IJCRR - 10(8), April, 2018
Pages: 25-27
Date of Publication: 28-Apr-2018
Print Article
Download XML Download PDF
Comparison of Somatostatin and Famotidine for the Treatment of Nonvariceal Acute Upper Gastrointestinal Bleeding
Author: Caglayan Geredeli
Category: Healthcare
Abstract:Introduction: It is aimed to be researched if somatostatin is as effective as H2 blockers to stop the bleeding in centres where the endoscopic treatment is not available for the non-variceal upper GI bleeding cases
Methods: The patients who were admitted to the Department of Internal Medicine of Dicle University with the diagnosis of upper GI bleeding, and were not suitable for endoscopic treatment are subjected in our paper. One group was given 250 mcg bolus dose of somatostatin followed by 120 hours of infusion at a speed of 250 mcg/hour whereas the other group was given 20 mg intravenous famotidine every 12 hours. The amount of time required to stop bleeding, the amount of blood transfusion made during that 120 hours time period, the ratio of recurrent bleeding, and the ratio of the necessity of surgical intervention were noted.
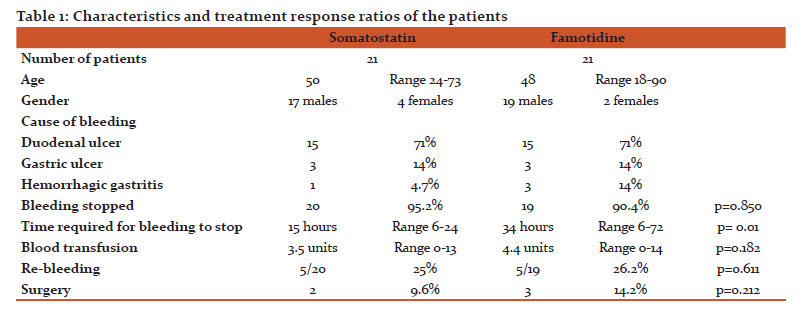
Results: 42 patients were included in the study. 21 patients were in somatostatin group, and the other 21 patients were in H2 receptor blocker group. In both somatostatin and H2 receptor blockers groups, the cause of gastrointestinal bleeding was found to be duodenal ulcers in 71% of the patients and gastric ulcer in 14% of the patients. While bleeding had stopped in the first 48 hours in 95.2% of the patients in somatostatin group, the same thing happened in 90.4% of the patients in H2 receptor group. There were no statistical difference between the two groups (p=0.90). The average time passed to stop the bleeding was 15 hours (6-24 hours) for the somatostatin group, whereas it was 34 hours (6-72 hours) for the H2 receptor blocker group. Somatostatin stopped the bleeding in a shorter time which was statistically significant (P=0.01).while the required blood transfusion amount was an average of 3.5 (0-13) units for the somatostatin group, it was 4.4 (0-14) units for the group of H2 receptor blocker. There were no statistically significant difference (p=0.182). The surgical intervention was needed in 9.6% of the patients in somatostatin group, whereas it was needed in 14.2% of the patients in H2 receptor group (p=0.212).
Conclusion: While somatostatin statistically significantly stops the bleeding in a shorter time period as compared with H2 receptor blockers, no significant difference in blood transfusion requirement and surgical necessity were detected. Cost-effectiveness of somatostatin should be considered when it is used in GI bleeding cases.
Keywords: Acute upper GI bleeding, Somatostatin, Famotidin
DOI: 10.7324/IJCRR.2018.10805
Full Text:
Introduction
Acute upper GI bleeding is a commonly seen health problem in the world(1). More than 100 of every 100000 patients who were admitted were found to have upper GI bleeding(1). 80% of upper GI bleeding stops spontaneously while 20% does not(1). Surgical intervention is required in 15% to 30% of the patients with an ongoing bleeding(1).
H2 receptor blockers, proton pump inhibitors and somatostatin are used in the medical treatment of upper GI bleeding(2,3). H2 receptor and proton pump inhibitors inhibit the acid secretion from stomach(3). Somatostatin, which is an endogenouspeptide that reduces the splenic blood flow, inhibits the motility and acid secretion of GI, and has a possible cytoprotective effect, is known to be effective in oesophageal variceal bleeding cases(4). However its effectiveness on non-variceal bleeding is uncertain. In our study, the comparison of the effectiveness of somatostatin in non-variceal bleeding with the routine H2 receptor blocker treatment was aimed.
Materials and Method
The study was conducted retrospectively at the Department of Internal Medicine of Dicle University. The patients who applied to the emergency service with the complaints of hematemesis and melena, and were undergone a gastroscopy in 24 hours following their admission to the internal medicine wards to find the cause of GI bleeding were selected. The patients who were older than 18 years old and had their arterial systolic blood pressures lower than 100mmHg, pulses higher than 100 and hematocrit levels below 30 were included. The patients who with serious coagulation defects and oesophageal variceal bleeding were excluded. The ones who were given famotidine (20 mg every 12 hours given intravenously) as treatment were selected for H2 receptor blocker group and the rest were given somatostatin (250 mcg bolus dose followed by 250mcg/h infusion for 72-120 hours). Both groups were given intravenous isotonic NaCl solution to stabilize them hemodynamically. The gastroscopy was repeated for the patients who stopped bleeding to confirm that the bleeding had stopped. The patients who stopped bleeding were then discharged 120 hours later with a prescription of famotidine 40mg. Patients' blood pressure and pulse rate were controlled hourly. Hematogram was performed every 6-12 hours to determine the need for blood transfusion. The patients whose blood pressure and pulse couldnot be stabilised in 24 hours and needed more than 8 units of blood transfusion were treated surgically. The comparison of the amount of time required for bleeding tobe stopped, the ratio of re-bleeding, the required amount of blood transfusion and the ratio of surgical intervention required was made.
Statistical Methods
The Statistical Package for the Social Sciences (SPSS) version 15.0 for Windows (SPSS Inc., Chicago, IL, USA) was used for the statistical analysis. The comparisons of the ratios in the groups were made using a chi-squared analysis and Mann-Wutnay U tests were used for the statistical analysis. P values smaller than 0.05 were decided to be evaluated as significant.
Results
42 patients were included in the study. 21 patients were given somatostatin, and 21 patients were given famotidine intravenously. Of the patients who received somatostatin treatment, 17 were male and 4 were female. 19 of the patients who were given famotidine were male whereas 2 of them were female. The mean age of the patients receiving somatostatin was 50 (24-73) while the mean age of the patients receiving famotidine was 48 (18-90). The causes of hemorrhage in both groups were similar (Table 1). Bleeding stopped in 95.2% of patients who were given somatostatin and 90.4% of patients who were given famotidine (Table 1). There was no statistical difference between the two groups by means of stoppage of the bleeding (p =0.850) The average time spent to stop the bleeding was 15 hours (6-24 hours) for the patients treated with somatostatin, whereas it was 34 hours (6-72 hours) for the ones treated with famotidine. There was a statistically significant difference between the two groups regarding the time required for the stoppage of bleeding (P=0.01). While the required blood transfusion amount was an average of 3.5 (0-13) units for the somatostatin receiving patients, it was 4.4 (0-14) units for the patients treated with famotidine. There was no statistically significant difference between the two groups in terms of blood transfusion (p=0.182). Re-bleeding developed in the 25% of the patients treated with somatostatin, whereas it re-occurred in 26.2% of the patients who were given famotidine. There was no statistically significant difference between the two groups in terms of rebleeding. (p=0.611) 9.6% of the patients treated with somatostatin needed surgical intervention, while 14.2% of the ones who were given famotidine did so. No statistical difference between the two groups with regards to surgical operation requirement was found (p=0.212). No mortality had been seen in neither of the groups during the 120 hours of admission.
Discussion
Somatostatin is effective in stopping the oesophageal variceal bleeding, but its effectiveness on non-variceal acute upper GI bleeding is controversial (5,6,7). Terres A et al, Antonioli A et al and Kayasseh et al has found somatostatin to be effective in stoppage of bleeding in upper GI bleeding cases. However in some other studies somatostatin is not found to stop the acute upper GI bleeding effectively (8-12). Our study showed that somatostatin effectively stops bleeding in cases with acute upper GI bleeding. In all the studies where the time required to stop the bleeding was evaluated (5,6,8,9) somatostatin was detected to decrease the bleeding time significantly. Our study also showed that the time required to stop the bleeding was 15 hours in cases where somatostatin was given, whereas it was 34 hours in the group that was given H2 receptor blockers. No significant difference between somatostatin and H2 blockers in terms of re-bleeding has been found in the studies made so far (8,10,11). In accordance with that, a significant difference couldnot be detected by means of re-bleeding in our study. Though some studies show that somatostatin decreases the need for blood transfusion in acute upper GI bleeding (6-8), a significant difference couldnot be detected in a few other studies. Our study also shows no significant difference between the two groups regarding blood transfusion (8-12). In some studies it has been detected that somatostatin decreases the requirement for surgical intervention (9,10). However, such effects of it couldnot be found in many other studies (5,7,9-12) An effect to reduce the necessity for surgical requirement couldnot be observed in our study as well.
Conclusion
To conclude, somatostatin had stopped the bleeding significantly and shortened the bleeding period significantly as compared with famotidine. However, no significant difference was found between the patients who were given somatostatin and H2 receptor blocker in terms of the ratio of re-bleeding, blood transfusion need and the necessity of surgical intervention.
Acknowledgements
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Source of funding: No funding source
Conflict of interest: No conflict of interest
References:
1.Longstreth GF. Epidemiology of hospitalization for acute upper gastrointestinal hemorrhage: a population-basedstudy. Am J Gastroenterol. 1995 Feb;90(2):206-10.
2.Walan A1, Bader JP, Classen M, Lamers CB, Piper DW, Rutgersson K, Eriksson S
Effect of omeprazole and ranitidine on ulcer healing and relapserates in patients with benign gastric ulcer. N Engl J Med. 1989 Jan 12;320(2):69-75.
3.Collins R, Langman M. Treatment with histamine H2 antagonists in acute upper gastrointestinal hemorrhage. Implications of randomized trials. N Engl J Med. 1985 Sep 12;313(11);660-6.
4.Imperiale TF, Birgisson S Somatostatin or octreotide compared with H2 antagonists and placebo in the management of acute nonvariceal upper gastrointestinal hemorrhage: a meta-analysis. Ann Intern Med. 1997 Dec 15;127(12);1062-71. Erratum in: Ann Intern Med 1998 Feb 1;128(3);245
5.Torres AJ, Landa I, Hernández F, Jover JM, Suárez A, Arias J, Cuberes R, Santoyo J, Fernández R, Calleja J, et al Somatostatin in the treatment of severe upper gastrointestinal bleeding: A multicentre controlled trial. Br J Surg. 1986 Oct;73(10):786-9.
6.Antonioli A, Gandolfo M, Rigo GP, Bianchi Porro G, Cheli R, Brancato F, Lazzaroni M, Parodi MC, Maringhini A, Raimondo S, et al Somatostatin and cimetidine in the control of acute upper gastrointestinal bleeding. A controlled multicenter study. Hepatogastroenterology. 1986 Apr;33(2):71-4.
7.Kayasseh L, Gyr K, Keller U, Stalder GA, Wall M Somatostatin and cimetidine in peptic-ulcer haemorrhage. A randomised controlled trial. Lancet. 1980 Apr 19;1(8173):844-6.
8.Galmiche JP, Cassigneul J, Faivre J, Tranvouez JL, Ouvry D, Colin R, Pascal JP, Klepping C. Somatostatin in peptic ulcer bleeding--results of a double-blind controlled trial.Int J Clin Pharmacol Res. 1983;3(5);379-87.
9.Basile M, Celi S, Parisi A, Castiglione N, Parisi S Somatostatin in the treatment of severe gastrointestinal bleeding from peptic origin. A multicentric controlled trial. Ital J Surg Sci. 1984;14(1):31-5
10.Coraggio F, Scarpato P, Spina M, Lombardi S Somatostatin and ranitidine in the control of iatrogenic haemorrhage of the upper gastrointestinal tract. Br Med J (ClinResEd). 1984 Jul 28; 289(6439):224.
11.Magnusson I, Ihre T, Johansson C, Seligson U, Törngren S, Uvnäs-Moberg K. Randomised double blind trial of somatostatin in the treatment of massive upper gastrointestinal haemorrhage. Gut. 1985 Mar;26(3):221-6.
12.Basso N, Bagarani M, Bracci F, Cucchiara G, Gizzonio D, Grassini G, Percoco M, Procacciante F, Toti F. Ranitidine and somatostatin. Their effects on bleeding from the upper gastrointestinal tract. Arch Surg. 1986 Jul;121(7):833-5.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License