IJCRR - 4(17), September, 2012
Pages: 67-73
Date of Publication: 14-Sep-2012
Print Article
Download XML Download PDF
In-vitro Estimation of Free Radical Scavenging Activity of Fruit juices by DPPH assay
Author: Singh Meenu, Pragzna Yanamadala, Dharmadev Bommi
Category: Healthcare
Abstract:Objective: A number of different beverage products claim to have antioxidant potency due to their perceived high content of polyphenols. The present study was designed to evaluate the in vitro estimation of free radical scavenging activity of three different fruit juices as follows: Grapes, Guava and Pineapple available in local market. Method: Above mentioned fruit juices were screened for their free radical scavenging property using diphenyl picryl hydrazyl (DPPH) radicals. The absorbance of these fruit juices at 517nm is found by using UV-spectroscopy at various time intervals such as 15 minutes, 30 minutes and 24 hours respectively. Then the absorbance was compared with each other and better antioxidant activity of the fruit juices was estimated. Results: The results obtained from the DPPH assay demonstrated that Grapes juice had better antioxidant activity than Pineapple and Guava juices. Conclusions: The present research demonstrates that although a number of popular beverages have evidence of antioxidant activity, there are clear differences in antioxidant potency. Some beverages with
lower potency would need to be consumed in much larger amounts to equal the antioxidant potency of
Grapes juice.
Keywords: Anti-oxidant activity, DPPH (diphenyl picryl hydrazyl), Grapes, Guava, Pineapple
Full Text:
INTRODUCTION
Clinical trials and epidemiological studies have established an inverse correlation between the intake of fruits and vegetables and the occurrence of diseases such as inflammation, cardiovascular disease, cancer, and aging-related disorders 1 . The defensive effects of natural antioxidants in fruit and vegetables are related to three major groups; vitamin, phenolics and carotenoids and are believed to be the effective nutrients in the prevention of these oxidative stress related diseases 2,3. There is therefore a parallel increase in the use of methods for estimating the efficiency of such substances as antioxidants 4,5. One such method that is currently popular is based upon the use of the stable free radical 2,2-di(4- tertoctylphenyl)- 1-picrylhydrazyl (DPPH) which was an easy and accurate method with regard to measuring the antioxidant capacity of fruit and vegetable juices or extracts 4 .
Pineapple fruit is considered a highly nutritious fruit because it contains a high level of vitamin C, a natural antioxidant which may inhibit the development of major clinical conditions including heart disease and certain cancers 6 . The fruit also contains phenolic compounds and β- carotene 7,8, which constitute natural sources of antioxidants. Guava (Psidium guajava L.) fruit is considered a highly nutritious fruit because it contains a high level of ascorbic acid (50–300 mg/100 g fresh weight), which is three to six times higher than oranges. Phenolic compounds such as myricetin and apigenin 9 , ellagic acid, and anthocyanins 10 are also at high levels in guava fruit. The grapes has been well recognized worldwide for over 2,000 years as one among the edible sweet fruits and recognized for its wide spectrum of biological properties. Resveratrol (3,5,40- trans-trihydroxystilbene) is a natural phytoalexin abundantly found in grapes and red wine, which has potent antioxidant property 11. Thus, red fruit juices such as grapes and others like guava and pineapple have received attention due to their antioxidant activity. Whereas there are numerous phytochemicals consumed in our diet, polyphenols constitute the largest group and have attracted much attention due to their antioxidant properties 12. In fact, the potential health benefits of plant foods are commonly linked to their polyphenol content. Currently, there are a number of commercial ready-to-drink (RTD) polyphenol-rich beverages, which base their marketing strategies on antioxidant potency. However, to the best of our knowledge, data on the direct comparison of antioxidant activity of these widely available leading beverage products have not been obtained. It is of great interest to the general public to know the antioxidant capacity of the beverages that they consume. However, it should be cautioned that because of the inherent complexity of food matrices, the use of one antioxidant capacity method to determine antioxidant potency is ineffective. This is because antioxidants respond to different reactive species in different tests, which is partially attributed to multiple reaction mechanisms and reaction phases 13,14. The aim of the current study was to compare the antioxidant activity of three marketed fruit juices i.e., Grapes, Guava and Pineapple juices by DPPH assay.
Materials and Methods
Ready-to-Drink Polyphenol-Enriched Beverages
The following preparations are obtained from the local provisional market of Hyderabad: Grape, Guava and Pineapple of “Real Company”. All fruit juices were analyzed in late March or early April prior to their expiration dates as stated on their packages. All beverages were kept at storage conditions as specified on their labels prior to analyses. Analysis of Ascorbic acid content (AAC) The AAC was determined by the iodine titration method 15 or the RP-HPLC method: Waters C18 column (3.9×150 mm, 5µm particle size), mobile phase 5% acetic acid (Sd Fine Chem Limited, Mumbai), flow-rate 0.5 mL/min and 254 nm detection wavelength. Analysis of Total phenol content (TPC) TPC was determined using the Folin-Ciocalteu?s reagent 16 (LOBACHEMIE, Mumbai). Samples (0.3 mL, triplicate) were introduced into test tubes followed by 1.5 mL of Folin-Ciocalteu?s reagent (diluted 10 times with water) and 1.2 mL of sodium carbonate, 7.5%w/v (Virat Lab, Hyderabad). The tubes were vortexed, covered with parafilm and allowed to stand for 30 min.
Absorption at 765 nm was measured. If the sample absorbance exceeded 1, the sample was appropriately diluted to give reading less than 1. Total phenol contents were expressed in gallic acid equivalents (mg per 100 g fresh fruit).
Free Radical Scavenging Capacity:The free radical scavenging capacity was analyzed by the DPPH assay 17,18. 2,2-diphenyl-1- picrylhydrazy, DPPH (Santa cruz biotechnology, Inc) is a radical generating substance that is widely used to monitor the free radical scavenging abilities (the ability of a compound to donate an electron) of various antioxidants. The DPPH radical has a deep violet color due to its impaired electron, and radical scavenging can be followed spectrophotometrically by the loss of absorbance at 517 nm, as the pale yellow non radical form is produced 19.
The DPPH assay was typically run by the following procedure: In this method five dilutions of each fruit juice with two replicates were analyzed. Reaction solution was prepared by mixing 50 µL of diluted fruit juice with 300 µL of methanolic
DPPH solution (1mM) and the final volume was brought to 3 mL with methanol (Sd Fine Chem Limited, Mumbai). The solution was kept in dark at room temperature for 15 minutes. The absorbance (Ajuice) was read against the prepared blank (50 µL diluted fruit juice, 2950 µL methanol) at 517nm. A DPPH blank solution was prepared (300 µL of 1mM DPPH solution, 2.7 mL of methanol) and measured. Percent inhibition of DPPH radical was calculated for each dilution of juice according to formula: % Inhibition = [(ADPPH-Ajuice)/ADPPH×100] Where ADPPH is the absorbance value of the DPPH blank solution, Ajuice is the absorbance value of the sample solution. IC50, the concentration of antioxidant required for 50% scavenging of DPPH radical in the specified time period was derived from the % Inhibition vs Concentration plot. Results are shown in table and graphs.
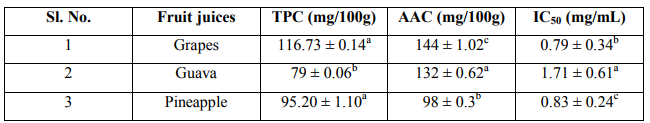
Table 1: Table showing total phenolic content, ascorbic acid content and IC50 of fruit juices of the Real company
RESULTS
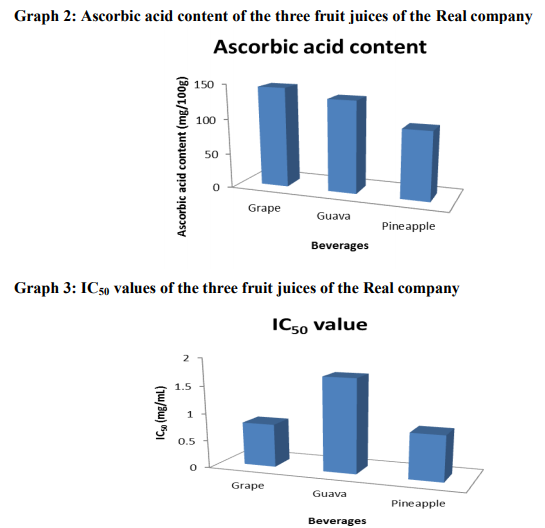
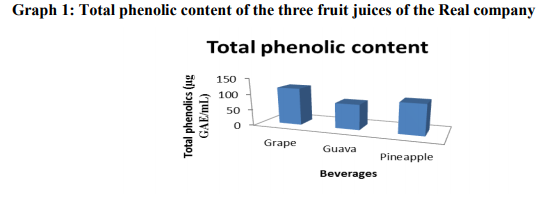
The results of total phenolic, ascorbic acid contents and IC50 of different fruit juices are summarized in Table 1. From the table the total phenolic content was found to be highest in Grapes juice 116.73 ± 0.14 mg/100g followed by Pineapple juice 95.20 ± 1.10 mg/100g and lowest in Guava 79 ± 0.06 mg/100g. The change in colorization from violet to yellow and subsequent fall in absorbance of the stable radical DPPH was measured at 517nm for various concentrations i.e. 50-250 μg/mL. The IC50 value for each fruit extract defined as the concentration of extract causing 50% inhibition of absorbance was calculated, since IC50 is a measure of inhibitory concentration, a lower IC50 value would reflect greater antioxidant activity of the sample. Antioxidant activity among the fruit samples was found to be maximum in Grapes having lowest IC50 value of 0.79 ± 0.34 mg/mL and minimum in Guava having highest IC50 1.71 ± 0.61 mg/mL as lower IC50 value would reflect greater antioxidant activity of the sample.
DISCUSSION
A polyphenol-rich food with health benefits has become a more common element in food marketing these days. The public is highly aware of the term “antioxidant”, which has been defined by the Institute of Medicine of the National Academy of Sciences as follows: “a substance in foods that significantly decreases the adverse effects of reactive species, such as reactive oxygen and nitrogen species, on normal physiologic function in humans.” Therefore, the marketing of many so-called “superfoods” is commonly based on their antioxidant potential. Multiple assays with different sensitivities and specificities for antioxidant activity are being used separately to justify health claims. Consumers have a difficult time distinguishing among the various antioxidant claims for widely available antioxidant beverages. Therefore, the present study was significant in comparing the most commonly available brands of beverages for antioxidant activity using the well-known and established laboratory methods for determining antioxidant capacity. Grapes juice had the highest antioxidant capacity and the most complete antioxidant coverage in vitro which may be contributed to its constituent, phytoalexin. The present research demonstrates that although a number of popular beverages have evidence of antioxidant activity in vitro, there are clear differences in antioxidant potency. The bleaching of the DPPH solution increases regularly with increasing amount of fruit in a given volume of solution. The bleaching action is mainly attributed to the presence of polyphenols and ascorbic acid extracted into the solution. The total phenolic content, IC50, and the ascorbic acid content of the fruit juices are summarized in Table 1, Graphs 1, 2 and 3. For a given amount of fruit, the higher the absorbance, the better is the reducing power. Correlation of IC50 with reducing power: DPPH assay measures the ability of the extract to donate hydrogen to the radical. In DPPH assay the lower the IC50 the better it is able to scavenge the radicals, particularly peroxy radicals which are the propagators of the autoxidation of lipid molecules and thereby break the free radical chain reaction 20. It is observed that grapes having low IC50, is a very potent radical scavenger. In terms of reducing power, grape rank highest but guava is significantly lower than that of pineapple. The high antioxidant potential (as characterized by low IC50 and high reducing power) of grape is attributed to its high TPC and AAC. The low antioxidant potential of guava (IC50 = 1.71 ± 0.61 mg/mL) is due its low TPC and AAC. Pineapple in spite of its relatively high TPC has low antioxidant potential (IC50 = 0.83 ± 0.24 mg/mL). Three possible reasons may be able to account for this: First, it has been reported that 21 reaction of DPPH with certain phenols such as eugenol and its derivatives is reversible, resulting in low readings for antioxidant activity (% disappearance). The second possible reason could be due to the slow rate of the reaction between DPPH and the substrate molecules 13 .The third possible explanation (for the relatively low reducing power) could be that certain phenols in the pineapple juice have a higher redox potential than that of other fruit juices. To clarify this anomaly further work is necessary. Finally, it is also observed that the antioxidant potential correlates well with AEAC.
CONCLUSION
For a body to maintain antioxidant level, external supplementation is necessary for healthy living. From the present research the order of the antioxidant activity in the fruit juices was found to be Grapes>Pineapple>Guava. These fruits can be used as alternative source of natural antioxidant rather than synthetic antioxidant like BHT (Butylated hydroxytoluene) and BHA (Butylated hydroxyanisole) because of carcinogenicity.
ACKNOWLEDGEMENT
We thank everyone who supported this study
References:
1. Swartzberg J, Margen S. Eat, Drink, and be Healthy-The HarVard Medical School Guide to Healthy Eating. Am J Epidemiol 2001;154(12):1160.
2. Ames BN, Gold LS, Willet WC. The causes and prevention of cancer. Proc Natl Acad Sci USA 1995;92:5258-5265.
3. Kaur C, Kapoor HC. Antioxidants in fruits and vegetables-the millennium?s health. Int J Food Sci Technol 2001;36(7):703-725.
4. Sanchez-Moreno C. Review: Methods used to evaluate the free radical scavenging activity in foods and biological systems. Food Sci Tech Int 2002;8(3):121-137.
5. Schwarz K, Bertelsen G, Nissen LR, Gardner PT, Heinonen MI, Hopia A, et al. Investigation of plant extracts for the protection of processed foods against lipid oxidation. Comparison of antioxidant assays based on radical scavenging, lipid oxidation and analysis of the principal antioxidant compounds. Eur Food Res Technol 2001;212:319-328.
6. Diplock AT. Antioxidants and disease prevention. Molecular Aspects of Medicine 1994;15:293-376.
7. Gardner PT, White TAC, McPhail DB, Duthie GG. The relative contributions of vitamin C, carotenoids and phenolics to the antioxidant potential of fruit juices. Food Chemistry 2000;68:471-474.
8. Charoensiri R, Kongkachuichai R, Suknicom S, Sungpuag P. Betacarotene, lycopene, and alpha-tocopherol contents of selected Thai fruits. Food Chemistry 2009;113:202-207.
9. Miean KH, Mohamed S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. Journal of Agricultural and Food Chemistry 2001;49:3106–3112.
10. Misra K, Seshadri TR. Chemical components of the fruits of Psidium guajava. Phytochemistry 1968;7:641–645.
11. Yadav M, Jain S, Bhardwaj A, Nagpal R, Puniya M, Tomar R, et al. Biological and Medicinal Properties of Grapes and Their Bioactive Constituents: an Update. J Med Food 2009;12(3):473-484.
12. Proteggente AR, Pannala AS, Paganga G, Van Buren L, Wagner E, Wiseman S, et al. The antioxidant activity of regularly consumed fruit and vegetables reflects their phenolic and vitamin C composition. Free Radical Res 2002;36:217-233.
13. Huang D, Ou B, Prior RL. The chemistry behind antioxidant capacity assays. J Agric Food Chem 2005;53:1841-1856.
14. Prior RL, Wu X, Schaich K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J Agric Food Chem 2005;53:4290-4302.
15. Suntornsuk L, Kritsanapun W, Nilkamhank S, Paochom, A. Quantitation of vitamin C content in herbal juice using direct titration. Journal of Pharmaceutical and Biochemical Analysis 2002;28:849-855.
16. Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphomolybdic– phosphotungstic acid reagents. American Journal of Enology and Viticulture 1965;16:144-158.
17. Malterud KE, Farbrot TL, Huse AE, Sund RB. Antioxidant and radical scavenging effects of anthraquinones and anthrones. Pharmacology 1993;47:77-85.
18. Nenseter MS, Halvorsen B, Rosvold Q, Rustan AC, Drevon CA. Paracetamol inhibits copper ion-induced, azo compound initiated, and mononuclear cells-mediatedoxidative modification of LDL. Arterioscler Thromb Vasc Biol 1995;15:1338-1344.
19. Seeram NP, Aviram M, Zhang Y, Henning SM, Feng L, Dreher M, et al. Comparison of Antioxidant Potency of Commonly Consumed Polyphenol-Rich Beverages in the United States. J Agric Food Chem 2008;56:1415-1422.
20. Frankel EN. Recent advances in lipid oxidation. Journal of the Science of Food and Agriculture 1991;54:495-511.
21. Bondet V, Brand-Williams W, Berset C. Kinetic and mechanisms of antioxidant activity using the DPPH free radical method. LWT-Food Science and Technology 1997;30(6):609-615.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License