IJCRR - 10(5), March, 2018
Pages: 30-35
Date of Publication: 15-Mar-2018
Print Article
Download XML Download PDF
Evaluation of Scientific and Ethical Status of Drug Promotional Literatures (DPLs) for Ethical Medicinal Drug Promotion
Author: Pratit P. Vyas, Amol L. Bhave
Category: Healthcare
Abstract:Background: The study was aimed to evaluate the scientific and ethical status of Drug Promotional Literatures (DPLs) using World Health Organization (WHO) guidelines for ethical medicinal drug promotion. This would in turn create awareness amongst healthcare providers about DPLs and thus would promote rational use of drugs.
Methods: This cross sectional observational study was carried out at Department of Pharmacology, Medical College Baroda. DPLs were collected and evaluated for scientific and ethical status as per guidelines by WHO for ethical medical drug promotion.
Results: Out of total 616 DPLs collected, 371 satisfied the inclusion criteria. Out of them, 180 DPLs [48.5%] were of single drug whereas, 191 were of fixed dose combinations (FDCs) [51.5%] of which only [31.9%] FDCs were approved as per drug controller general of India (DCGI). Drugs acting on endocrine system [19.67%] were the most commonly promoted drugs.11.05% of DPLs were having catchy terms. Graphical presentations were present in [19.40%] of DPLs of which bar diagram [47.22%] was maximum, followed by line diagram [19.44%], tables [16.67%]. We observed that [11.61%] of DPLs were having irrelevant pictures while rest [88.39%] were with relevant pictures. In DPLs with relevant pictures, maximum pictures provided were of package of dosage form [41.36%] followed by photo/picture of woman [26.06%].35.04% of DPLs turned out to be promoting polypharmacy.
Statistical representation of data in the form of p-value related to drug under promotion was mentioned in [3.77%] of DPLs.
Conclusions: We hereby concluded that drug manufacturers didn't follow the WHO guidelines for ethical medicinal drug promotion, thus failing to fulfil the rational promotion of drugs.
Keywords: WHO guidelines, Drug promotional literatures, Ethical medicinal drug promotion, Drug marketing
DOI: 10.7324/IJCRR.2018.1055
Full Text:
INTRODUCTION
According to World Health Organization (WHO), Medicinal Drug Promotion refers to “all informational and persuasive activities by manufacturers and distributors, the effect of which is to induce the prescription, supply, purchase, and/or use of medicinal drugs” (WHO 1988).[1]
For the purpose of drug promotion pharmaceutical companies usually use Direct-to-physician(DTP) technique as a major marketing strategy, the primary goal being to convince physicians to prescribe the manufacturer’s product through these advertisements.[2]Other than this, pharmaceutical companies also use visual aids, flip charts, leave-behinds , advertisements , audio-visuals and gifts as modes of drug promotion.[3][4]
In India, promotional activities standards are set by self-regulatory code of pharmaceutical marketing practices, January (2007) and governed by Organization of Pharmaceutical Producers of India (OPPI), and by National legislation.[5]However, it has been consistently shown by many studies that the information/data provided through drug advertisements is inconsistent with the code of ethics.[2][12-14]As it has been repeatedly shown by various studies that physician’s behaviour is influenced by pharmaceutical promotions[4],hence promotion in the form of financial or material benefits should not be offered to or sought by health care practitioner to influence them in prescribing drugs.[1]
Majority of the health professionals are dependent on commercial sources of drug information from medical representatives, drug advertisement brochures etc., and it has great impact on physician’s behaviour of drug prescribing pattern.[6]Furthermore, most of the of physicians were of the view that drug promotional activities has undoubtedly have an influence on their prescribing practices.[8][15]Even physicians who are using scientific literatures as commercial source of information to obtain knowledge could be influenced by the same without being aware of it.[16]
Often on many occasions, the materials provided through promotions are often misleading and confusing,[6][7] but on the contrary doctors get motivated by the intensive marketing made by pharmaceutical companies.
WHO has published criteria for ethical medicinal drug promotion in order to support and encourage the improvement in health care system through rational use of drugs.[1] These promotional activities highly influence the prescribing behaviour of health care providers. In this era of growing popularity of evidence based medicine, it becomes utmost essential for critical analysis of drug promotional literatures to promote rational drug prescribing. [8][9]
Hence this study is aimed for evaluating the scientific and ethical status of the drug promotional literatures using WHO criteria for ethical medicinal drug promotion in order to overcome their inaccuracies and inadequacies. Such studies would play a pivotal role in pursuing health care providers towards the ultimate goal of rational prescribing.
MATERIALS AND METHODS
STUDY DESIGN
Cross sectional observation study
DATA COLLECTION:
SAMPLE SIZE
INCLUSION CRITERIA
All DPLs were collected from following sectors:
- Pharmaceuticals firms
- Medical representative association
- Private practitioners
- Multispecialty hospitals.
EXCLUSION CRITERIA
- DPL for medicinal devices and equipments (insulin pump, blood glucometer, etc)
- DPL for Homeopathic and Ayurvedic medicines
- Drug list
- Drug monographs
- Doubling of DPLs
METHODS OF SAMPLE COLLECTION
Following telephonic contact and conversation, details of our study were explained and appointment timings were fixed for collection of DPLs. As per inclusion and exclusion criteria, DPLs were collected and sorted.
The same protocol was followed for all sectors during the period of data collection.
DATA ANALYSIS
Data analysis was done under following headings :
- Type of drugs:
- Total DPL collection,
- Classification of collected DPLs,
- Product type (single/FDC)
- Status of FDC as per DCGI.[18]
- Pharmacological groups and DPL collection through various clinical disciplines
- Catchy terms
- Graphical Presentations :
- Bar diagrams
- Line diagrams
- Tables
- Cost comparison
- Pseudographs, etc
- Pictorial contents :
- Statistical representation, data of Post marketing surveillance and Polypharmacy
Data were entered using Microsoft Office Excel 2007. Descriptive statistics were produced for each outcome.
RESULTS
- TYPE OF DRUGS:
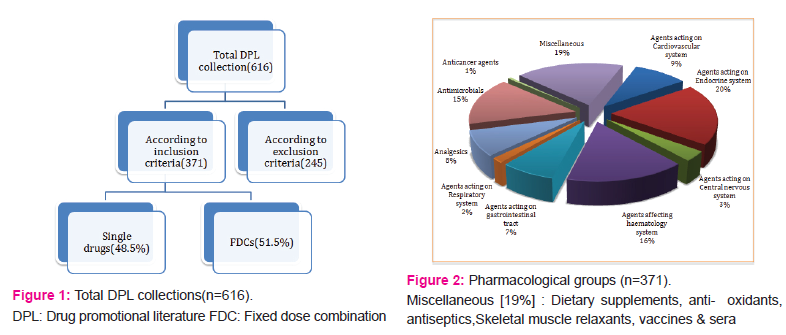
A total of 616 DPLs were collected, out of which 371 DPLs met the inclusion criteria and rest 245 DPLs were excluded. Out of total DPLs evaluated, 180 DPLs [48.5%] were of single drug whereas, 191 were of Fixed dose combinations [51.5%].In addition, we further evaluated the status of all 191 FDCs and found that out of total, only [61; 31.9%] FDCs were approved as per DCGI.[18][Figure no. 1]
- PHARMACOLOGICAL GROUPS and DPL COLLECTION THROUGH VARIOUS CLINICAL DISCIPLINES :
Drugs acting on endocrine system [73; 19.67%] were the most commonly promoted drugs; followed by miscellaneous agents [70; 18.88%], agents affecting haematology system [59; 15.9%], antimicrobials [57; 15.36%], etc. In miscellaneous group, the most commonly promoted were dietary supplements [27; 38.57%].[Figure no. 2]
Maximum DPLs collected belonged to obstetrics and gynaecology [142; 38.27%] discipline followed by medicine [64; 17.25%], surgery [57; 15.36%], skin and v.d.[46; 12.4%], etc.
- CATCHY TERM :
In our study we have observed that 41 [11.05%] DPLs having catchy terms.
- GRAPHICAL PRESENTATION :
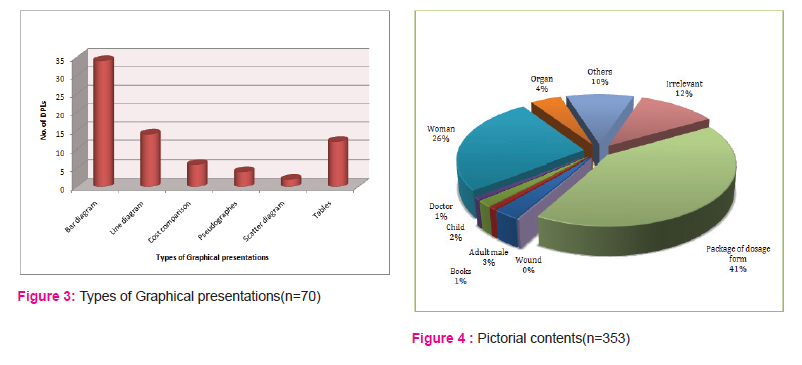
In our study some forms of graphical presentations [72; 19.40%] were present in DPLs. We observed that bar diagram [34; 47.22%] was the most commonly used graphical presentation, followed by line diagram[14; 19.44%], tables[12; 16.67%], cost comparison[6; 8.33%], etc.[Figure no. 3]
- PICTORIAL CONTENT :
Of the total DPLs [371], different pictorial contents were observed amongst 353 DPLs and were evaluated. 41 [11.61%] DPLs were having irrelevant pictures while the rest [312; 88.39%] had relevant ones. In DPLs with relevant pictures, maximum pictures provided were of package of dosage form [146; 41.36%] followed by photo/picture of woman [92; 26.06%], picture of human organ [16; 4.53%], picture of adult male [10; 2.83%], etc.[Figure no. 4]
- STATISTICAL REPRESENTATION :
We also evaluated that statistical representation of data in the form of p-value related to drug under promotion and found it to be mentioned in 14 [3.77%] DPLs.
- DATA OF POST MARKETING SURVEILLANCE:
1 DPL [0.27%] was found to provide data/information regarding post marketing surveillance
- POLYPHARMACY :
130 [35.04%] DPLs turned out to be promoting polypharmacy.
- MISCELLANEOUS :
Paper quality, print and colour were excellent in almost all DPLs. Considerable difference/dissimilarity was found in brand name and generic/INN with respect to font size and colour. We found in our study that font size of brand name was 2-4 times the font size of generic/INN which does not meet the standard guidelines.
DISCUSSIONS
Drug Promotional Literature (DPL) is readily available, easily accessible and important source of drug information. Direct-to-physician (DTP) marketing is one of the important facet of the promotion of pharmaceuticals.[4][8][17] Every year, new drugs enter Indian market of which majority are “me-too” products. Very few of them are genuine innovations and rest are with some altered formulations. As a result, more number of FDCs are added to more than 20,000 drug formulations present already in the market. [6]
Promotional activities were concentrated not much on innovative medicines’ exposure, but on publicizing fixed dose combinations not recommended by WHO. Drug manufactures spend more than $ 11 billion each year in drug promotion and marketing. Around $ 8000 to $ 13000 per year is spent on each healthcare professionals for drug promotional activities. [7] Pharmaceutical industries do not follow WHO guidelines while promoting their drug products, thus accelerating their commercial motive rather than ethical educational aspect. As a result, little therapeutic information is provided to help physicians to reach any rational decision about promoted drug.
Each DPL was analysed keeping in mind the objectives of the evidence-based medicine with the help of available evidences in the medical literature for its concurrence with WHO guidelines for ethical medicinal drug promotion.
A total of 616 DPLs were collected in our study, out of which 371 satisfied the inclusion criteria and rest 245 were excluded. Out of 245 excluded DPLs, most common were drug list (68.17%) followed by doubling of DPLs (15.51%), ayurvedic products (7.76%) , protein supplements(4.49%),etc. It was observed from studies viz. Mali et.al. and Khakhkhar et.al. that DPLs with Fixed dose combinations (FDCs) mounted to 41% and 49% respectively. Our study findings also fall in line with above studies with total number of DPLs having FDCs amounting to 51.5% and rest (48.5%) are having single drugs. Similar findings were also supported by another study by Jadav et.al. in which DPLs promoting FDCs were 46%. In addition to that, we further evaluated the status of FDCs and observed that of total, only 31.9% FDCs were approved as per DCGI. Hence the efforts of pharmaceutical corporate in providing only the rational elements seem to be questionable.
Some studies viz. Khakhkhar et.al. and Jadav et.al. respectively have revealed that anti-microbial agents (19% , 18%) followed by drugs acting on Cardiovascular system(19% , 16%) are the most commonly promoted drugs in DPLs. In another study by Mali et. al., it was observed that chemotherapeutic agents (26%) were the maximally promoted groups. In contrast to the above studies we observed that drugs acting on endocrine system (19.67%) were the most commonly promoted drugs in DPLs; followed by miscellaneous group (18.88%), agents acting on blood (15.9%), anti-microbial (15.36%), etc. Amongst miscellaneous group, most common promoted were dietary supplements (38.57%). In contrast to our findings, in another study carried out in Nepal by Kadiralam et.al. revealed dietary supplements to be only in 3% of DPLs.
Studies like Khakhkhar et.al. and Jadav et.al. have observed catchy terms to be there in 72% and 95% of DPLs respectively. In contrast to the above study findings; in our study we have found that DPLs with catchy terms were only 11.05%.
Our study observation revealed some of the drug promotional literatures using graphical presentations to depict some literary information. A total of 72 graphical presentations were evaluated in 19.4% of DPLs. Similar findings have been also highlighted in other studies viz. Mali et.al. and Jadav et.al. in which graphical presentations were given in 16% and 12% of DPLs respectively.
Unlike some other studies[2][11] in which pseudographs (29%) and columns were being maximum respectively, our study observations state, bar diagrams (47.22%) to be most commonly used graphical presentation followed by line diagram (19.44%), tables(16.67%) , cost comparison(8.33%),etc, whereas pseudographs falling almost towards the end.
As a part of persuasive communication, these promotional brochures were made striking using various types of pictures and devoting majority of the literature area to non-specific and less accurate contents which represent the tendency of pharmaceutical companies of wasting money in printing eye catching glossy paper promotional literature deprived of important therapeutic information. Unlike other studies viz. Mali et al and Khakhkhar et. al., which showed the presence of irrelevant pictures in 90% and 69% of DPLs respectively, our study found that only 11.61% of DPLs were having irrelevant pictures. DPLs with relevant pictures (88.39%) observed package of dosage form (41.36%) to be maximum followed by photo portrait woman (26.06%), picture of human organ (4.53%), picture of adult male (2.83%), etc. Having maximum photo images of women indicates the urge to create attraction for particular product.
We also evaluated that statistical representation of data in the form of p-value related to drug under promotion and found it to be mentioned in only 3.77% of DPLs. Our findings are supported by another study of Khakhkhar et.al. showing only 7% of literature having data related to statistical representation. Data of post marketing surveillance were only given in 1 DPL. These important aspects related to long term safety of drugs were also grossly neglected. We have observed in our study that 35.04% DPLs were promoting polypharmacy. Of the various references we compared, none of them were having data regarding post marketing surveillance and polypharmacy.
Paper quality, print and colour were excellent in almost all DPLs. Considerable difference/dissimilarity was found in brand name and generic/INN with respect to font size and colour. We found in our study that font size of brand name was 2-4 times the font size of generic/INN which does not stand ideal.
Printed promotional material is an important source of information. Most health professionals are dependent on commercial sources of drug information from medical representatives, drug advertisement brochures etc., and it has great impact on prescribing behaviour [6].
In India, there are regional Ethics Committees for complaints against unethical drug promotion advertisements. Drug controller authority takes necessary legal steps in response to such complaints to against drug manufacturers and distributors[6]. Forwarding more complaints about irrational promotion to regulatory authority by cautious doctors might lead pharmaceutical industry to incline toward self-regulation. Government regulatory bodies must play a proactive role where code of ethics is failing. Wherever the hospitals are attached to the academic institutes, prior scrutiny of the promotional material for authenticity of the content could be done by respective department of pharmacology.
The strength of our study stands in the way of having critically evaluated drug promotional literatures which are the major source of establishing interaction between clinicians andmanufacturers which in turn promote rational usage of various agents in order to better the health status of community.
We do accept and feel that in this vast field of drug promotion strategies, we have evaluated only one type of promotional activity, i.e. Drug Promotional Literatures (DPLs). Hence the need always stands to assess other forms of drug promotional activities also i.e. visual aids, leave behinds, leaflets and audio visuals.
CONCLUSIONS:
We hereby concluded that drug manufacturers didn’t follow the WHO guidelines for ethical medicinal drug promotion, thus failing to fulfil the rational promotion of drugs. On the basis of the observations of our study, we have observed that many of the literatures were having non-scientific and unethical information based on which it is suggested that physicians need to be aware of the flaws in promotional literatures before accepting it as valid source of drug information.
ACKNOWLEDGEMENT
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
DECLARATIONS
Source of Funding: No
Conflict of interest: No
Ethical approval: Approval taken from Institutional Ethics Committee for Human Research (IECHR) Medical College and SSG Hospital, Baroda.
EC Reg No: ECR/85/Inst/GJ/2013
References:
- Ethical criteria for medicinal drug promotion. World Health Organization [Online]. 1988 May 13; Available from:
http://www.who.int/medicinedocs/collect/edmweb/pdf/whozip08e/ whozip08e.pdf.
- Mali SN, Dudhgaonkar S, Bachewar NP. Evaluation of rationality of promotional drug literature using World Health Organization guidelines. Indian J Pharmacol 2010;42:267-72.
- Cooper RJ, Schriger DL. The availability of references and the sponsorship of original research cited in pharmaceutical advertisements. CMAJ 2005;172:487-91.
- Cardarelli R, Licciardone JC, Taylor LG. A cross-sectional evidence-based review of pharmaceutical promotional marketing brochures and their underling studies: Is what they tell us important and true? BMC Fam Pract 2006;7:13.
- OPPI code of pharmaceutical marketing practices. Available:http://www.indiaoppi.com/OPPI%20Code%20of%20 Marketing%202007.pdf [Online]. 2007 Jan.
- Gopalakrishnan S, Murali R. India: Campaign to tackle unethical promotion. World Health Organization. Essential drugs monitor [Online] 2002. p. 22. Available from: http://www.apps.who.int/medicinedocs/pdf/s4937e/s4937e.pdf
- Rohra DK, Gilani AH, Memon IK, Perven G, Khan MT, Zafar H, et al.Critical evaluation of claims made by pharmaceutical companies in drug promotional material in Pakistan. J Pharm PharmSci 2006;9:50-9.
- Villanueva P, Peiro S, Librero J, Pereiro I. Accuracy of pharmaceutical advertisements in medical journals. Lancet 2003;361:27-32.
- Lexchin J. Enforcement of codes governing pharmaceutical promotion: What happens when companies breach advertising guidelines? CMAJ 1997;156:351-6
- Khakhkhar T, Mehta M, Shah R, Sharma D. Evaluation of drug promotional literatures using WHO guidelines. J Pharm Negative Results 2013;4:33-8.
- Jadav SS, Dumatar CB, Dikshit RK., Running title: Drug promotional literatures (DPLs) evaluation as per World Health Organization (WHO) criteria. J App Pharm Sci, 2014; 4 (06):084-088.
- Stryer D, Bero LA. Characteristics of materials distributed by drug companies. An evaluation of appropriateness. J Gen Intern Med. 1996;11:575–83.
- Smart S, Williams C. Evidence based advertising. Half of drug advertisements in BMJ over six months cited no supporting evidence? BMJ. 1997;315:1622–3.
- Mindell J, Kemp T. Evidence based advertising. Only two fifth of advertisements cited published, peer reviewed references? BMJ. 1997;315:1622.
- Drug Promotion what we know, what we yet to learn. Geneva: World Health Organization and Health Action International; 2005.
- Gitanjali B, Shashindran CH, Tripathi KD, Sethuraman KR. Are drug advertisements in Indian edition of BMJ unethical? BMJ [serial online] 1997.
- Medhi B, Prakash A. 2010. Ideal characteristics of promotional literature. In: Medhi B and Prakash A, ed. Practical Manual of Experimental and Clinical Pharmacology. 1st edition. India: JBPMP 342-45.
- FIXED DOSE COMBINATIONS APPROVED BY DCG (I) SINCE 1961 TILL NOVEMBER, 2014. Available from:http://cdsco.nic.in/writereaddata/Aprroved%20FDC%20list%20till%20november%202014.pdf
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License