IJCRR - 10(2), January, 2018
Pages: 06-15
Date of Publication: 19-Jan-2018
Print Article
Download XML Download PDF
Formulation of Cost Effective Alternative Bacterial Culture Media Using Fruit and Vegetables Waste
Author: Pratibha Jadhav, Mrunalini Sonne, Arati Kadam, Suraj Patil, Kirti Dahigaonkar, Jaspal Kaur Oberoi
Category: General Sciences
Abstract:Vegetable stalks and fruit peels are generally used for composting or merely disposed off as waste. Considering their nutritional values, these kitchen wastes can be utilized for production of alternative cultivation media. Cost of conventional culture media is very high. Use of these alternatives would reduce the cost drastically. The present study is aimed at replacing the nutrient source by various locally available cheap materials, such as vegetables and fruits waste, that contains considerable amount of protein and starch. These raw materials are cheap and easily available in local shops and vegetable markets and also as kitchen
wastes. They were selected as a natural nutrient source to prepare the alternative culture media. Growth of bacteria and pigment production efficiency was analyzed using nine formulations. The results showed that drumstick formulations having seed and peel extract, formulations B and D supported growth of E.coli, Serratia sp., Pseudomonas sp.
Keywords: Alternative media, Cost effective media, EDS analysis
DOI: 10.7324/IJCRR.2018.1022
Full Text:
INTRODUCTION
Microbiological studies depend on the ability to cultivate and maintain microorganisms under laboratory conditions by providing suitable culture media that offers favorable conditions. A nutrient media prepared for the growth of microorganisms in a laboratory is called culture media. Microorganisms can obtain energy directly from sunlight while carbon can be made available in organic forms such as carbohydrates or inorganic forms such as carbon dioxide and water. Nutrient agar medium is commonly used as general purpose medium for the cultivation of broad range of bacteria. It is a basic medium composed of peptic digest of animal tissue, beef extract and yeast extract, sodium chloride and agar. Commercially available media such Nutrient Agar, Cetrimide Agar, MacConkey Agar are used for the growth of microorganisms but these are very expensive.
In today’s world waste disposal is also a major problem. So lot many researches are carried so as to use domestic waste for production of cheap media. Higher cost of cultivation media is a matter of concern[2]. Therefore, various alternative media are formulated and alternatives for agar are tested, so as to reduce the cost involved.
Agar is a solidifying agent and very few studies have concentrated on replacing agar for solidification. By comparing other studies carried out in this area by a number of researchers, now it would be possible to use a number of sources as alternative culture media[1]. Even then, increasing cost of culture media has necessitated continuous search for more readily available culture media at affordable price.
Different media for the growth and isolation of organisms have been reported from different substrates[6]. Some vegetables and fruits have been used to cultivate both fungi and bacteria, such as Gooseberry[36],Carrot, Tomato, Cabbage, Pumpkin etc [8]. with easily available low cost material as substitutes for Nutrient Agar. Some others have used cow pea, green gram and black gram as starch and protein substitutes to reduce the cost of microbial media[40].
Microorganisms are omnipresent and very diverse. Preparation of suitable culture media is one of the necessary to study them. Different microorganisms grow in different environments and have variety of growth requirements; like nutrients, pH, osmotic conditions and temperature[7]. The current limitations of cultivation of microbes in lab need to be addressed by formulation of newer media.
Microbial culture media can be of different types, depending on the nutritional growth requirements of the microorganisms. Microorganisms require various macro elements namely C, H, O, N, S, P, K, Ca, Mg and Fe. For Carbohydrates, Lipids, Proteins and Nucleic acids synthesis C, H, O,N S, P are used, while K, Ca, Mg, Fe exist in the cell as cations, playing a variety of roles. In addition to macro elements, all microorganisms require several microelements like Mn, Zn, Co, Mo, Ni and Cu. These are generally part of enzymes and cofactors. Microorganisms also require organic compounds as growth factors.
MATERIAL AND METHODS
Collection of samples
Vegetables and fruits like Drum stick (seeds and peels), Orange peel, Potato peel, Cauliflower stalk and Fenugreek stem were collected from local vegetable market. The collected samples were transported to the laboratory and processed immediately.
Treatment of samples
Peels, stalks and seeds were sun dried for 2-3 days. Dried material was grinded to powder using electronic blender. The powdered samples were kept in air tight containers until its use.
Test organisms used
E.coli, Serratia sp., Pseudomonas sp.
Formulation of Media
The dry powder was kept in warm water for 2-3 hours. Then filtered with the help of filter paper and filtrate was used to nine different solid formulated media. Then agar, which is solidifying agent was added in 100 ml distilled water[9].
In all experiments pH of the media was adjusted to 6.5 - 7.0. The dissolved media was sterilized in autoclave (Dixons, ST19T) at 121°C for 20 minutes under 15 psi pressure and were poured into sterile Petri dishes separately.
Preparation of fresh culture
Bacterial cultures used for analysis were E.coli, Pseudomonas sp., Serratia sp.. Then these bacteria were streaked on the freshly prepared NA medium from the original stock culture. The cultures were allowed to incubate at 37°C for 24 hours.
Microbial inoculation into alternative media
The young cultures of test bacteria such as E.coli, Pseudomonas sp., Serratia sp.. were inoculated in triplicates in each alternative culture medium. Then all the plates were incubated at 37°C for 48 hours. After the incubation all the plates were observed for bacterial growth and pigmentation.
Analysis of bacterial growth in formulated media
Bacterial growth was checked at 37°C for total duration of 24 hrs. Time interval selected for growth curve was one hour. Fresh culture of test organisms was inoculated in Nutrient broth and formulated medium. Growth was measured at 600nm.
Estimation of proteins and carbohydrates
Protein was estimated by Folin Lowry’s method while carbohydrates content was analyzed by DNSA method.
Chemical analysis of dehydrated powder
The chemical composition, macronutrient content of the tested samples was determined by EDS (Energy Dispersive Spectroscopy) analysis.
RESULTS
Alternative media supported the growth of test organisms like E.coli, Pseudomonas sp., Serratia sp. There was no significant variation in the colony morphology. Pigmentation was slightly affected in some cases.
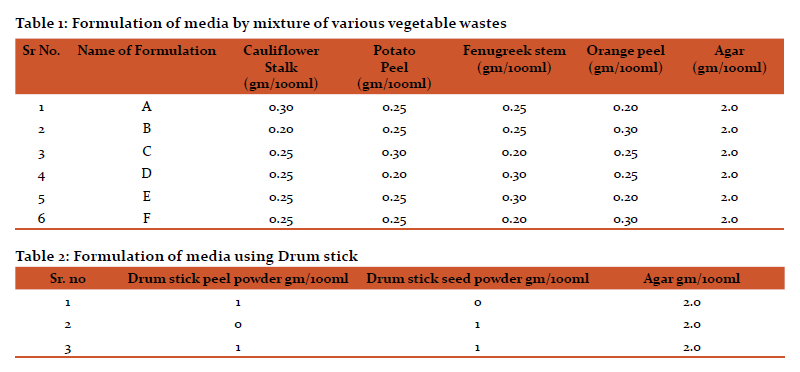
Growth of Serratia sp. on different media
Serratia sp. grown on control (NA) showed pigmentation. Serratia sp. grown on formulated media showed no significant variation in colony morphology. Serratia sp. showed maximum growth and pigmentation on DP, DS, B, D formulations. Pigmentation was slightly affected in A, E, F formulations.
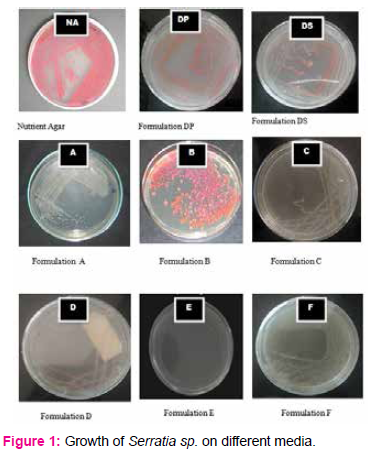
Growth of Pseudomonas sp. on different media
Pseudomonas sp. grown on control (NA) showed pigmentation. When Pseudomonas sp. was grown on formulated media there was no significant variation in colony morphology but showed no pigmentation on DP, DS, A, B, C, D, E, F formulations.


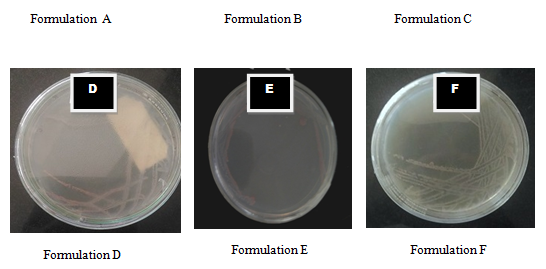
Growth of E.coli sp. on different media
E.coli grown on control (NA) showed growth. When E.coli was grown on all formulated media there was no significant variation in colony morphology.
Fig 3 Growth of E.coli on different media:
Microbial growth curve:
The microbial growth was tested and measured at different incubation intervals. Growth was measured at 600 nm on spectrophotometer.
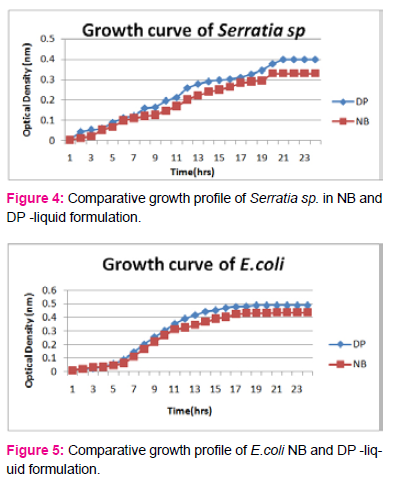
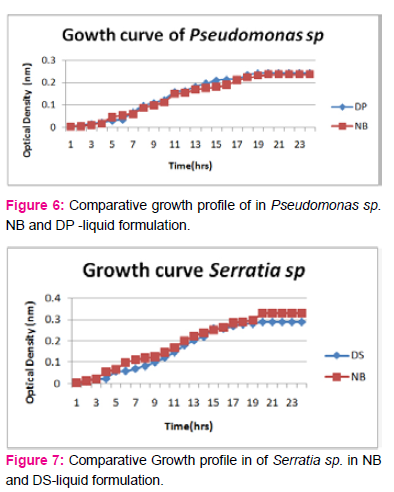
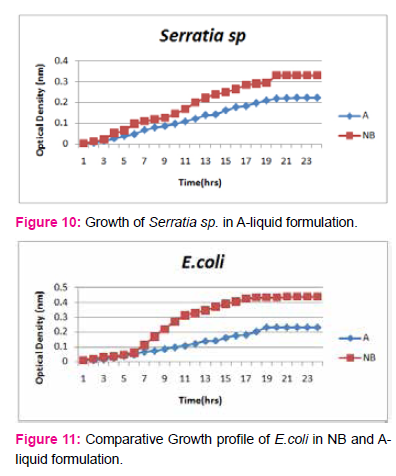
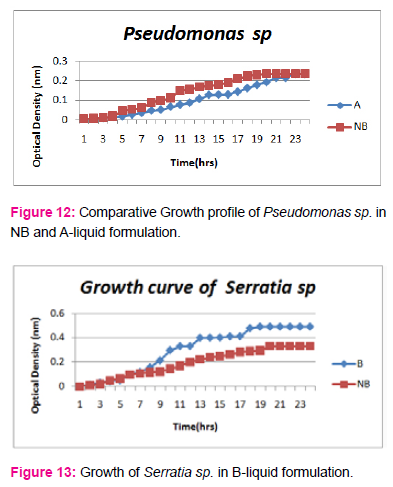
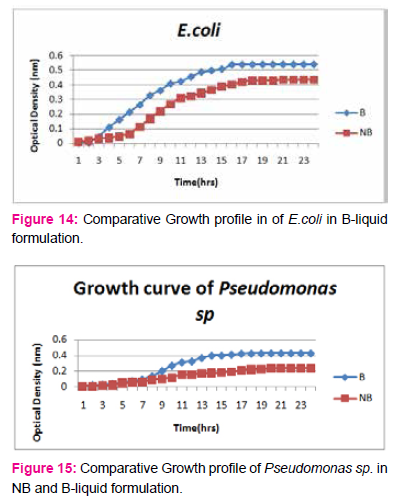
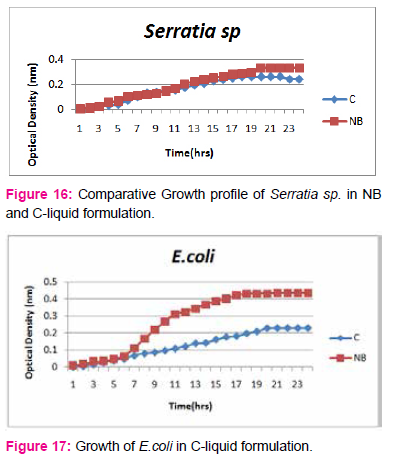
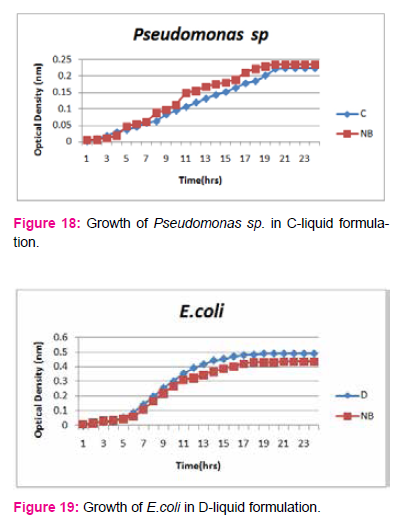
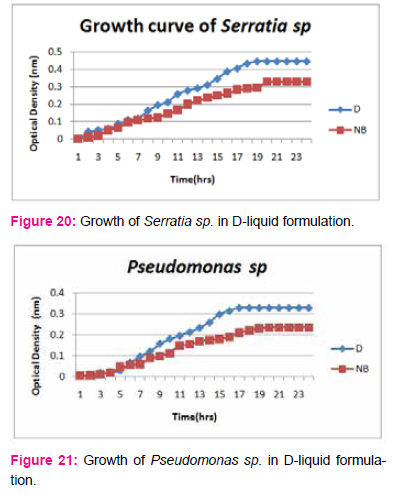
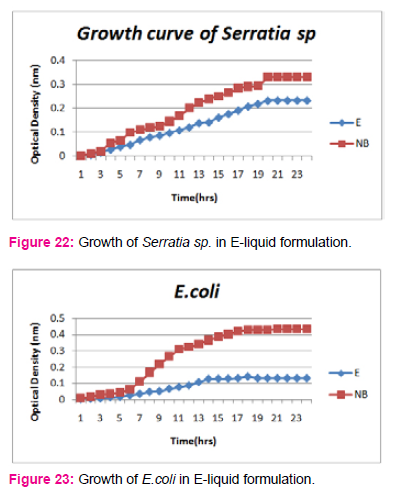
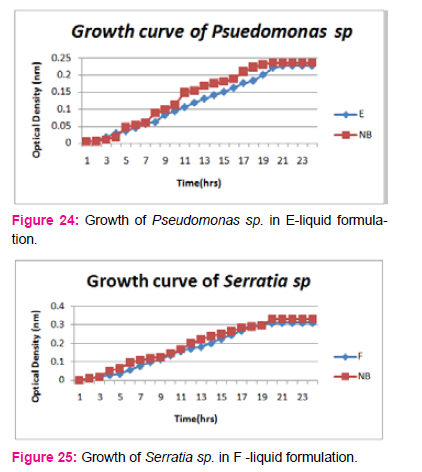
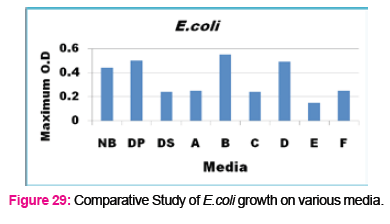
When growth of Serratia sp. in NA was compared with DP,DS,A,B,C,D,E,F, it was found that maximum growth was found in DP,DS,B and D and less growth was found in A,C,E,F,DS. Percent increase in growth in comparison to NA was DP-25%, B-42%, D-33%.
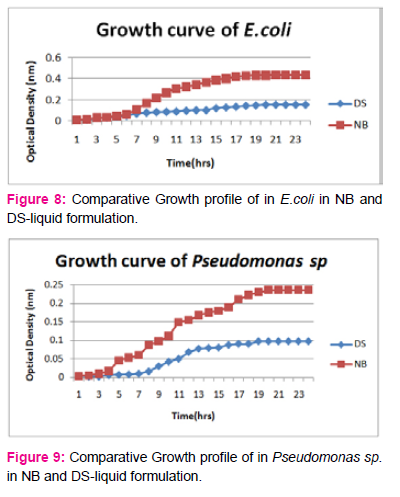
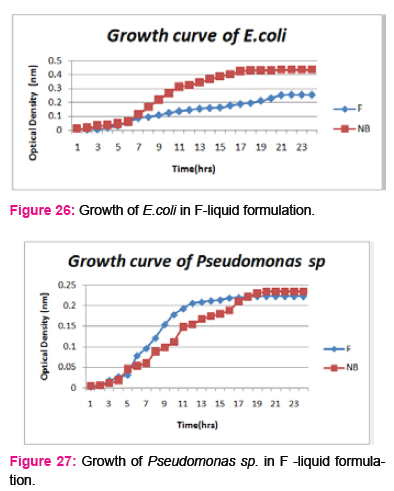
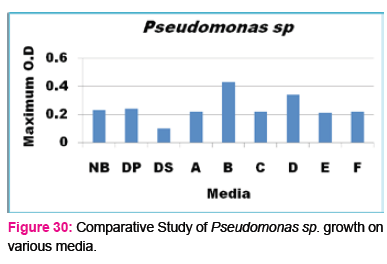
When growth of E.coli sp.in NA was compared with DP,DS,A,B,C,D,E,F, it was found That max growth was found in DP, DS, B and D and less growth was found in A,C,E,F,DS.
Percent increase in growth in comparison to NA was DP-16%, B- 20.75%, D-14%.
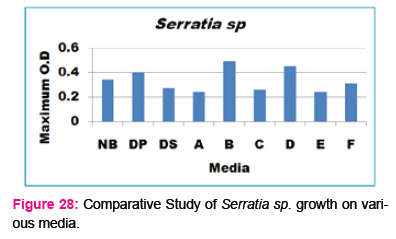
When growth of Pseudomonas sp. in NA was compared with DP,DS,A,B,C,D,E,F, it was found that maximum growth was found in DP, DS, B, and D and less growth was found in A,C,E,F,DS. Percent increase in growth in comparison to NA was DP-8 %, B-47.61%, D-33%
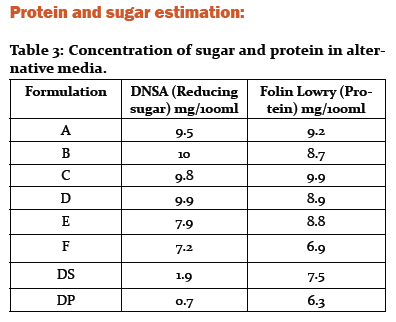
Protein and Sugar concentration was estimated by standard method. B media contains 10 mg/100 ml of Sugar and 8.7 mg/100 ml of protein. D media contains was 9.9 mg/100ml of Sugar and 8.9 mg/100 ml of protein. DP media it was 0.7 mg/100ml of Sugar and 6.3 mg/100 ml of protein. Thus, being rich in sugar and proteins, the media is able to support growth of microorganisms.
Chemical analysis of the dehydrated powder:
To determine chemical composition and nutritional content of formulated media EDS (Electronic Dispersive Spectroscopy) analysis was done.
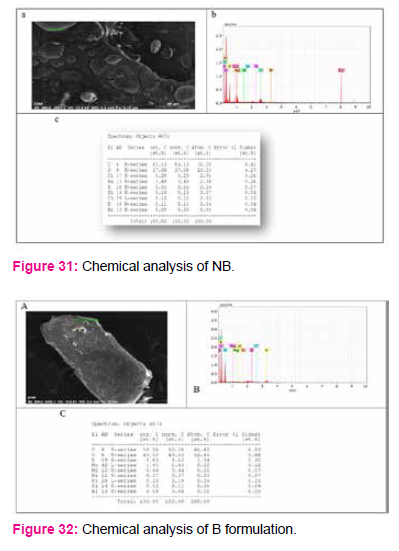
In B formulation Magnesium (Mg), Molybdenun(Mo), Nikel(Ni) was found which was absent in control NB. But Cu (copper), Chloride(Cl), Sulfur(S) are present in control NB and absent in B formulation media.
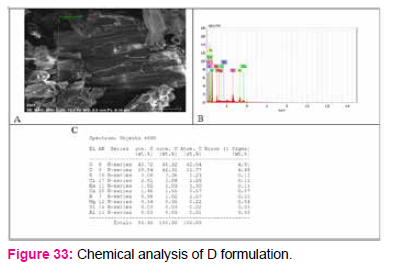
In D formulation Magnesium (Mg), Calcium (Ca), N (Nitrogen), was found which was absent in control NB. Cu (copper), Sulfur (S) was present in control NB but absent in D formulation.
In DP formulation Magnesium (Mg), Phosphorus (Ca), N (Nitrogen), was found which was absent in control NB. Cu (copper), Aluminium (Al) was present in control NB but absent in formulated DP media.
Comparative cost study of media:
The cost of Alternative media is drastically less than the commercial liquid media. The commercial liquid media containing agar for solidification is also very cost effective as compared to commercial solid media

The cost of alternative media is drastically less than commercial liquid media. The B, D, and DP media containing agar for solidification is also very cost effective as compared to commercial solid media. The rise in cost of B, D and DP agar media is due to incorporation of agar which adds on to the cost Alternative cheap source of solidifying agent needs to be found so that this cost be brought down.
DISCUSSION
Cultivation of microorganisms in laboratory requires nutrient environment which serves as source of nutrients for growth. Our investigation was aimed at replacing nutrient media by alternatives such as vegetable and fruit waste. In reported studies; carrot, tomato, cabbage, pumpkin waste was used in formulation of media[8] but in our research we introduced cauliflower, drum stick, orange peel, potato peel and fenugreek stem for formulation of media.
CONCLUSION
The formulated medias A to F supported growth of bacteria such as E.coli, Pseudomonas sp., Serratia sp. In preliminary study it was found that E. coli grew well in B formulation in 24 hours. Pseudomonas sp. grew best in B formulation in 24 hours while pigment production was observed after 48 hours. Serratia grew best in all formulations and pigment production was observed in 24 hours at room temperature. EDS analysis was done to find the elements present in NA, DP, B and D formulation that supported growth of E.coli, Serratia sp. and Pseudomonas sp.
After comparing the growth of organisms in commercial and alternative media, the growth of Serratia sp on alternative media shows 25%, 42%, 33% rise in DP, B and D media respectively. E.coli shows 16%, 21%, 14% rise in growth on DP, B and D media respectively. Pseudomonas sp. shows 8%, 48%, 33% rise in growth on DP, B and D respectively, which is higher than that of growth on commercial media. Alternative media could be used as cheap media for routine experiment in laboratory. On comparison with Nutrient Broth, our formulated media DP, D and B gave better results. Formulated media was found to be highly cost effectively.
References:
- A. Ravathie, P. Sevvel, R. Nirmala, N. Kularajany (2012). Journal of natural product and plant resources, 2; 697-700.
- Adesemoye AO and Adedire CO, Use of Cereals as basal medium for the formulation of alternative culture media for fungi, World J Microbiol and Biotech, 2005; 21: 329-336.
- Adoki A., (2008). Factors affecting yeast growth and protein yield production from orange, Platinum and banana waste processing residues using Candida sp. African. J. Biotechnology 7(3), 290-295.
- Anupama and Ravindra P. Value added Food: single cell protein (2000). Biotechnology advances, 18, 459- 479.
- Asad M. J., Asghan M., Yaqub M. and Shahzad K (2000) Production of single cell protein,. (2000). Delignified Corn Cob By Arachniotus species, Pak. J. Of Agric. Sci., 2000. 37, 3-4.
- Basu S, Bose C, Ojha N, et al. Evolution of bacterial and fungal growth media. Bioinformation. 2015;11(4):182-184. doi:10.6026/97320630011182
- Bhattacharya S, Vijayalakshmi N, Parija S C. Uncultivable bacteria : Implications and recent trends towards identification. Indian J Med Microbiol 2002;20:174-7
- Deivanayaki M and Iruthayaraj AP, Alternative vegetable nutrient source for microbial growth, Inter J Biosci, 2012; 2: 47-51.
- Famurewa and O.M. David, 2008. Formulation and Evaluation of Dehydrated Microbiological Media from Avocado Pear (Peasea americana Cmill). Research Journal of Microbiology, 3: 326-330.)
- Jamel P., Alam M. Z. and Umi N.,( 2008) Media optimization for bio proteins production from cheaper carbon source,.. J. of Engi. Sci. and Techno., 3(2) 124-130.
- Janssen PH, Yates PS, Grinton BE, Taylor PM, SaitM (2002) Improved culturability of soil bacteria and isolation in pure culture of novel members of the divisions Acidobacteria,Actinobacteria, Proteobacteria, and Verrucomicrobia.. Appl Environ Microbiol.;68:2391–6.
- Kasanadze AK (2000) Replacement of Agar by cassava flour in microbial media. BSc Dissertation University of Malawi, Bunda College of Agriculture, Lilongwe.
- Konstantinidis KT, Tiedje JM. (2005) Genomic insights that advance the species definition for prokaryotes..ProcNatlAcadSci USA;102(2567–2572):99.
- Kuria P, Demo P, Nyende AB, Kahangi EM. (2008) Cassava starch as an Alternative gelling agent for the in vitro micro propagation of potato (Solanum tuberosum L.).. African Journal of Biotechnology, .7: 301–307.
- Leach HW, McCowan LD, Schoch TJ. (1959) Swelling and solubility patterns of various starches..Cereal Chemistry. 36: 534-544.
- Lucyszyn, N., M.Quoirin, M. M. Homma and M. R. Sierakowski. (2008) Agar/galactomannan gels applied to shoot regeneration from tobacco leaves.. Biol. Plant. 51(1):173-176.
- Maliro MFA, Lameck G. (2004) Potential of cassava flour as a gelling agent in media for plant tissue cultures. African Journal of Biotechnology ,. 3: 244-274.
- Mbanaso ENA. (2008) Effect of multiple subcultures on Musa shoots derived from cassava starch-gelled multiplication medium during micropropagation. African Journal of Biotechnology Vol 7, No 24 (2008)
- Mohamed, M. A. H., A. A. Alsadon and M. S. Al Mohaidib .( 2009) Corn and potato starch as an agar alternative for Solanumtuberosum micro propagation. .Afr. J. Biotechnol. 9(1): 012- 016.
- Murashige, T. and F.Skooga.( 1994) A revised medium for rapid growth and bio assays with the tobacco tissue cultures.. Physiol. Plant. 15: 473-497.
- N. Ravimannan, R. Arulanantham, S. Pathmanathan and K. Niranjan. (2014) Annals of Biological Research,. 5(1), 36-39.
- Naik, P. S. and D. Sarker. ( 2001) Sago An Alternative Gelling Agent For Potato In Vitro Culture..Biol.Plant. 44(2): 293.
- Najafpur, Ghasem D. (2007) Single Cell Protein. Biotechnology advances. Biochemical Engineering and Biotechnology Advances, .332-347.
- Nambiar VS, Bhdalkar k, Daxini. (2003) Drumstick Leaves As Source Of Vitamin A In ICDS-SFP.. Indian Journal of Pediatrics.70: 383-387.
- Nambiar VS, Mehta R, Daniel M. (2005) Polyphenol Content of Three Indian Green Leafy Vegetables.2005. Journal of Food science and Technology.,42: 312-315.
- Nambiar VS, Seshadri S. (1998.) Beta Carotene Cotent Of Green Leafy Vegetables Of Western India By HPLC. Journal of Food Science and Technology. 35: 365-367.
- Nichols D. (2007) Cultivation gives context to the microbial ecologist..FEMS Microbiol E.coli.; 60:351–7.
- Ozel, C. A., K. M. Khawar and O. Arslan (2008).Comparison Of The Gelling Of Isubgol, Agar And Gelrite On In Vitro Shoot Regeneration And Rooting Of Variety Samsun Of Tobacco (Nicotiana tabacuml.).. Scient.Horticul.2008. 117(2): 174-181.
- Pandhre GR, Satwase AN, Hashmi SI. (2011) Effect Of Different Pretreatments And Drying Methods On Color Characteristics Fenugreek Leaves., Int J Cur Sci Res 1: 115-119.
- Pham VHT, Kim J. Cultivation Of Unculturable Soil Bacteria.(2012). Trends Biotechnology . 30:475–84.
- Porndarun, H. Vichai. and C.Walairut. (2010) Department of product development, Faculty of agro industry , Kasetsart University, Bangkok 10900,Thailand,. 100-106.
- Priadi, D., H. Fitriani and E. Sudarmonowati. (2008) The growth of cassava (Manihotesculenta Crantz)on various alternative gelling agents. Biodiversitas 9(1): 9-12.
- Prokash, S., M. I. Hoque and T. Brinks.( 2000) Culture media And Containers,. Biotechnology And Eco Development Research Foundation, Bangalore, India..
- S. Tharmila, E. C. Jayaseelan and A. C. Thavaranjith (2011) Archives of applied science Research,.3; 389-393.
- S. Tharmila, E. C.Jeyaseelan and A. C. Thavaranjit (2011) Archives of Applied Science Research. Vol.3 (3):389-393.
- Sathiya Vimal S, Vasantha Raj. S , Senthil kumar. R. P , and Jagannathan. (2013) Natural Sources of Gooseberry Component used for Microbial Culture Medium Journal of Applied Pharmaceutical Science Vol. 3 (11), 040-044.
- Vartoukian SR, Palmer RM, Wade WG. (2010) Strategies for culture of ‘unculturable’ bacteria. FEMS Microbiol Lett. 309:1–7.
- Zapata, A. Cost Reduction In Tissue Culture Of Banana.(2001). (Special Leaflet), International Atom Energy Agency Labs., Agric. And Biotech. Lab., Vienna, Austria.
- Zhe, W. R., C. Debasis, E. J. Hahn and K. Y. Paek. (2005) Growth Of Doritaenopsis In Peat substituted Growing Medium. J. Korean Soc. Hort. Sci.. 46(1): 76-81.
- Wasas AD, Huebner RE, Klugman KP. Use of Dorset Egg Medium for Maintenance and Transport of Neisseria meningitidis and Haemophilus influenzae Type b. Journal of Clinical Microbiology. 1999;37(6):2045-2046.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License