IJCRR - 2(8), August, 2010
Pages: 33-44
Print Article
Download XML Download PDF
PHARMACOLOGICAL PROPERTIES OF BIOFOULING ASCIDIAN, POLYCLINUM MADRASENSIS SEBASTIAN, 1952 FROM TUTICORIN COAST OF INDIA
Author: Bragadeeswaran Subramanian, Ganesan Kittusamy, Prabhu Kolandhasamy, Balasubramanian Thangavel, Meenakshi Kesavan Vithiyanathapuram, Y.Venkateshwaralu
Category: Healthcare
Abstract:Ascidians are sessile, conspicuous and important members of shallow benthic communities having rich source of nitrogenous secondary metabolites and of peptides and alkaloids. In the present study, biofoulant ascidian, Polyclinum madrasensis was collected from the pearl oyster cages of Tuticorin coast. The protein content was showed 790 \?g/ mL of crude extract and molecular weight determination using SDS page indicated well defined bands ranging from 21 to 90 kDa. The methanol and 1:1 Methanol/ Dichloromethane
extracts exhibited strong hemolytic activity on chicken, goat, cow and human erythrocytes (O, A, B and AB groups). The methanolic extract the highest cytotoxicity in brine shrimp (LC50 = 97 \?g/mL). Different polar and non polar solvent extracts were assayed for antibacterial activity against ten human pathogens. The crude compound was characterized by 1H NMR and results represented.
Keywords: Polyclinum madrasensis, biofoulants, protein estimation, hemolysis, cytotoxicity, antibacterial, 1H NMR Study.
Full Text:
Introduction
Nature is a large treasure trove of organic molecules that have countless biological functions. Over billions of years, nature has produced these organic molecules, which have varied uses, some of which are yet to be determined (Kita and Uemura 2006a and b). The Ascidians, commonly called sea squirts (Subphylum: Urochordata, Class Ascidiacea) are dominant organisms in many marine communities, having a wide geographic distribution (Seed and O?Connor 1981). Ascidians contain a wealth of interesting pharmacological substances (Rinehart et al., 2006). One of the reasons for this ecological success is the ability of these animals to synthesise secondary metabolites with important defensive roles, including antimicrobial peptides (Lee et al., 1997), cytosine like compounds (Raftos and Nair 2004), lectins (Green et al., 2003) and antileukemic compounds (Takeara et al., 2008). Saclike filter feeder ascidians have been reported to be an important source in drug discovery. Tetrahydroisoquinolone alkaloid „Ecteinascidin 743? from Ecteinascidia turbinata, cyclic depsipeptides „Dehydrodidemnin B? and „Didemnin B? from Trididemnum solidum,
cyclic peptide „Vitilevuamide? from Didemnin cuculiferum and „Diazonamide? from Diazona angulata are a few tunicate compounds in anticancer preclinical or clinical trials (Jain et al., 2008). In the present study, biofoulant ascidian was collected from Tuticorin coast of India for new pharmaceutical and biomedical substances exploration.
Materials and Methods
Ascidian collection
Bulk samples of Polyclinum madrasensis Sebestian, 1952 (548 gms. in wet wt.) was collected from the cement blocks, pilings and oyster cages of Tuticorin coast (Lat. 8 0 47? 20” and Long. 780 09? 70”), Tamil Nadu, India by SCUBA diving at the depth ranging from 4 to 6 m during June, 2008.
Extraction
The extraction method suggested by Malla Reddy et al. (2005) was followed. The freshly collected ascidian was soaked in methanol at the site of collection until workup. The initial methanol extract was decanted and the ascidian material was extracted with 1:1 methanol: dichloromethane (3 x 0.5 L) at room temperature. The combined extract including initial methanol extract was filtered, and the solvent was removed under reduced pressure to give predominantly an aqueous suspension, which extracted into ethyl acetate (3x 0.5 L), and concentrated under reduced pressure to give a dark brown gummy mass of 10.98 gms. For antibacterial activity, extraction method suggested by Chellaram et al (2004) was followed. The freshly collected ascidians each 100 gms in wet weight were soaked in methonal, dichloromethane, ethanol, acetone,chloroform, n-butanol, ethyl acetate and diethyl ether at the site of collection until workup. The initial respective solvent extract was decanted and repeated for three times (3 x 0.2 L) at room temperature. The combined extract was filtered, and the solvent was removed under reduced pressure to give a dark brown gummy mass. This crude extract was further studied for antimicrobial activity.
Protein estimation
Protein content was estimated by the method of Bradford et al. (1976). The Standard protein sample was prepared at 2 mg/ mL of BSA. The assay relies on the binding of the dye Coomassie Blue G250 to the protein molecule measured calorimetrically at 595 nm. Dilutions of protein standards with concentrations of 20, 40, 60, 80 and 100 μg/100 μL were assayed.
Molecular weight determination - SDS - PAGE
Crude protein of Polyclinum madrasensis was subjected to electrophoresis following the method of Laemmli (1970) in 12 % polyacrylamide slab gels. 50 µg of the crude protein in each case was diluted with sample buffer (3:1) and heated at 94 0C for 30 seconds, then loaded. The Molecular weight markers (Fermentas SMO431 14.4 - 116 KDa) used viz., consisted of Lysozyme (14.4), beta - lactoglobulin (18.4), RE Bsp 981 (25.0), Lactate dehydrogenase (35.0), Glutathione S (29.0), Ovalbumin (45.0), Bovine Serum Albumin (66.2) and Phophorylase b (98.0). 10 µl of the marker was loaded in the extreme right wells and the proteins were loaded subsequently. Upon completion of electrophoresis, the gel was washed gently with distilled water to remove excess SDS, stained in Coomassie Blue R250 (Coomassie brilliant blue R250, 1.25 g; Methanol 227 mL; Glacial acetic acid 46 mL; distilled aqueous to make up to 500 mL) for two hours at room temperature and then destained (Methanol 7 mL; glacial acetic acid 7 mL and distilled aqueous to make up to 100 mL) for 48 hours. Protein bands were visualised as dark blue bands on a light blue background. The samples were solublised in reducing sample buffer and equal amount of protein was loaded into 12 % SDS- Polyacrylamide gel and electrophoresis was carried out at constant current (30 mA). The molecular weight of the documented figure was analysed using the Total Lab Package Version 2.01.
Pharmacological study
The extracts of Polyclinum madrasensis was tested for hemolytic, cytotoxicity using brine shrimp lethality and antibacterial activities.
Hemolytic activity
Methanolic and 1:1 DCM/ MeOH extracts of Polyclinum madrasensis was assayed on chicken, goat, cow and human erythrocytes (A, B, AB and O blood groups) following the method of Pani Prasad and Venkateshvaran, (1997). The chicken, goat and cow blood samples were obtained from the nearby slaughterhouse in Parangipettai, while clinically healthy human blood samples were obtained from local hospital using 2.7 % ethylenediaminetetraacetic acid (EDTA) solution as an anticoagulant at 5 % of the blood volume and brought to the laboratory. The blood was centrifuged thrice at 5,000 rpm for five minutes. 1 % erythrocyte suspension was prepared for hemolysis study.
Cytotoxicity using brine shrimp lethality assay
To determine the toxic effects on Artemia salina (brine shrimp) the method of Meyer et al (1982) was adopted. The extracts were dissolved in 0.01 mL of DMSO and incorporated into 5 mL of sea water (pH= 8.8 and Salinity = 28 ‰) containing ten Artemia sp. Each concentration (10, 20, 40, 60, 80 100 µg/mL) was tested thrice, and a control DMSO was done each time. The vials were maintained under illumination. Survivors were counted after 24 hrs and the percentage of deaths at each dose and control (DMSO and saline water). The LC50 values of brine shrimp were obtained from counts using the probit analysis method described by Litchfield and Wilcoxon (1941).
Antibacterial Activity
Antibacterial activity was carried out by using standard disc diffusion method (McCaffrey and Erdean, 1985; Murugan and Santhana Ramasamy 2003). The following microorganisms, Staphylococcus aureus, Salmonella typhi, Salmonella paratyphi, Klebsiella oxytoca, Klebsiella pneumonia, Vibrio cholerae, Pseudomonas aeruginosa, Escherichia coli, Proteus mirabilis, and Lactobacillus vulgaris were used. The extracts were applied to 6 mm sterile discs in aliquots of 30 µL of solvent, allowed to dry at room temperature and placed on agar plates seeded with microorganisms. The bacteria were maintained on nutrient agar plates and incubated at 37° C for 24 hrs. Zones of growth inhibitions were measured following incubation. All extracts were tested thrice at a concentration of 30 µg disc-1 .
Preliminary characterisation of the extracted compound by 1H NMR 1H NMR
spectra were performed at Bruker Avance 300 MHz NMR Spectrophotometer operating at 400.24 and 100.614 observations. The crude sample was dissolved in DMSO/CDCl3 as solvents, TMS as internal reference. Spectra were acquired using a 5 mm 1H dual tuned probe. Temperature was maintained at 25 0C.
Results
Protein Estimation
The protein content in Polyclinum madrasensis was 790± 0.5 µg/ mL of crude. This result clearly indicates the high amount of protein and other biochemical components present in their body.
Molecular weight détermination - SDS - PAGE
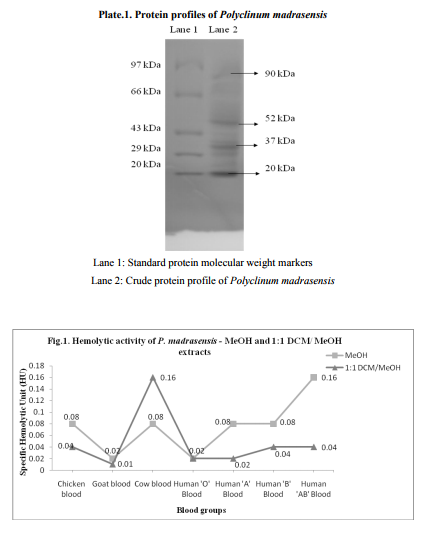
Upon SDS - PAGE on 12 % gel, Crude protein of P. madrasensis yielded 9 bands ranging from 20 to 97 kDa with well defined bands of 20, 37, 52 and 90 kDa (Plate.1).
Biomedical properties study Hemolytic activity
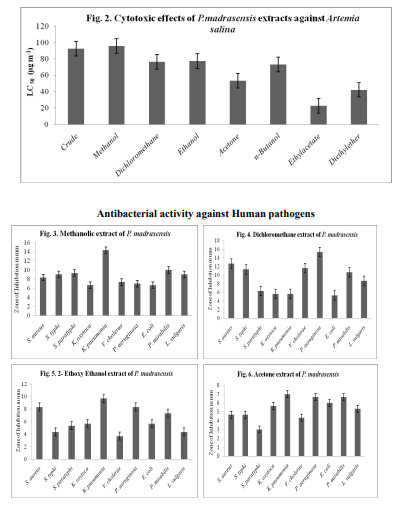
All blood groups showed promising activity. Chicken, goat, cow and A, B, O and AB of human erythrocytes were vulnerable to lysis. The MeOH and 1:1 DCM: MeOH, extracts on human AB blood group and cow blood showed maximum of 0.16 specific Hemolytic Unit (HU) and minimum of 0.01 specific Hemolytic Unit was recorded in goat blood from 1:1 DCM: MeOH extract (Fig. 1)
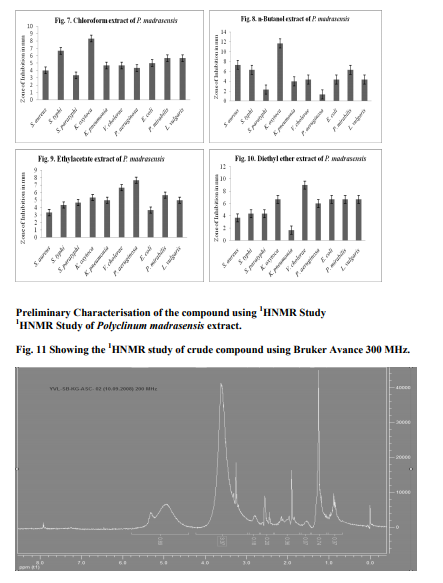
Cytotoxicity using brine shrimp lethality assay Polyclinum madrasensis extracts tested at 10, 20, 40, 60, 80 and 100 µg/mL-1 and showed highest cytotoxicity, indicating the presence of cytotoxic compounds. The LC50 values are given in Fig. 2. The different solvent system extracts showed cytotoxic properties against Artemia salina larvae. The maximum cytotoxicity (97 µgmL-1 ) was observed in MeOH extracts and minimum (20 µgmL-1 ) was observed in ethyl acetate extract.
Antibacterial activity
Fig. 3-10 shows the results of in vitro antimicrobial activity against pathogenic bacteria. The crude extracts showed high and moderate antibacterial activity against 10 pathogens assayed. Among pathogens bacteria tested, P. aeruginosa was the most sensitive against DCM extract (15 mm) and minimum of 5 mm were recorded in diethyl ether and n-butanol extracts against Klebsiella oxytoca and P. aeruginosa respectively.
1H-NMR study
The 1H-NMR spectrum was taken in DMSO D6. From the spectral signal at 7.9 indicates the presence of guanidine and thymidine containing alkaloid, however the compound is to be isolated and confirmed.
Discussion
As far as we know, human consumption of ascidians as a table food occurs in Japan and Chile. Some ascidians (e.g. Halocynthia roretzi) is widely enjoyed as food in Japan, particularly in the Hokkaido and Johoku districts because of the high amount of protein, carbohydrate and other essential micronutrients (Nanri et al., 1992). The amount of carbohydrate, protein, lipid and minerals such as phosphorous and calcium in ascidians is previously reported by (Rajesh and Ali 2008). They also found that the concentration of total carbohydrate was higher in the test (tunic) in their body. This corresponds to the higher amount of cellulose and crude fibre. The present investigation shows that Polyclinum madrasensis contain high amount of protein indicating their high food value and thus surpass many marine food sources in terms of value added marine food.
In the present study, the molecular weight of the protein yielded 9 bands ranging from 21 to 87 kDa with well defined bands of 20, 37, 52, 57and 90 kDa. It supports the previous studies i.e., the tunicate, Styela plicata from Sydney harbour, Australia, analysed for molecular weight of the proteins by SDS PAGE revealed that it contained a single protein of approximately 14 kDa (Nair et al., 2001). Green et al. (2003) demonstrated the molecular weight of protein from hemolymph (43 kDa) of the solitary ascidian Styela plicata from Australian waters. Santos et al. (1992) demonstrated the electrophoresis of the sulfated polysaccharides from different species of ascidians. Chemical analysis of polysaccharides from ascidians reveals sulfate ester, high galactone content and small amounts of glucose and hexosamine in proportions that vary among the different species. The sulfate content may account for part of the variation in electrophoresis motilities as observed in Herdmania momus, Ciona oblonga and C. intestinalis. The endoderm specific alkaline phosphate protein with molecular mass of 86 kDa and 103 kDa were reported by Kumaro et al. (1996) from Halocynthia roretzi from Japan. Hemolytic activity of extracts the MeOH and 1:1 MeOH: DCM extracts on human AB blood group and cow blood showed maximum of 0.16 specific Hemolytic Unit (HU), followed by 0.08 HU observed in MeOH extracts on chicken, cow, human A and B erythrocytes. Minimum of 0.01 specific Hemolytic Unit was recorded in goat blood from 1:1 DCM: MeOH extract. Present study supports the previous reports. i.e., Jimenez et al. (2003) revealed the hemolytic activity of ascidians E. vannamei, Euherdmania sp., D. psammatodes, D. ligulum and Polysyncraton sp. and concluded that lytic protein substances present in these animals, invertebrates and prochordates may be involved in immunity or prey capture. Lee et al. (2001) revealed that hemolytic activity of extracts from the ascidian, Halocynthia aurantium showed 21 % lysis against human red blood cells. The hemolytic activity of tunicate, Halocynthia aurantium disrupted 8 % and 16 % of human O and B erythrocytes respectively (Jang et al., 2002). Gouiffes et al. (1988) demonstrated that the Bistramide A, compound derived from the ascidian, Lissoclinum bistratum from UA islet in New Caledonia, France showed cytotoxicity against Artemia salina larvae in less than 1 µg/ mL. Methanolic and crude extract of P.madrasensis are more toxic and they showed LC50 values of 97 and 95 respectively, than digitalin (LC50 = 151 µg/mL-1 ) and Caffeine (LC50 = 306 µg/mL-1 ) and it is correlated with previous works done by Meyer et al. (1982) and Jimenez et al. (2003). The minimum of (LC50 = 21 µg/mL-1 ) was showed in ethylacetate extracts of Polyclinum madrasensis. The species studied in the present work, Polyclinum madrasensis has not been previously investigated for the presence of cytotoxic compounds from Indian water. The epidioysterol is toxic against A. salina larva, derived from morocco ascidian Cynthia savignyi. The LC50 value showed 71 µg/mL. The percentage of death increased at 10, 30, 50 and 100 µg/ mL for 20 %, 25 %, 42 % and 63 % respectively. Three cyclotetrapeptides were isolated from the ascidian Cystodytes dellechiajei (Polycitoridae), only the valine containing peptide had significant cytotoxic effect and it showed LC50 value of 1.5 µg/mL (Aracil et al., 1991). Extracts of Ecteinascidia turbinata (Phelebobranchia, Perophoridae) exert several biologically significant effects; they are more cytotoxic. A new class of metabolites, the Eudistomins based on the β-carboline ring system, have been isolated from several species of Eudistoma (Ploycitoridae). Antibacterial activity has been previously reported from extracts of some ascidians. From present study, the bacteria tested P. aeruginosa is the most sensitive against 1:1 DCM: MeOH extracts (15 mm) followed by MeOH extract against Klebsiella pneumonia (14 mm) inhibitory activity. Minimum of 5 mm were recorded in diethyl ether and n-butanol extracts against Klebsiella oxytoca and P. aeruginosa respectively. Abourriche et al. (2003) evaluated the antibacterial activity against A. tumifaciens, E.coli, P. aeruginosa and S. aureus from the extracts of Moroccan Atlantic sea ascidian, Cynthia savignyi. It shows that, except for the dichloromethane extract, all extracts were active against bacteria. A. tumifaciens was the most sensitive. Activity of hexane and diethyl ether extracts of this ascidian against A. tumifaciens was slightly less, but higher than the activity of Lissoclinum fragile extracts.
Lissoclinotoxins, on the other hand, are cyclic peptides (lissoclinotoxin A and B), isolated from the tissues of Lissoclinum perforatum. Lissoclinotoxins A and B are powerful antimicrobial agents, with strong activity against a range of bacteria, including ichthyopathogenic strains, such as Aeromonas salmonicida and Vibrio anguillarum, at concentrations between 0.1 to 0.6 µg/mL (Litaudon and Guyot, 1991 and Litaudon et al., 1994). Ascidians have attracted attention as a source of antimicrobial proteins because they are sessile, filter feeding prochordates which hold a phylogenetically strategic position close to the origin of the vertebrate line. They are thus considered to contain antibacterial agents of relevance to either antifouling technology or clinical pharmacology and the tissues of several solitary species have been subjected to broad spectrum screening for bactericidal, antiviral or cytotoxic activity (Rinehart et al., 1983).
Conclusion
All extracts were prepared using the described procedures above and were evaluated for consistency by taxonomic analysis of the ascidians specimens assayed for biomedical properties and preliminary characterisation of the compounds. Much chemical research remains to be done in order to achieve the structural elucidation of the active principles contained in ascidians studied in the present work which demonstrated bioactivity. In these ascidians, the active compounds were present at very low concentrations. Large scale collection will be necessary to obtain sufficient quantities of pure compounds for structural elucidation and further pharmacological evaluation. It is therefore likely that other factors, besides bioactive compounds might play an additional role in the defense mechanisms of the species investigated. Some options for sustainable use of marine resources are chemical synthesis, controlled harvesting, aquaculture of the source organism, in vitro production through cell culture of the macro or microorganism source and transgenic production.
Acknowledgements
Sincere thanks are due to authorities of Annamalai Universities for the necessary facilities provided. The sample processing and NMR spectral studies facilities provided by the Director, Indian Institute of Chemical Technology is acknowledged. Project grant provided by Department of Biotechnology (DBT), New Delhi is gratefully acknowledged.
References:
1. Abourriche A., Y. Abboud, S. Maoufoud, H. Mohou, T. Seffaj, M. Charrouf, N. Chaib, A. Bennamara, N. Bontemps and C. Francisco, 2003. Cynthichlorine: A bioactive alkaloid from the tunicate Cynthia savignyi. I. Framaco., 58: 1351-1354.
2. Aracil, J. M., A. Badre, M. Fadli, G. Jeanty, B. Banaigs, C. Francisco, F. Lafargue, A. Heitz and A. Aumelas, 1991. Nouveaux cyclotetrapeptides isoles de lascidie Cystodytes dellechiajei. Tetrahedron Lett., 32: 2609-2612.
3. Bradford, M. M., 1976. A rapid and sensitive method for the quantification of microgram quantities of protein using the principle of protein dye binding. Anal. Biochem., 72: 248- 254.
4. Chellaram, C., M. E. Gnanambal and J. K. Patterson Edward, 2004. Antibacterial activity of the winged oyster Pteria chinensis (Pterioida: Pteridae). Indian J. Mar. Sci., 33: 369- 372.
5. Gouiffes, D., M. Juge, N. Grimaud, L. Welin, M. P. Sauviat, Y. Barbin, D. Laurent, C. Roussakis, J. P. Henichart and J. F. Verbist, 1988. Bistramide A: A new toxin from the Urochordata Lissoclinum bistratum Sluiter: isolation and preliminary characterization. Toxicon. 26: 1129-1136.
6. Green, P. L., S. V. Nair, D. A. Raftos, 2003. Secretion of a collectin- like protein in tunicates is enhanced during inflammatory responses. Dev. Comp. Immunol. 27: 3- 9.
7. Jain, R., S. Sonawane and N. Mandrekar, 2008. Marine organisms: Potential source for drug discovery. Cur. Sci., 94: 292pp.
8. Jang, W. S., K. N. Kim, Y. S. Lee, M. H. Nam and I. H. Lee, 2002. Halocidin: a new antimicrobial peptide from hemocytes of the solitary tunicate, Halocynthia aurantium; FEBS Letters. 521: 81- 86.
9. Jimenez, P. C., S. C. Fortier, T. M. C. Lotufo, C. Pessoa, M. E. A. Moraes, M. O. Moraes and L.V. Costa- Lotufo, 2003. Biological activity in extracts of ascidians (Tunicata, Ascidiacea) from the north- eastern Brazilian coast. J. Exp. Mar. Biol. Ecol., 287: 93- 101.
10. Kitam, M and D. Uemura, 2006a. In Progress in Marine Molecular Biotechnology. Shellfish Poisons. W E G Muller, H G Schroder (ed.) (Berlin, Heidelberg Verlag Press: Springer) 43: 25-32.
11. Kita, M and D. Uemura, 2006b. In Topics in Heterocycles, Bioactive Heterocyclic Alkaloids from Marine Origin. S Eguchi, (ed.) (Berlin, Heidelberg Verlag Press: Springer); 6: 157-163.
12. Kumaro, G., H. Yokosavva and H. Nishida, 1996. Biochemical evidence for membrane- bound endoderm specific alkaline phosphate in larvae of the ascidian Halocynthia roretzi. Eur. J. Biochem., 240: 485-489.
13. Laemmli, U. K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680- 685.
14. Lee, I. H., Y. Cho and R. I. Lehrer 1997. Styelins, broad spectrum antimicrobial peptides from the solitary tunicate, Styela clava. Comp. Biochem. Physiol., 118: 515- 521.
15. Lee, I. H., Y. Lee, C. Kim, L. T. Hong, L. B. Menzel, J. Pohl, M. Sherman, A. Waring and R. Lehrer 2001. Dicynthaurin: an antimicrobial peptide from hemocytes of the solitary tunicate, Halocynthia aurantium. Biochim. Biophys. Acta., 1527: 141-148.
16. Litaudon, M and M. Guyot, 1991. Lissoclinotoxin A, an antibiotic 1, 2, 3 - trithiane derivative from the tunicate Lissoclinum perforatum. Tetrahedron Lett., 32: 911-914.
17. Litaudon, M., F. Trigalo, M. T. Martin, F. Frappier and M. Guyot, 1994. Lissoclinotoxins: Antibiotic polysulphur derivatives from the tunicate Lissoclinum perforatum Revised structure of Lissoclinotoxin A. Tetrahedron Lett., 50:5323- 5334.
18. Litchfield, J. T and P. J. Wilcoxon 1949. Simplified method of evaluating doseeffect experiments. J. Pharmac. Exp. Ther., 96(2):99-113.
19. Malla Reddy S, Srinivasulu M, Satyanarayana N, Kondapi A K and Y. Venkateswarlu, 2005. New potent cytotoxic lamellarins alkaloids from Indian ascidian Didemnum obscurum; Tetrahedran, 61: 9242- 9247.
20. McCaffrey, E. J and R. Erdean, 1985. Antimicrobial activity of tropical and subtropical sponges; Mar. Biol., 89:1- 8.
21. Meyer, B. N., N. R. Ferrigi, J. E. Putnam, L. B. Jacobson, D. E. Nicolas and J. L. Mclaughin, 1982. Brine shrimp: a convenient general bioassay for active plant constituents. Planta Med., 45: 31- 34.
22. Murugan, A and M. Santhana Ramasamy, 2003. Biofouling deterrent natural product from the ascidian Distaplia nathensis. Indian J. Mar. Sci., 321: 62-164.
23. Nair, S. V., M. Burandt, A. R. L. Hutchinson, D. Raison and A. Raftos, 2001. A C - type from the tunicate, Styla plicata that modulates cellular activity. Comp. Biochem. Physio.129: 11- 24
. 24. Nanri, K., J. Ogawa and T. Nishikawa 1992. Tunic of pyurid ascidian Microcosmus hartmeyeri Oka is eaten locally in Japan. Nanki Seibutu., 34: 135
. 25. Pani Prasad and K. Venkateshwaran, 1997. Microhaemolytic assay, International Training Manual on Advance Techniques in Marine Biotoxinology; CIFE, India. 41pp
26. Raftos, D and S. Nair, 2004. Tunicate cytokine-like molecules and their involvement in host defense responses. Prog. Mol. Subcell. Biol., 34: 165-182.
27. Rajesh M and H. Abdul Jaffar Ali, 2008. Nutritional value and antimicrobial activity of marine ascidian species; Ascidian News. http://depts.washington.edu/ascidian/
28. Rinehart, K. L., J. B. Gloer, G. R. Wilson, R. G. Hughes, L. H. Li, H. E. Renis and J. P. McGovern 1983. Antiviral and antitumor compounds from tunicates. Federation Proceedings, 42: 87- 90.
29. Rinehart, K. L., P. D. Shaw, L. S. Shield, J. B. Gloer, G. C. Harbour, M. E. Koker, D. Samian, R. E. Schwartz, A. A. Tymiak, D. L. Weller, G. T. Carter and M. H. G. Munro, 2006. Marine natural products as sources of antiviral, antimicrobial and anti-neoplastic agents; Pure and Applied Chemistry. 53:795- 817.
30. Santos, J. A., B. Mulloy and D. A. S. Mourao, 1992. Structural diversity among sulphated α- L- galatones from ascidians (tunicates). Studies on the species Ciona intestinalis and Herdmania momus. Eur. J. Biochem. 204: 669- 679.
31. Sebestian V O. A new species of synascidian from Madras. Curr. Sci., 1952; 21: 316- 317.
32. Seed, R and R. J. O'Connor, 1981. Community organization in marine algal epifaunas, Ann. Rev. Ecolog. Syst. 12: 49- 74.
33. Takeara, R., P. C. Jimenez, D. V. Wilke, M. O. Demoraes, C. Pessoa, N. P. Lopes, C. J. L. Lopes, T. M. C. Lotufo and L. V. Costa-Lotufo, 2008. Antileukemic effects of Didemnum psammatodes (Tunicata: Ascidiacea) constituents; Comp. Biochem. Physiol. Part A: Mol. and Integ. Physiol., 151: 391- 398.
34. Thompson, J. E, R. P. Walker and D. J. Faulkner, 1985. Screening and bioassays for biologically active substances from forty marine sponge species from San Diego, California. USA. Mar. Biol., 88: 11- 21.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License