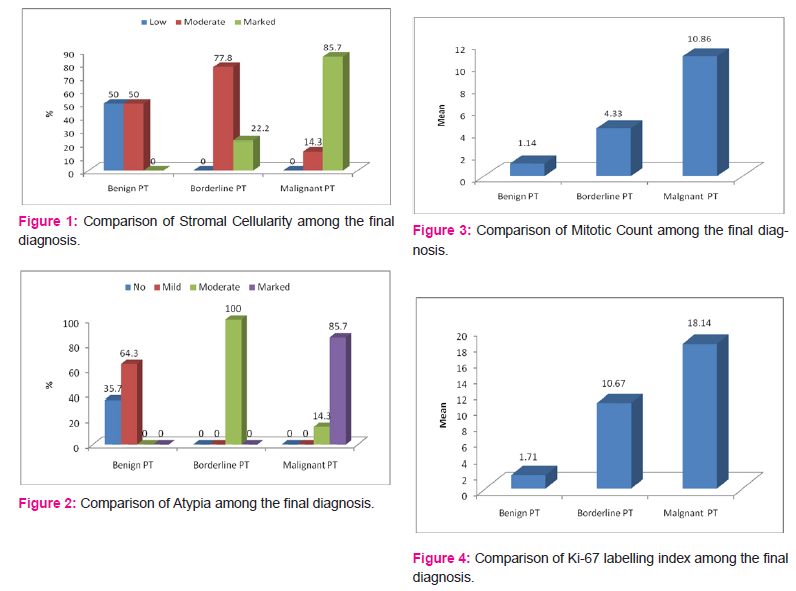
In our study out of 30 cases, maximum number of cases belonged to benign phyllodes tumor, 14 cases (47%) whereas Borderline and Malignant phyllodes tumor constituted 9 and 7 cases (30% and 23%) respectively. More than half of the patients in benign phyllodes tumor (57.1%), borderline phyllodes tumor (77.8%) and malignant phyllodes tumor (71.4%) were more than 35 years of age. The age was significantly different among the groups (p=0.02). The post-hoc test revealed that the age was significantly (p=0.01) different between benign phyllodes tumor (31.29±10.30) and malignant phyllodes tumor (45.86±12.98). There was significant difference (p=0.0001) in the tumor size among benign phyllodes tumor (5.57±3.71), borderline phyllodes tumor (10.22±2.86) and malignant phyllodes tumor (16.43±3.99). Majority of cases of benign phylloides showed mild cellularity, borderline cases showed moderate cellularity, while malignant cases had marked cellularity in most of cases. The comparison of atypia among the various category showed that mild atypia was present in 64.3% of benign phyllodes tumor, moderate atypia was in all the patients of Borderline phyllodes tumor and 14.3% in malignant phyllodes tumor. Marked atypia was found in 85.7% in malignant phyllodes tumor. The association between atypia and final diagnosis was statistically significant (p=0.0001). The Mitotic Counts were significantly different in benign phyllodes tumor (1.14±1.09), Borderline phyllodes tumor (4.33±1.00) and malignant phyllodes tumor (10.86±0.69). The Ki-67 labelling index was significantly (p=0.0001) lower in Benign phyllodes tumor (1.71±0.82) compared with Borderline (10.67±6.20) and Malignant phyllodes tumor (18.14±9.26).
DISCUSSION
Our result demonstrated the several findings. Firstly, the mean age of patient in benign, borderline and malignant phyllodes tumor were 31.39, 37.67 and 45.86 years respectively and this difference was found to be statistically significant (p=0.02). Thus the mean age of patient increases as we progress from benign to malignant phyllodes tumor. Similar results were also noted by Tse GM et al 4 and K.S. Kim et al 5 in their studies. Mean tumor size in our study was 5.57, 10.22 and 16.43cm respectively in benign, borderline and malignant phyllodes tumor and this difference was statistically significant. This increasing in tumor size from benign to borderline to malignant phyllodes tumor was also found in the studies done by Tse GM et al and Ili? I, et al.6 Stromal cellularity, atypia and mitotic counts increased as lesions progresses from benign towards malignant category and the difference was statistically significant. Ki Beom Ku et al 7 and Timothy W. Jacobs et al 8 have also mentioned similar findings in their studies. Important finding in our study is that mean Ki-67 labelling index was lower in Benign phyllodes tumor (1.71) compared with Borderline (10.67) and Malignant phyllodes tumor (18.14) and this difference was statistically significant (p=0.0001). There are many studies which support this finding of our study. In one study done by Yu-Jan Chan et al 9 in year 2004, immunohistochemical analysis of 63 phyllodes tumor cases (50 benign and 13 malignant) were done and compared for the positive staining of tumor cell with the tumor grading and clinical outcome. They found that Ki-67 antigen was well correlated with tumor grading. In another study by Nicole Nicosia Esposito et al 10 the mean Ki-67 scores for benign, borderline and malignant phyllodes tumor were 3%, 26% and 32% respectively. Thus Ki-67 was differentially expressed among different tumor grades (p<0.001) but it did not differentiate between borderline and malignant phyllodes tumor (p>0.99). In few other studies by Y Singh et al 11 in 1996 and Umekita Y et al 12 in 1999 have also found the significant difference in the expression of Ki-67 antigen in benign, borderline and malignant phyllodes tumor, thus further supporting the findings of our study.
CONCLUSION
Patient age and tumor size increases as the lesions progress from benign to borderline to malignant. Ki-67 antigen in combination with other histomorphological parameters also increases in severity and helps in predicting the clinical behaviour and in distinguishing the benign, borderline and malignant phyllodes tumor.
ACKNOWLEDGEMENT
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Conflict of interest: None
Source of Funding: None
References:
1. Muller J, Ueber den feinen bau unddie furmen der krankhafte Geschwulste. Berlin: G Reimer; 1838; 1: 54-57.
2. Tavassoli FA, Devilee P, et al. Pathology and Genetics of Tumors of the Breast and Female Genital Organs. Lyon, France: International agency for research on cancer; 2003.
3. Fattaneh A, Tavassoli FA, Devilee P. WHO classification of tumors: Tumors of the breast and female genital organs. IARC, Lyon, pp 100-103, 2003.
4. Tse GM, Tsang AK, Putti TC, Scolyer RA, Lui PC, Law BK, Karim RZ, Lee CS. Stromal CD10 expression in mammary fibroadenomas and phyllodes tumors. J Clin Pathol. 2005; 58(2):185-189.
5. K.S. Kim. Correlation of CD10 and EGFR Expression in Phyllodes Tumors of the Breast. Ann Oncol (2012)23 (suppl 2): ii17-ii24.
6. Ivan Ili?, Pavle Ran?elovi?, Ratko Ili?, Vuka Kati?, Maja Milentijevi?, Ljubinka Veli?kovi?, Miljan Krsti?. An approach to malignant mammary phyllodes tumors detection. Vojnosanit Pregl .2009; 66(4): 277–282.
7. Ki Beom Ku, Mi Ji Bang, Jong Woo Choi, Yoon Sik Lee, Jin Hyun Park, Hyun Ok Kim, Min Hee Jeong, Hoon Gyu Oh, and Jin Gu Bong. Clinical, Pathologic and Immunohistochemical feature of Phyllodes Tumors of the Breast. J Breast Cancer. 2004; 7(3):185-192.
8. Timothy W. Jacobs, Yunn-Yi Chen, Donald G. Guinee, Joseph A. Holden, Imok Cha, Donald E et al. Fibroepithelial Lesions with Cellular Stroma on Breast Core Needle Biopsy. Are There Predictors of Outcome on Surgical Excision? Am J Clin Pathol 2005;124:342-354.
9. Yu-Jan Chan, Be-Fong Chen, Chin-Long Chang, Tsen-Long Yang, Chi-Chen Fan. Expression of p53 protein and Ki-67 Antigen in Phyllodes Tumor of The Breast. J Chin Med Assoc.2004; 67:3-8.
10. Nicole Nicosia Esposito, Deepak Mohan, Adam Brufsky, Yan Lin, Malathy Kapali, David J. Dabbs. Phyllodes Tumor: A Clinicopathologic and Immunohistochemical study of 30 cases. Arch Pathol Lab Med. 2006; 130:1516–1521.
11. Y Singh, T Hatano, Y Uemura, N Shikata, H Senzaki, K Hioki, A Tsubura. Immunohistochemical profile of phyllodes tumors of the breast. Oncology Reports.1996; 3(4):677-681.
12. Umekita Y, Yoshida H. Immunohistochemical study of MIB1 expression in phyllodes tumor and fibroadenoma. Pathology international.1999; 4 9(9):807-10.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License