IJCRR - 9(17), September, 2017
Pages: 37-40
Date of Publication: 12-Sep-2017
Print Article
Download XML Download PDF
A Review on Lorcaserin - A Selective 5-HT Serotonin Receptor Agonist in Obesity Management
Author: Venkatesh P., Venkhat Balaji B.R., Venu Gopal K., Vinodhini S., Vinodhini T., Vinodhini C., Chitra K.
Category: Healthcare
Abstract:Lorcaserin is a novel anti-obesity agent. The study retrospect's the pharmacokinetic effects, mode of action, adverse drug reactions and various uses of Lorcaserin. Lorcaserin is pro-opiomelanocortin neurons stimulator present in the nucleus of hypothalamus resulting in a peak melacortin-4 receptor activity, which results in satiety and decreased food intake. Though some side effects were reported, the potential benefits of Lorcaserin outweigh the risks. Serum drug monitoring is not required. Lorcaserin is a 5HT2C receptor agonist whose property may also be studied to treat anxiety, Alzheimer's disease, depression and parkinsonism. Literature review was conducted to identify relevant studies. The study reviewed the pharmacokinetics, pharmacodynamics and clinical trials proposed so far on lorcaserin.
Keywords: Lorcaserin, 5-HT agonist, Anti-obesity
DOI: 10.7324/IJCRR.2017.9176
Full Text:
Introduction
Physical interventions such as exercise, diet and surgery, behavioural therapies, and pharmacological treatments are the approaches taken for the management of weight reduction in obese individuals. This may be done alone or in combination for greater efficiency.
Administration of anti-obesity drugs may lead to a reduction in the absorption of nutrients and appetite. It may also results in an increased satiety and energy expenditure. The better results were achieved with the pharmacotherapy for weight loss of about 2 to 7.9kg when compared to that treated with placebo.
5-HT is a monoamine neurotransmitter is mostly seen in the central serotogenic neurons and enterochromaffin cells with a broad spectrum of behavioural and physiological function. Hence, 5-HT receptor is considered as an anti-obesity drug target.
There is a wide range of 5-HT2c receptor modulating drugs having the ability to deal with a variety of conditions by changing the central serotogenic function. Such conditions are addiction, depression, anxiety, Alzheimer’s disease, parkinson’s disease and obesity1.
For the treatment of obesity only limited numbers of drugs are in use. In 1999, Orlistathas got approval by the Food and Drug Administration (FDA). Later in June 2012, a new drug Lorcaserin was approved and promoted for prescription by FDA. But In October 2012, it was rejected initially due to some cancer signal detection in animal studies. Finally, from further research such as BLOOM and BLOSSOM the drug was approved in the same year2.
Lorcaserinis chemically [1R]-8-chloro-2,3,4,5-tetrahydro-1-methyl-1H-3-benzapine and acts as a selective 5-hydroxy tryptamine (5-HT, serotonin)2c receptor agonist which is developed particularly to aim human appetite expression. Lorcaserin, a selective serotonin (5HT2c) receptor agonist is capable of suppressing appetite and food intake. Induction of this receptor gives rise to a number of reactions that finally stimulates the release of 2-melanocortin stimulating hormone, which acts on melanocortin-4-receptors to control appetite.3



Conclusion:
Lorcaserin is a novel anti-obesity agent. Since, it is a 5HT2C receptor agonist it may have potential in treating depression, anxiety, Alzheimer’s disease and Parkinsonism. The advantages of lorcaserin are high rate of renal excretion and minimal drug interaction. The common adverse drug reactions are nausea, dizziness, headache, vomiting, and cardiovalvulopathy. The attempt on review of Lorcaserin will pave the way for budding researchers to explore and fill the gaps in analytical methodology which are not so far reported and also useful for physicians and other health professionals to challenge their research on Lorcaserin.
Acknowledgement:
We sincerely thank the Management, Central Library, Publication oversight committee of Sri Ramachandra University for providing necessary scientific sources. The authors are also grateful to authors/ editors/ publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1.Lisa L, Loannider Demos, Loretta Piccenna, John J Meneil. Pharmacotherapies for obesity; past, current and future therapies. Journal of Obesity 2011; Article ID 179674: 18pages.
2.Dick BS Bashim, A K Sharma, Navdeep Dahiya and Anjan Khadka. Lorcaserin; A novel anti-obesity drug. Journal of Pharmacology and Pharmacotherapeutics 2014; 5(2):175-9.
3. Jason C G, Halford and Joanne A Harold. Lorcaserin and the role of 5-HT2c agonist in the treatment of obesity. Clinical Medicine Reviews on Therapeutics 2011; (3): Page 347-354.
4. Lorcaserin. Clinical Pharmacology.[internet database].Gold Standard,inc.,2012.
Available at: http://www.clinicalpharmacology.comaccessed : November 12, 2012.
5.Lorcaserin. Lexi-drugs [database online].Lexi _comp, Inc: November 12, 2012.
6.Belviq [package insert].Woodcliff Lake, NJ: Eisai Inc., Ltd,: 2012
7.Lorcaserin. In: DRUGDEX System [Internent database]. Greenwood village, Colo: Thomson micromedex. Updated periodically.Accessed: Novemer 12: 2012.
8.Lorcaserin Facts and Comparisons [Internet Database]. Wolters Kluwer. Available at: http://online.factsandcomparison.com. Accessed: November 12, 2012.
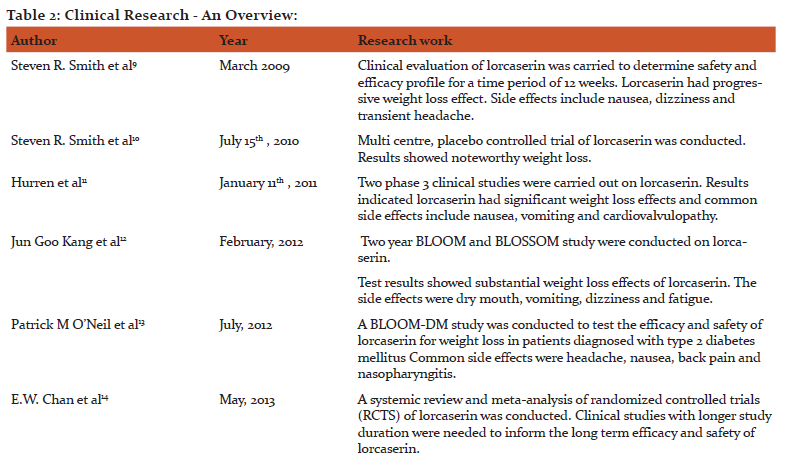
9 Steven R Smith, Warren A Prossor, David , J Donahur, Michael E Morgan, Christen, MAndenon, et al. Lorcaserin (APD356), a selective 5-HT2c agonist, reduces body weight in obese men and women. Obesity 2009 March; 17(3): 494-593.
10. Steven R, Smith MD, Neil J. Weissman M, D Christen, M Anderson. Matilde S Multicenter, Placebo-controlled Trail of Lorcaserin For Weight Management.EnglJmed2010july 15, (363): 245-256.
11. Hurren KM, Berlie HD. Lorcaserin :An investigational serotonin 2C agonist for weight loss. American journal of health system pharm 2011 Nov 11; 68(21): 2029-37
12. Jun Goo Kang, Cheol–Young park. Anti –obesity Drugs :A Review about their effects and safety. Diabetes Metab 2012 Feb 17th; 36(1): 13-25.
13. ONeil PM, Smith SR , Weissman NJ , Fidler MC, Sanchez M ,Zhang J et al. Randomised placebo-controlled clinical trials of lorcaserin for weight loss in type 2 diabetes mellitus: the BLOOM –DM study. Journal of obesity 2012 July; 20(7): 1426-1436.
14.Chan EW, Hey Y, Chui CS, Wong AY, Lau WC, Wong IC. Efficacy and safety of lorcaserin in obese adults:a meta- analysis of 1-year randomized controlled trials (RCTS)and narrative review on short – term RCTS. Obes rev 2013May; 14(5): 383-392.
15. Joshua W, Fleming Katies MC, Clendon Daniel, M Riche. New obesity agents: Lorcaserin and pheneterimine / topiramate. Pharmacother 2013 June 25; 47(7): 1007-1016
16.Neil J Weismam, Matildesanchez, Gary G Koch, steven R smith, William R Shanahan, christen M Anderson. Echo cardiographic assessment of cardiac valvular regurgitation with lorcsaserin from analysis of 3 phase-3 clinical trials. Circulation: cardiovascular imaging 2013 July 16; 6:560-567.
17. Amal A Bajrai, Essam Ezzeldin, Khalid AAl-Rashood, Mohammad Raish and Muzaffar Iqbal. A validated UPLC-MS-MS Assay for the rapid determination of lorcaserin in plasma and brain tissue samples. Journal of analytical toxicology 2015 November 14; 40: 133-139.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License