IJCRR - 10(5), March, 2018
Pages: 56-59
Date of Publication: 15-Mar-2018
Print Article
Download XML Download PDF
Evaluation of Methyl Blue Sabouraud Dextrose Agar Medium for Differentiation of Candida dubliniensis from Candida albicans
Author: Abiroo Jan, Gulnaz Bashir, Asiya Yaqoob Beigh, Bashir A. Fomda, Nazir Ahmad Var, Nadeem Ahmed, Junaid Ahmed
Category: Life Sciences
Abstract:Background: Candida dubliniensis that was first identified as a new species by Sullivan et al. (1995) in Dublin, Ireland (and was subsequently named after its place of origin) while performing an epidemiological investigation of oral candidiasis in HIV-infected and AIDS patients in the early 1990s. This pathogenic Candida species shares many phenotypic features with Candida albicans which cause problems its identification. Several phenotypic based tests have been developed to distinguish C. albicans from C. dubliniensis but none has been demonstrated being sufficient alone for accurate differentiation of the two species.
Aim: To facilitate the differentiation of these species, we evaluated methyl blue Sabouraud dextrose agar medium.
Method: Two hundred Candida spp. were tested including 186 stock strains of C. albicans and 14 strains of C. dubliniensis. Identification of all these strains was confirmed by polymerase chain reaction-restriction fragment length polymorphism (PCRRFLP) using BlnI (AvrII) enzyme. All isolate were inoculated on the medium, incubated at 37 \?C in ambient air for 24 to 96h. Examination was done in Fluorescent chamber with illumination at 365 nm.
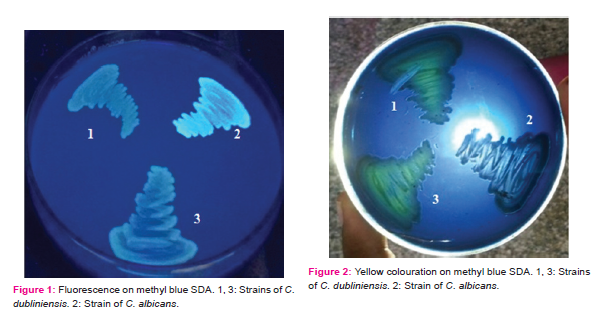
Result: On this medium, 156 C. albicans isolates showed fluorescence at 48h of incubation while none of the 14 C. dubliniensis isolates did so even on extending the incubation period. Also after 96h of incubation colonies of all 14 test strains and the two reference strains of C. dubliniensis showed yellow colour when viewed against light while others did not.
Conclusion: In conclusion, based the results of our study, methyl blue SDA test offers an additional simple means for identification of C. dubliniensis.
Keywords: Candida dubliniensis, Candida albicans, Methyl blue Sabouraud dextrose agar medium, Phenotypic identification, Fluorescence
DOI: 10.7324/IJCRR.2018.10510
Full Text:
INTRODUCTION
The augmentation of mycoses has been favored by the increased numbers of immunocompromised individuals and species previously not associated with human disease and novel species have been identified as potential pathogens. 1,2,3A clear paradigm of this phenomenon is Candida dubliniensis that was first identified as a new species by Sullivan et al. (1995) in Dublin, Ireland (and was subsequently named after its place of origin) while performing an epidemiological investigation of oral candidiasis in HIV-infected and AIDS patients in the early 1990s.4,5 The earliest known isolates of C. dubliniensis precede the AIDS pandemics with one isolate deposited in the Central Bureau voor Schimmel cultures in Holland as C. albicans in 19526 and another in the British National Collection for Pathogenic Fungi as C. stellatoidea in 1957.4
Although the first isolate of C. dubliniensis, had been recovered way back in 1950s, it was not until the late 1980s or early 1990s that the next isolates of C. dubliniensis were identified.5 This clearly highlights the fact that due to phenotypic similarity with C. albicans, C. dubliniensis is generally misidentified. Afterwards, C. dubliniensis isolates were identified in a wide range of clinical settings.7,8,9,10,11,12,13 C. dubliniensis is most frequently isolated from the oral cavity because of greater adherence to human buccal epithelial cells, mucin, and the oral bacterium Fusobacterium nucleatum. 14
Although primarily associated with recurrent episodes of oral candidiasis in AIDS and HIV-infected patients, C. dubliniensis has also been implicated in cases of superficial and disseminated candidiasis in patients without HIV infection.4 The incidence of this yeast species is increasing whereas its epidemiology still remains to be elucidated. To gain a more complete understanding of the precise epidemiological role played by C. dubliniensis in human disease, it is essential that rapid and reliable tests for its identification be available in routine clinical microbiology laboratory. However, the introduction of such tests has been complicated by the fact that C. dubliniensis shares many phenotypic characteristics with C. albicans.
A variety of methods have been developed for phenotypic discrimination of isolates of C. dubliniensis from C. albicans. Variable results have been reported by different authors for each phenotypic method and none has been found sufficient alone for differentiation of the two species.15,A potentially more stable identification would be one based on the analysis of genetic variability5 but the tests based on genotypic analysis are not readily applicable for the identification of this species in most average mycology laboratories. So an easy-to-perform phenotypic test, if reliable, would be a valuable tool for differentiation of Candida dubliniensis from Candida albicans. Therefore, this study was designed to evaluate methyl blue Sabouraud dextrose agar medium for differentiation of the two species.
MATERIALS
This study was conducted in the Mycology Division of Department of Microbiology of a tertiary care hospital in Kashmir, India. The study was approved by the Institute’s Ethics Committee.
Test strains
A total of 200 Candida spp. were tested in this study. These included 186 stock strains of C. albicans tentatively identified by phenotypic methods such as germ tube formation, colony colour on HiCrome Candida differential agar (HiMedia) and characteristic morphology on corn meal agar. These were isolated from cancer patients with oral candidiasis/colonization and held in stock collection of Mycology Laboratory, Department of Microbiology. Remaining 14 isolates were strains of C. dubliniensis which were kindly provided by Dr. Ziauddin Khan (Professor and Chairman Department of Microbiology, Kuwait University). Identification of all these strains was confirmed by Polymerase Chain Reaction-restriction fragment length polymorphism (PCR-RFLP) using BlnI (AvrII) enzyme which produced two strong bands of 200 bp and 340 bp in C. dubliniensis and only one band of 540 bp in C. albicans.8
Reference strains
C. albicans 90028 obtained from National Culture Collection of Pathogenic Fungi, Department of Medical Microbiology, PGIMER, Chandigarh and C. dubliniensis (type strain CD36) and C. dubliniensis (CBS 7987) which were kindly provided by Dr. Ziauddin Khan (Professor and Chairman Department of Microbiology, Kuwait University) were included in the study.
METHODS
Methyl blue SDA was prepared as per manufacturers guidelines. The media was surface inoculated by overnight growth of test as well as reference isolates. Each plate was inoculated with 6 isolates. Incubation was done at 37 °C in ambient air for 24 to 96h. Examination was done in Fluorescent chamber with illumination at 365 nm. 4
RESULTS
Out of 186 test strains of C. albicans 156 strains fluoresced on methyl blue SDA when exposure to long-wave UV light while 30 (16.13%) strains did not. All the test strains of C. dubliniensis failed to fluoresce under these conditions at 48h of incubation and even on extending the incubation period further (Figure 1). Also after 96h of incubation colonies of all the test strains of C. dubliniensis showed yellow color when viewed against light while none of the test strains of Candida albicans formed such colored colonies. (Figure 2) The reference strain of C. albicans fluoresced on methyl blue SDA while those of C. dubliniensis did not but they showed yellow color when viewed against light while.
DISCUSSION
The aniline dye and the cell wall specific polysaccharides of C. albicans react to produces the fluorescent metabolite. No other species of Candida, when grown on SDA with aniline blue medium produces fluorescence when exposed to long-wave ultraviolet light.16 This property of C. albicans has been exploited to differentiate it from C. dubliniensis.
In our study we found that fluorescence on methyl blue SDA was 83,87% accurate in identifying C. albicans and 100% in identifying C. dubliniensis. We also found that 16.1% of C. albicans failed to produce fluorescence. Our findings are in agreement with Kirkpatrick WR. et al. (1998) 17 Kantarc?oglu AS et al. (2002)15 and Akgu¨l O et al. (2009)18 who found that fluorescence was not visible in all C. albicans isolates. Sullivan et al. (1998) reported that fluorescence may not be reproducible in isolates subjected to storage and repeated subculture.5
We also found that after prolonged incubation of 96h yellow colouration was produced by the two reference strains of C. dubliniensis and not by any of the reference or test strain of C. albicans. This feature of C. dubliniensis which could be observed by examining the growth against light can also help in the differentiation of C. dubliniensis from C. albicans. To the best of our knowledge, production of yellow colour by C. dubliniensis on methyl blue SDA has not been reported previously. Combining the above two features, methyl blue SDA appears to be a medium well-suited for medical mycological use. It can serve as a primary isolation and differentiation medium for C. dubliniensis.
Conclusion
In conclusion, methyl blue SDA test offers an additional simple means for identification of C. dubliniensis. Our study was first to report production of yellow coloration by colonies C. dubliniensis on methyl blue SDA. This feature also differentiates C. dubliniensis from C. albicans with 100% accuracy. So this medium can be used as a screening medium for identification of C. dubliniensis. Further studies over a large number of isolates in multiple laboratories are suggested to evaluate the reproducibility of this simple method.
Acknowledgements
The authors thank Sher-i-Kashmir Institute of Medical Sciences, Soura, Srinagar for funding the project. Authors thank National Culture Collection of Pathogenic Fungi (NCCPF), Department of Medical Microbiology, PGIMER Chandigarh and Dr. Ziauddin Khan; Professor and Chairman Department of Microbiology, Kuwait University for providing control strains for the project. Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
COMPLIANCE WITH ETHICAL STANDARDS
Funding: This work was supported by research grant from our parent institute, Sher-i-Kashmir Institute of Medical Sciences, Soura, Srinagar
Conflict of Interest: The authors declare that they have no conflict of interest.
Informed consent: Informed consent was obtained from all individual participants included in the study.
References:
- Coleman DC, Rinaldi MG, Haynes KA, Rex JH, Summerbell RC, Anaissie E J et al. Importance of Candida species other than Candida albicans as
opportunistic pathogens. Med. Mycol 1998; 36:156–65.
- Pontón J, Rüchel R, Clemons KV, Coleman DC, Grillot R, Guarro J et al. 2000. Emerging pathogens. Med Mycol 2000; 38: 225–36.
- Sandven P. Epidemiology of candidemia. Rev Iberoam Micol 2000;17: 73–81.
- Gutiérrez J, Morales P, González MA and Quindós G. Candida dubliniensis, a new fungal pathogen. J Basic Microbiol 2002; 42: 207–27.
- Sullivan D and Coleman D. Candida dubliniensis: Characteristics and Identification. J Clin Microbiol 1998; 36: 329.
- Odds FC, Nuffel LV and Dams G. Prevalence of Candida dubliniensis isolates in a yeast stock collection. J Clin Microbiol 1998; 36: 2869–73.
- Coleman D, Sullivan D, Bennet GP, Moran G, Barry H and Shaley D. Candidiasis, the emergence of a novel species, Candida dubliniensis. AIDS 1997;11: 557–67.
- Ceballos A, Gaitán LA, Ruesga MT, Ceballos L and Quindós G. Prevalence of oral lesions by Candida spp., their varieties and serotypes in a population of patient with AIDS under a Highly active antiretroviral therapy. Rev Iberoam Micol 1998; 15: 141–45.
- Meiller TF, JabraRizk M A, Baqui AA, Kelley JI, Meeks V I, Merz WG and Falkler WA. Oral Candida dubliniensis as a clinically important species in HIV-seropositive patients in the United States. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1999; 88: 573–80.
- Meis JF, Rhunke M, De Pauw BE, Odds FC, Siegert W and Verweij PE. Candida dubliniensis candidemia in patients with chemotherapy induced neutropenia and bone marrow transplantation. Emerg Infect Dis 1999; 5: 150–3.
- Polacheck I, Strahilevitz J, Sullivan D, Donnelly S, Salkin IF and Coleman DC. Recovery of Candida dubliniensis from non human immunodeficiency virus infected patients in Israel. J Clin Microbiol 2000; 38: 170–74.
- Redding SW, Bailey CW, Lopez Ribot JL, Kirkpatrick WR, Fothergill AW, Rinaldi MG and Patterson TF. Candida dubliniensis in radiation-induced oropharyngeal candidiasis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod., 2001; 91: 659–62.
- Willis AM, Coulter WA, Sullivan DJ, Coleman DC, Hayes JR, Bell PM et al. Isolation of C. dubliniensis from insulin-using diabetes mellitus patients. J Oral Pathol Med 2000; 29: 86
- Jabrarizk MA, Ferreira SM, Sabet M, Falkler WA, Merz WG, Meiller TF. Recovery of Candida dubliniensis and other yeasts from Human Immunodeficiency Virus-associated periodontal lesions. J Clin Microbiol 2001; 39: 4520–22
- Kantarc?oglu AS and Yücel A. The presence of fluconazole-resistant Candida dubliniensis strains among Candida albicans isolates from immunocompromised or otherwise debilitated HIV-negative Turkish patients. Rev Iberoam Micol 2002; 19:44-8.
- Gold Schmidt M C, Fung DYC, Grant R et al. New aniline blue dye medium for rapid identification and isolation of Candida albicans. Journal of Clinical Microbiology 1991; 29( 6): 1095-99
- Kirkpatrick WR, Revankar SR, Mcatee RK, Lopez Ribot JL, Fothergill AW, Mccarthy DI. Detection of Candida dubliniensis in oropharyngeal samples from human immunodeficiency virus-infected patients in North America by primary CHROMagar candida screening and susceptibility testing of isolates J Clin Microbiol. 1998; 36: 3007–12
- Akgul O and Cerkcioglu N. Hypertonic Sabouraud Dextrose Agar as a substrate for differentiation of Candida dubliniensis. Mycopathologia 2009; 167:357-59.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License