IJCRR - 3(6), June, 2011
Pages: 58-76
Print Article
Download XML Download PDF
PILOT SCALE PRODUCTION OF BIODIESEL FROM MACRO ALGAE COLLECTED FROM COROMANDAL
COAST, EAST COAST INDIA
Author: Ramganesh Selvarajan, Senthil Kumar, Rangasamy Murugesan, Sanniyasi Elumalai
Category: General Sciences
Abstract:Bioenergy is one of the most important components to mitigate greenhouse gas emissions and substitute of fossil fuels. The need of energy is increasing continuously, because of increases in industrialization and population. The basic sources of this energy are petroleum, natural gas, coal, hydro and nuclear. The ability of algae to fix Co2 can also be an interesting method of removing gases from power plants, and thus can be used to reduce greenhouse gases with a higher production algal biomass and consequently higher biodiesel yield. Present research such as biodiesel from macroalgae having species Sargassum ilicifolium and Turbinaria ornata. Biodiesel production (methyl ester) was found maximum in Sargassum ilicifolium (8.01%) and minimum in Turbinaria ornate (7.03%).More over sediment (glycerine, pigments and other elements) were higher in Turbinaria ornate(4.26g) than in Sargassum ilicifolium(2.93g). It was observed that Turbinaria ornata could produce large amount of poly unsaturated fatty acids (PUFA) as the major storage lipid which accounted 80.44% than the Sargassum ilicifolium is about 75.98% respectively. Since intracellular lipids can be enhanced by controlling the conditions such as nutrient limitation, limitation of light and salt stress, it can be expected that the TAG yield of macro algae can further be increased by the same approaches. This may be a First report in India that the biodiesel producing macroalgae, their detailed ultrastructure and the lipid droplets storage. If we go further to demonstrate this model in the coastal India, there will be a larger opportunities to produce cost effective biofuel or
biodiesel and glycerine production as well as the rural employment creation.
Full Text:
INTRODUCTION
Biodiesel is defined as the monoalkyl esters of long chain fatty acids, an alternative for fossil fuel. The most common biodiesel constituent used today is fatty acid methyl esters. Bioenergy is one of the most important components to mitigate greenhouse gas emissions and substitute of fossil fuels. The need of energy is increasing continuously, because of increases in industrialization and population. The basic sources of this energy are petroleum, natural gas, coal, hydro and nuclear. Biodiesel is non toxic completely biodegradable fuel with reduced sulfur emission. The release of sulfur content and carbon monoxide would be cut down by 30% and 10%, respectively, by using biodiesel as energy source. Using biodiesel as energy source, the gas generated during combustion could be reduced, and the decrease in carbon monoxide is owing to the relatively high oxygen content in biodiesel. Biodiesel (monoalkyl esters) is one of such alternative fuel, which is obtained by the transesterification of triglyceride oil with monohydric alcohols. It has been well-reported that biodiesel obtained from canola and soybean, palm, sunflower oil, algal oil as a diesel fuel substitute. The burning of an enormous amount of fossil fuel has increased the Co2level in the atmosphere, causing global warming. Biomass has been focused on as an alternative energy source, since it is a renewable resource and it fixes Co2in the atmosphere through photosynthesis. If biomass is grown in a sustained way, its combustion has no impact on the Co2 balance in the atmosphere, because the Co2 emitted by the burning of biomass is offset by the Co2 fixed by photosynthesis [Macedo]. Among biomass, algae (macro and microalgae) usually have a higher photosynthetic efficiency than other biomass. The ability of algae to fix Co2 can also be an interesting method of removing gases from power plants, and thus can be used to reduce greenhouse gases with a higher production algal biomass and consequently higher biodiesel yield. Micro-algae have received so far more attention with respect to macro-algae as agents for enhanced Co2 fixation due to their facile adaptability to grow in ponds or bioreactors and the extended knowledge on several strains used for fish feeding. Macro-algae are extensively grown and used as food in Asiatic Countries, or as source of chemicals. They are usually collected from natural water basins where they are seasonally available. Only recently they have been considered for energy production, and the potential of some Pacific Ocean strains has been preliminarily studied. In fact algae are the highest yielding feedstock for biodiesel. It can produce up to 250 times the amount of oil per acre as soybeans. In fact, producing biodiesel from algae may be only the way to produce enough automotive fuel to replace current gasoline usage. Algae produce 7 to 31 time greater oil than palm oil. It is very simple to extract oil from algae. The idea of using algae as a source of fuel is not new but it is now being taken seriously because of the escalating price of petroleum and, more significantly, the emerging concern about global warming that is associated with burning fossil fuels. No literature is found regarding present research such as biodiesel from macroalgae having species Sargassum ilicifolium and Turbinaria ornata. Therefore the present work will focus on existing gap between the fossil fuel and Microalgae based Biofuel.
MATERIALS AND METHODS
Study Site
The Present experiment was carried out in the research lab of Department of Plant Biology and Biotechnology, Presidency College (Autonomous), Chennai, India and Sopisticated Analytical Instrumentation Facility (SAIF) Indian Institute of Technology Madras (IITM), Chennai, India.
Macro algae Sample collection
Two different Algae (Sargassum ilicifolium and Turbinara ornata) were collected from the village Vadakadu, Rameshwaram - Taluk, Ramanathapuram, Tamil Nadu, India. It is located at 9.28°N 79.3E. It has an average elevation of 10 meters (32 feet).
Anatomical studies of the Macrolgae:
The Collected Macrolgae was fixed in FAA (Formalin-5ml + Acetic acid-5ml + 70% Ethanol-90ml) for two hours. The materials were washed in distilled water and dehydrated through graded series of Tertiary butyl alcohol (Sass, 1940). Following dehydration, the materials were infiltrated with paraffin wax controlled temperature (55°C). After infiltration, the specimens were cast into paraffin blocks and the blocks were stored in refrigerator for sectioning. Serial sections to the thickness of 6-8μm were prepared with the help of rotary Microtome. The sections were dewaxed and stained with 0.05% Toluidine blue O (O‘Brien et al., 1964) (dissolved in water) for general anatomical studies. Since it is a Meta chromatic dye, it gave good results for studying gross anatomical features of the inner parts.
Nile blue Staining:
For localization of the Lipid Bodies in the Algae, sections were stained with Nile Blue Stain. The sample or frozen sections are fixated in formaldehyde, then immersed for 20 minutes in the Nile blue solution and rinsed with water. For better differentiation, it is dipped in 1% acetic acid for 10–20 minutes until the colors are pure. This might take only 1–2 minutes. Then the sample is thoroughly rinsed in water (for one to two hours). Afterwards, the stained specimen is taken on a microscope slide and excess water is removed. The sample can be embedded in glycerol or glycerol gelatin. Both external and microtome sections were photographed with NIKON Coolpix-8400 Digital camera and NIKON Labphoto-2 microscopes. Magnifications of the micrographs are shown by the scale-bars.
Lipid Extraction and Oil Conversion from Macroalgae
Macroalgae were ground with motor and pestle as much as possible. The ground algae were dried for 20 min at 80°C in a incubator for releasing water. The yield of lipid contents were compared using different solvent system such as, organic solvents (1-butanol, ethanol, n-hexane / Ether), catalyst (NaOH), and different levels of feeding frequency, pH value, temperature, and reaction time. Hexane and ether solution (1:1) were mixed with the dried ground algae and amber colored oil extracts were collected on top of the solution Then the mixture was kept for 24 h for settling.
Biomass collection
The Macroalgal biomass was collected after filtration and weighted.
Evaporation
The extracted oil was evaporated in vacuum to release hexane and ether solutions using rotary evaporator.
Transesterification
Transesterification is the process of exchanging the organic group R" of an ester with the organic group R' of an alcohol. These reactions are often catalyzed by the addition of an acid or base catalyst. The reaction process is called transesterification

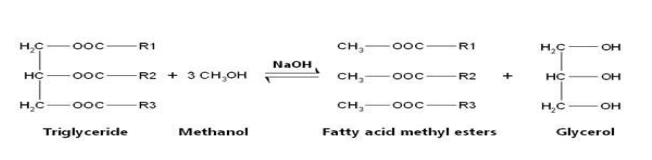
Mixing of catalyst and methanol
NaOH (0.25 g) was mixed with methanol (24 mL) and stirred properly for 20 min. The conical flask containing solution was shaken for 3 h by electric shaker at 300rpm.
Settling
After the manual shaking the biomass solution was kept for 16 h to settle the biodiesel and sediment layers clearly.
Separation of biodiesel
The biodiesel was separated from sedimentation by separating funnel carefully. Quantity sediment (glycerin, pigments, etc.) was measured.
Washing
The collected Macroalgal biodiesel was washed by 5% water until it become clean.
Storage
Biodiesel production was measured by using measuring cylinder; pH was measured and stored at room temperature for further analysis.
Gas Chromatography and Mass Spectroscopic Studies
The collected Macroalgal biodiesel from sample was processed with GC and MS (JEOL GC mate). Lipid fraction was resuspended in nhexane and applied to silica gel column chromatography. Aliphatic hydrocarbon fraction passes through the column fatty acid and carotenoid fractions were trapped. Passing through fraction was defined as hydrocarbon fraction, lipid components in hydrocarbon fraction were identified by GC/MS. The sample (1µl) was evaporated in a split less injector at 300°C. The results were recorded and compared with the Fosssil diesel and gasoline oils. The methyl esters of fatty acids were quantified by a gas chromatograph (Agilant-JEOL GC and MS). The column (HP5) was fused silica 50m x 0.25 mm I.D. Analysis conditions were 20 minutes at 100°C the 3°/ min to 235°C for column temperature, 240°C for injector temperature, helium was the carrier gas. The weight percentages of fatty acids were approximated by the area of the detector response. The fatty acid methyl esters were identified by gas chromatography coupled with mass spectrometry.
Fourier Transform Infra-Red Spectrometry
In our study Perkin Elmer model spectrum-I PC was used. FT - IR spectra (Resolution: 4 cm-1, Scan Number: 3) were performed after evaporation of the Lipid fraction on the Thalium bromide tablets.
RESULTS AND DISCUSSION
Percent Dry weight of algae before oil extraction was lower in Sargassum ilicifolium than in Turbinaria ornata (Table 1). However Extracted oil was higher in Sargassum ilicifolium than in Turbinaria ornata. However biomass after oil extraction was found maximum in Turbinaria ornata than the Sargassum ilicifolium (Table 1). Biodiesel production (methyl ester) was found maximum in Sargassum ilicifolium and minimum in Turbinaria ornata.More over sediment (glycerine, pigments and other elements) were higher in Turbinaria ornata than in Sargassum ilicifolium (Table 1). There is no significant difference in pH between both species. In (Fig 5) photograph shows esterification (biodiesel and residual layer) and biodiesel.
Sargassum ilicifolium The Macroalgal body consist of flat leaf like lateral appendages central thick stipe. The stipe or stem has an epidermal layer of squarish cells, wide, thin walled, less compact cortical tissues and central core of thick walled angular medulla.
Sections stained with Nile Blue
Epidermal cells stain dark and Cortical cells and medullary cells stain purple. Darkly stained Lipid bodies are seen in the cells of the medulla. (Fig. 1.1, 1.2.)
Sections stained with Toludine Blue O
The epidermal cells of macroalgal stain dark. The palisade (cortical) cells and the medulla stain dark violet. The oil droplets stains dark in color. (Fig. 2.1, 2.2)
Turbinaria ornata
The plant body is solid cylinder and appears lobed in transactional view. The epidermis is thin with small squarish cells. The cortical zone comprises fairly large, angular, thin wall compact cells. Medulla is wide and includes small, slightly thick walled compact cells.
Nile Blue Stain
Nile Blue stains cortex and medulla dark purple. The cell wall appears thick. Within the medullary cells are seen some granular or crystalline bodies. This is the area where oil droplets accumulates during stressful condition. (Fig. 3.1, 3.2)
Toludine Blue O
In Toludine blue staining, the epidermis appears dark. The cortex and medulla appears bright purple. The cells inclusions are not visible (Fig. 4.1, 2).
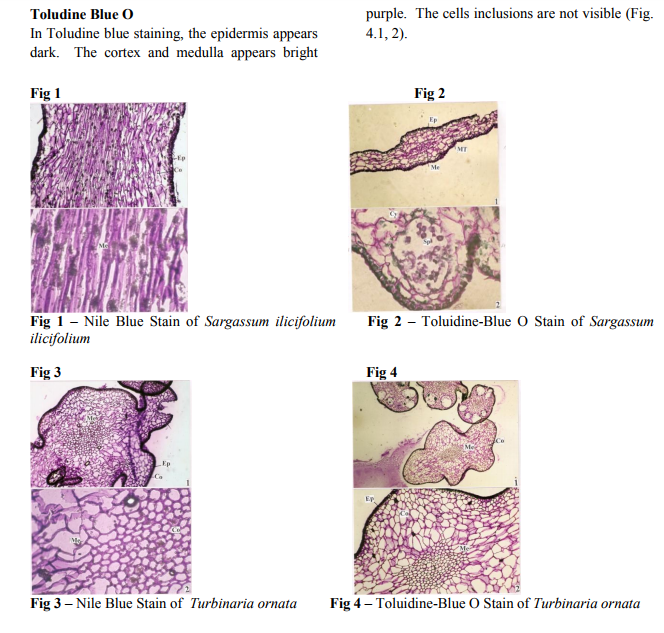
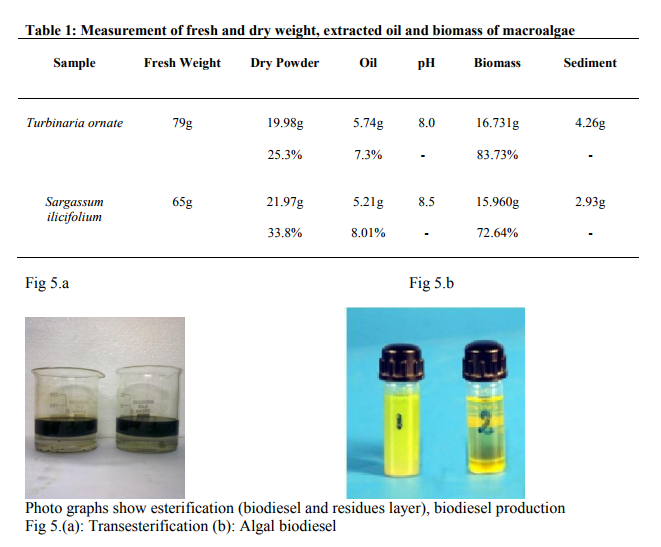
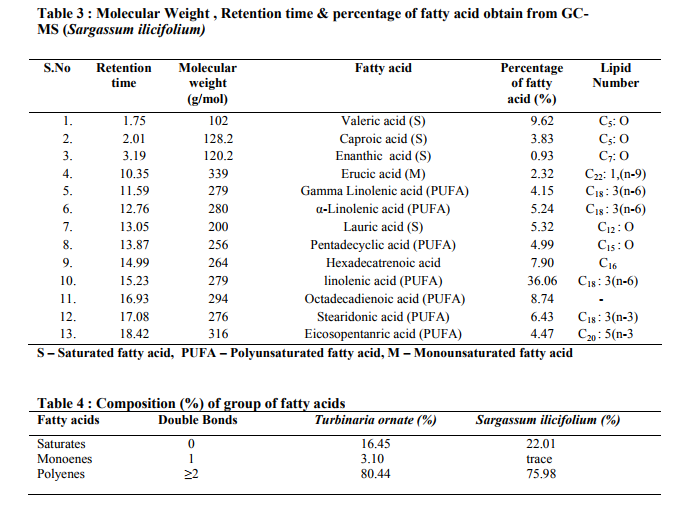
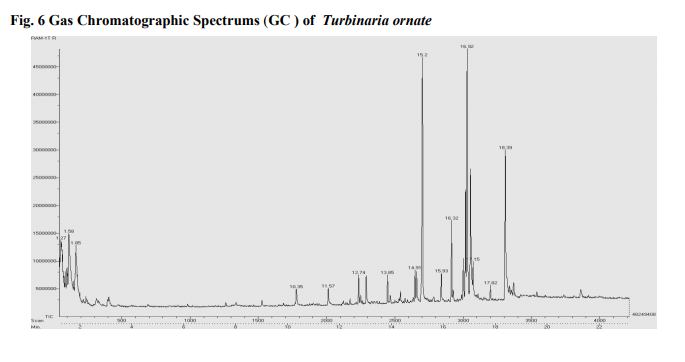
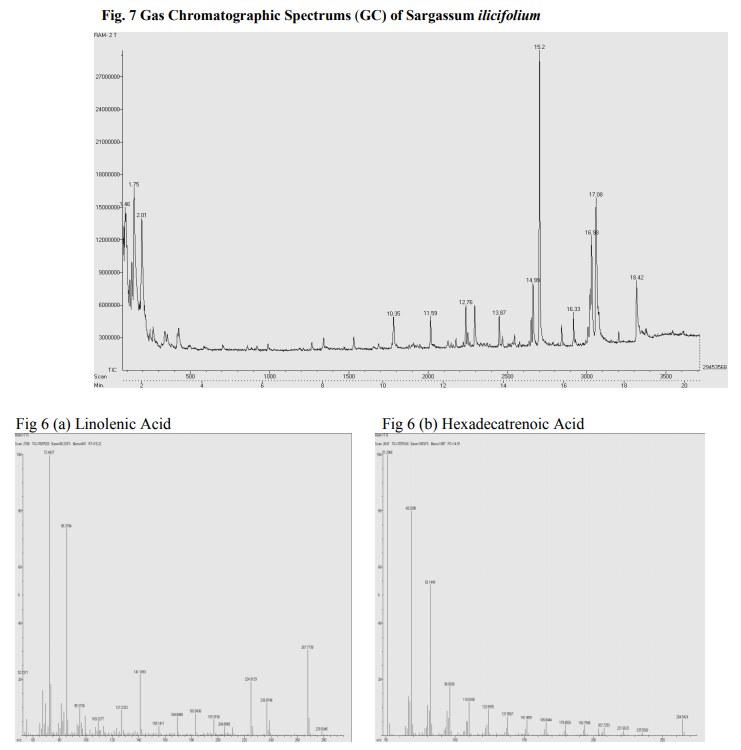
Photo graphs show esterification (biodiesel and residues layer), biodiesel production Fig 5.(a): Transesterification (b): Algal biodiesel Biodiesel can be produced from macro algae because of lipid contents. Sijama stated that docosahexaenoic acid (DHA) was a polyunsaturated fatty acid composed of 22 carbon atoms and six double bonds that belonged to the so called ω-3 group. They also reported that fish oil was the major source of DHA, but alternatively it might be produced by using of organisms. Marine organisms might contain large quantities of DHA and were considered a potential source of this important fatty acid. Some of these organisms could be grown heterotrophically on organic substrates without light. It has been reported that macro algae contain lipid content of 1.3-7.8% (dw). In addition in heterotrophic condition lipid content can be more in algae. The samples harvested are expected to produce lipid fractions containing Omega-3 fatty acids. After extraction and esterification to form the methyl esters, gas chromatographic analyses show (Fig 6 and 7) that the ω-3 fatty acids may constitute as much as higher of the total fatty acid fraction. They are generally contained in phospholipids, glycolipids, mono, or triglycerides and sulfolipids, or as the free acids, but are not limited to these forms (Long, T.V.,) The composition and structure of fatty acid esters, such as unsaturation degree and carbon.
chain length, determine the properties (e.g., cetane number, viscosity, cold flow, oxidative stability, and iodine value) of biodiesel.We therefore analysed the fatty acids of individual lipid classes from both Turbinaria and Sargassum. The fatty acid profiles of two Macro algae are presented (Table 2 and 3) respectively. However, saturated fatty acids (SFA), monounsaturated fatty acids (MUFA), and polyunsaturated fatty acids (PUFA) displayed a significant interactive effect of growth modes.
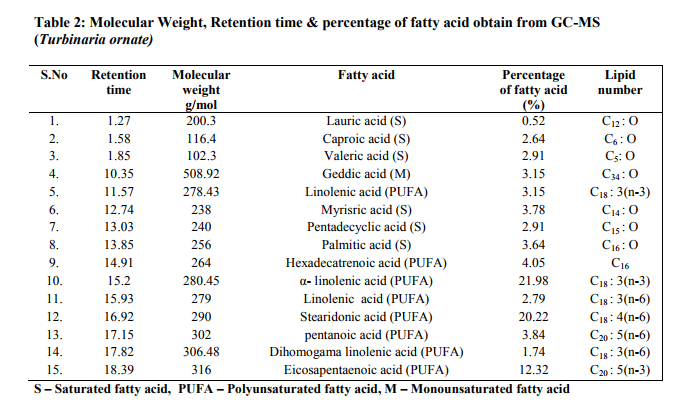
The overall distribution of total fatty acid (TFA) in the lipid classes of both macro algae is depicted (Table 4). It was observed that Turbinaria ornata could produce large amount of poly unsaturated fatty acids (PUFA) as the major storage lipid which accounted 80.44% than the Sargassum ilicifolium is about 75.98% respectively. Since intracellular lipids can be enhanced by controlling the conditions such as nutrient limitation, limitation of light and salt stress, it can be expected that the TAG yield of macro algae can further be increased by the same approaches.
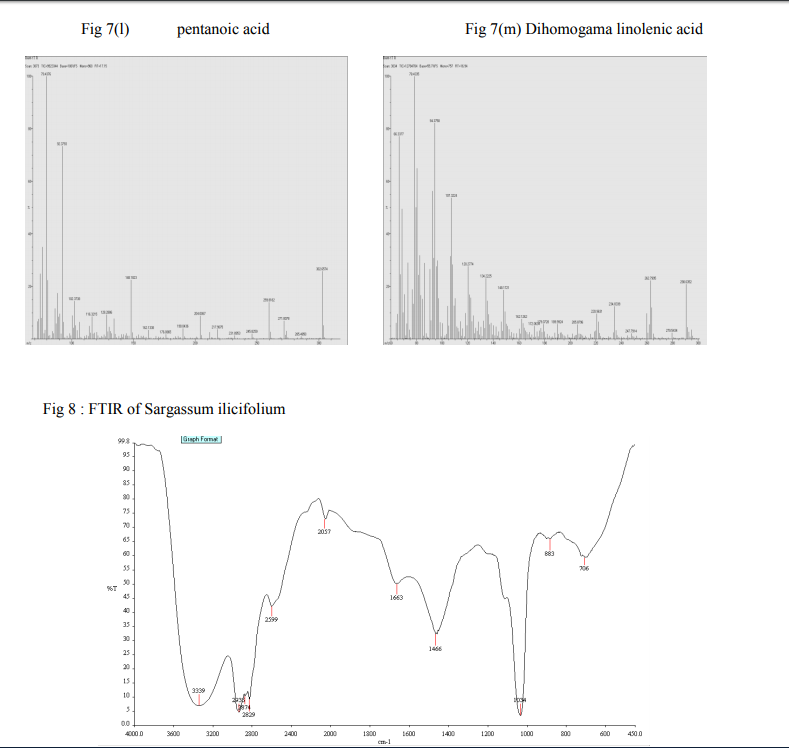
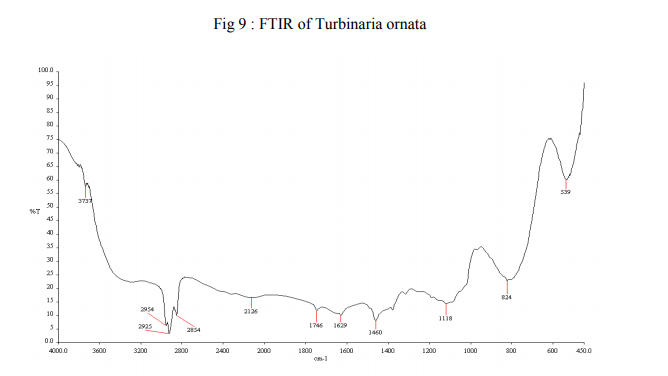
Fourier Transformer InfraRed (FTIR) gives five maxima of the generation of volatile products can be found in the spectrum. All collected extractions give bands at 674 and 2957 cm-1 (Fig ) so all are cis isomer, as expected from algal lipid because trans isomers have a strong nearby 970 cm-1 band and a weak nearby 3012 cm-1 while cis isomers gave medium nearby 720 and 3012 cm-1.bands An analysis of the IR spectrum obtained (Fig. 8 and 9) for the main composition stage, reveals the existence of the absorption bands characteristic of these five different bonds: • C?O: The main characteristic of the IR spectra of carbonylic compounds (aldehydes, acids, etc.) is the strong C?O stretching absorption band in the region of 1870-1540 cm-1. In the case of esters, this band appears in the 1750–1735 cm-1 • C-O-C: corresponding to ethers. These stretching vibrations produce a strong band in the 1200–900 cm-1 region. • C-H: absorption bands characteristic of the vibrations of C-H bonds, as an example, 2960 and 2875 cm-1 correspond to the asymmetric and symmetric vibrational modes of methyl groups, respectively, and 2929 and 2850 cm-1 correspond to the asymmetric and symmetric vibrational modes of methylene groups, respectively. • CO2: they produce strong bands in between 2800-2000 cm-1 as well as in 700 cm-1 region.
•H2O: the adsorption bands of water can be observed in the range of 1800-1200cm-1
CONCLUSION
Algae are an economical choice for biodiesel production, because of its availability and low cost. In this paper we have presented the preliminary results of an ongoing study on biodiesel produced from two macroalgae Sargassum ilicifolium and Turbinaria ornata. Many researchers reported that microalgae might better for higher biodiesl production. But research has not done yet in this regard. Till date there is no adequate research work or publications in India regarding macroalgal based biofuel Production. Our experimental results recorded maximum biodiesel production from macroalgae that confirms the previous findings. This may be a First report in India that the biodiesel producing macroalgae, their detailed ultrastructure and the lipid droplets storage. If we go further to demonstrate this model in the coastal India, there will be a larger opportunities to produce cost effective biofuel or biodiesel and glycerine production as well as the rural employment creation.

References:
1. Ackman,R.G., Tocher,C.S., McLachlan,J., 1968. Marine phytoplankter fatty acids. J. Fisheries Res. Board Canada, 25, 1603– 1620.
2. Chisti, Y., 1980-1981. An unusual hydrocarbon. J.Ramsay Soc., 27-28: 24-26.
3. Elumalai,S., 2010.Comparision of lipid production in microalgae from unique environment of ancient temple tank in tamil nadu (india) under day and light conditions.(under press).
4. E.W. Wilde, J.R. Benemann, Bioremoval of heavy metals by the use of micro-algae, Biotechnology Advances 77 (1993) 781- 812.
5. Goldemberg, J., 2000. World Energy Assessment, Preface.United Nations Development Programme, new York, NY, USA.
6. J. Sheehan, T. Dunahay, J. Benemann, P. Roessler. A Look Back at the U.S. Department of Energy's Aquatic Species Program-Biodiesel from Algae. National Renewable Energy Laboratory, Golden, CO, 1998. 80401 NREL/TP-580-24190.
7. Kapdan, I.K. and F. Kargi, 2006. Biohydrogen production from waste materials. Enzyme Microbiol. Technol., 38: 569-82.
8. Knothe, G., 2005. Dependence of biodiesel fuel properties on the structure of fatty acid alkyl esters. Fuel Process. Technol. 86, 1059-1070.
9. Kulkarni, M.G. and A.K. Dalai, 2006. Waste cooking iol-an economical source for biodiesel: A review. Ind. Eng. Chem. Res., 45: 2901-2913.
10. K. Gao, K.R. Mckinley, Use of macro-algae for marine biomass production and CO2 remediation: a review, Journal of Applied Phycology 6 (1994) 45-60.
11. Lang, X., A.K. Dalai, N.N. Bakhshi, M.J. Reaney and P.B. Hertz, 2002. Preparation and characterization of biodiesels from various Bio- Oils. Bioresour. Technol., 80: 53-62.
12. Long, T.V., 1989. Microorganism prodution of Omega-3 (N-3) lipids. World International Property Organization. International Search Report. LU, USA.
13. Macedo, I.D.C., 199. Energy from biomass and wastes. Biomass Bioenergy, 3: 77-80.
14. Miao X., Wu Q., 2006. Biodiesel production from heterotrophic microalgal oil. Bioresour Technol 97:841-846. doi:10.1016/ j.biortech.2005.04.008
15. Milne TA., Evans RJ., Nagle N., 1990. Catalytic conversion of microalgae and vegetable oils to premium gasoline, with shape selective zeolites. Biomass 21:219- 232. doi:10.1016/0144-4565 (90)90066-S.
16. M. Aresta, A. Dibenedetto, I. Tommasi, E. Cecere, M. Narracci, A. Petrocelli, C. Perrone, The use of marine biomass as renewable energy source for reducing CO2 emissions, October 2002. Elsevier, Special Issue Dedicated to GHGT-6, Kyoto;
17. M. Aresta, Energy from marine biomass, Polish Journal of Environmental Studies II (Suppl. I) (2002) 35- 40;
18. M. Aresta, A. Dibenedetto, G. Barberio, Utilisation of macroalgae for enhanced CO2 fixation and energy production, Preprints of Papers-American Chemical Society. Division of Fuel Chemistry 49 (1) (2004) 348- 349.
19. Renaud, S. and J. Luong-Van, 2006. Seasonal variation in the chemical composition of tropical australian marine macroalgae. J. Applied Phycol., 18: 381- 387.
20. Sawayama, S., S. Inoue, Y. Dote and S.Y. Yokoyama, 1995. CO2 fixation and oil production through microalga. Energy Convers Manage., 36: 729-31.
21. Shay, E.G., 1993. Diesel fuel from vegetable oils: Status and Opportunities. Biomass Bioenergy, 4: 227-242.
22. Shay, E.G., 1993. Diesel fuel from vegetable oils: Status and Opportunities. Biomass Bioenergy, 4: 227-242.
23. Sijtsma, L. and M.E. Swaaf, 2004. Biotechnological production and applications of the ω-3-polyunsaturated fatty acid docosahexaenoic acid. Applied Microbiol. Biotechnol., 64: 146-153.
24. Tadashi Matsunaga., Mitsufumi Matsumoto., Yoshiaki Maeda., Hiroshi Sugiyama., Reiko Sato., Tsuyoshi Tanaka., 2009 Characterization of marine microalga, Scenedesmus sp Strain JPCC GA0024 toward biofuel production, Biotech lett DOI 10.1007/s10529-009-0029-y.
25. Yusuf, C., 2007. Biodiesel from microalgae. Biotechnol. Advances, 25: 294-306.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License