IJCRR - 3(9), September, 2011
Pages: 141-148
Print Article
Download XML Download PDF
BIOCHEMICAL STUDIES ON BIODEGRADATION OF COPPER ON INDIAN MAJOR CARP CATLA CATLA BY
PSEUDOMONAS PUTIDA
Author: Ruckmani Devi S, Jayamathi Govindaraj
Category: Healthcare
Abstract:Copper is an essential trace element that is vital to the health of all living things (humans, plants, animals, and microorganisms). Copper ions are quite toxic to fish when concentrations are increased in environmental exposures often resulting in physiological, histological, biochemical and enzymatic alterations in fish, which have a great potential to serve as biomarkers. Pseudomonas putida has the unique ability to transform a toxic pollutant into a biodegradable product. The objective of this study was to examine the copper toxicity in Catla Catla, and the efficiency of Pseudomonas putida on copper biodegradation was assessed by biochemical parameters such as total protein, carbohydrate, phosphatase, protease, lipase, transaminases, lipid peroxidation, reduced glutathione and copper content and histopathology of muscle tissues of Catla Catla. To conclude that Pseudomonas putida effectively reduced the copper toxicity on Catla Catla.
Keywords: Lipid peroxidation, Transaminases, Oxidative stress, Histopathology
Full Text:
INTRODUCTION
The discharge of industrial effluents and urban wastes into aquatic system leads to contaminate the environment and physiological activities of living things in it1 . Copper is an essential trace metal for several fish metabolic functions such as for the activation of variety of enzymes involved in physiological processes which includes cellular respiration, free radical defense, neurotransmitter function, connective tissue biosynthesis etc2,3. This metal can exert adverse toxicological effects, when the concentration exceeds the capacity of normal physiological level in the body4 . High concentration of this heavy metal was detected in some aquatic ecosystems collecting vineyard run off water and it is also highly concentrated in ground water. Heavy metal contamination has been reported in aquatic organisms5 . Fishes are widely used to evaluate the health of aquatic ecosystems and the associated physiological changes serve as biomarkers of environmental pollution6 . Common Carp (Cyprinus Carpio), Prussian Carp (Carassius gibelio) and peppered Cory (Corydoras Palaeatus) were evaluated as target species to perform genotoxicity and other tests for assessing the heavy metal toxicity. Among this, Common Carp (Catla Catla) is an important commercial species around the world to feed the population. So we have chosen this variety to assess the copper toxicity in aquatic system7 . Copper is a required trace element for many organisms, yet it can exert an inhibitory effect on bacterial growth at relatively low concentrations. However, there are some bacterial species that can tolerate high levels of copper. It also has been reported that copper resistance is plasmid-encoded in Escherichia coli, Proteus vulgaris and a Pseudomonas syringae isolate. It is not known if copper is effluxed from the cell, detoxified by binding to copper-binding proteins, or binds to cell-surface components. Actual data on copper transport in bacteria is also lacking, indicating that this area of research deserves serious attention8 . Pseudomonas putida induces plant growth and protects the plants from pathogens. Because Pseudomonas putida assists in promoting plant development, researchers use it in bioengineering research to develop biopesticides and to the improve plant health. There is of great interest in sequencing the genome of Pseudomonas putida has the unique ability to transform a toxic pollutant into a biodegradable plastic9,10 . The bioremediation of heavy metals using microorganisms has recieved a great deal of attention in recent years, not only as a scientific novelty but also for its potential application in industry11 . A novel attempt is made to study the copper toxicity in Catla Catla, and the efficiency of Pseudomonas putida towards bioremediation by assessing various biochemical parameters and copper toxicity in different concentration was also studied.
MATERIALS AND METHODS
Study material, Catla Catla was collected from the local ponds near Pattukkotai, Tanjore District, Tamilnadu were acclimatized to the laboratory conditions and feeding schedule during 2009-2010.
Feed
Feed was given to the fishes daily 1hr from 10 am -11am, after that the left over fish meals were removed. The experiments were conducted for the period of 20 days. Water was changed in alternated days. Triplicate of the control and experimental ones were conducted simultaneously.
Heavy Metal
Copper was prepared at 2ppm concentration.
Acute toxicity study
Different concentrations of copper (1ppm/l, 2 ppm/l, 3 ppm/l, 4 ppm/l, 5ppm/l) were taken in a cylindrical aquarium containing 20 l of water. The fishes were observed for behavioral changes and mortality percentage was observed at different durations of exposure such as 24h, 48h, 72h, 96h etc. After the experimental period the mortality percentage of Catla Catla was assessed in different concentrations of copper on different exposure time. Pseudomonas putida was cultured and harvested according to12
Experimental design and protocol Group 1( n = 10) : Control (Aquarium tank containing 20 l water + 1 kg of sterilized soil) Group 2 ( n = 10) Fishes were introduced in the aquarium tank containing 20 l of copper (2 ppm/l) treated water + 1 kg of sterilized soil Group 3 ( n = 10) : Fishes were introduced in the aquarium tank containing 20 l water + 1 kg of sterilized soil + 50ml of Pseudomonas nutrient broth Group 4 ( n = 10) : Fishes were introduced in the aquarium tank containing 20 l of copper (2 ppm/l) treated water + 1 kg of sterilized soil + 50ml of Pseudomonas nutrient broth. After the 20th day of experimental period, the fish muscles were dissected out according to the method of Lockwood et al, (1977) and the efficiency of Pseudomonas putida towards bioremediation was assessed by assaying various biochemical parameters such as total protein13 and carbohydrate14, the enzyme assays includes phosphatase15, protease16, lipase17, aspartate transaminases (AST)18, alanine transaminases (ALT)19, lipid peroxidation20 and reduced glutathione21, and also the copper level22 in muscle tissue.
Histological manifestations
Fish liver was dissected and the histopathological manifestations were carried out.
RESULTS
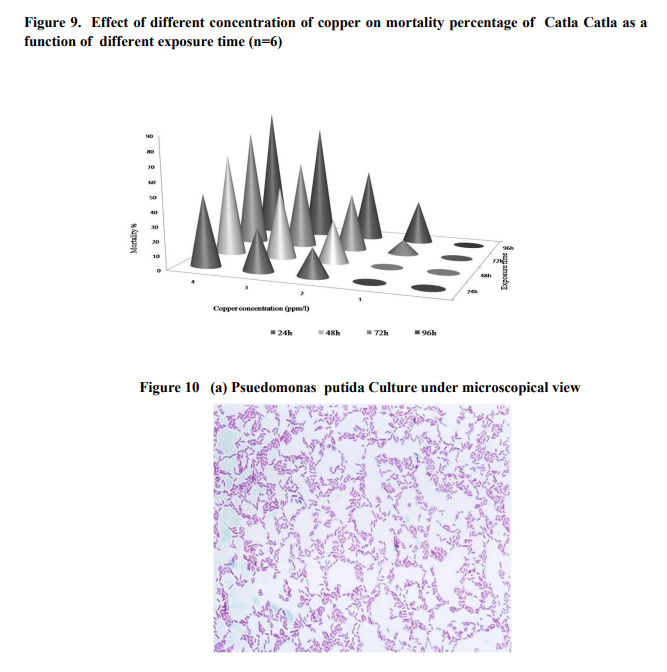
Effect of different concentration of copper on mortality percentage of Catla Catla as a function of different exposure time was given in Figure 1. The mortality percentage decreased with decreased concentration of heavy metals of copper. Figure 2 (a,) showed the mass culture of Pseudomonas Putida nutrient broth . Table 1. represented the biochemical levels of total protein, carbohydrate content, phosphates, protease, lipase, AST, ALT, lipid peroxidation, reduced glutathione and copper. Total protein content of Catla Catla treated with 2ppm /l concentration of copper (Group 2) was reduced (12.8 mg/g) when compared to Group 1 (24.7 mg/g) and Group 3 (15.9 mg/g) was observed closer to normal. Pseudomonas treated groups (Group 4) showed significant increase of total protein (22.3 mg/g). Carbohydrate content of Catla Catla treated with 2ppm /l concentration of copper (Group 2) was reduced (9.2mg/g) when compared to Group 1 (10.5 mg/g) and Group 3 was observed to be normal. Pseudomonas treated groups (Group 4) showed significant increase of carbohydrate content (10.4 mg/g) Activities of enzymes such as phosphatase, protease, lipase, ALT and AST levels were found to be reduced (Group 2), compared to control (Group 1) and was reverted back to normal in Group 4. No such alterations were found in Group 3. Phosphatase levels in Pseudomonas treated groups (Group 4) was (16.6mg/g) closer to Group 1 (18.7mg/g) whereas it was reduced in Group 2 (15.5 mg/g). Protease levels were found to be reduced (4.5 mg /g) in Group 2, compared to control (6.8 mg /g) and was reverted back to normal in Pseudomonas treated groups (5.9 mg /g), in Group 4. No such alterations were found in Group 3. Lipase levels were found to be increased (2.05mg /g) in Group 2, compared to control (1.08 mg /g) and was reverted back to normal in Pseudomonas treated groups (1.5 mg /g), in Group 4. No such alterations were found in Group 3. AST levels were found to be reduced in Group 2 (3.36mg /g), compared to Group 1 (4.52 mg /g) and was reverted back to normal in Group 4 (3.8 mg /g). No such alterations were found in Group 3 (4.2 mg /g) whereas, ALT levels were found to be increased in Group 2 (0.69 mg /g), compared to Group 1 (0.44 mg /g) and was reverted back to normal in Group 4 (0.5 mg /g). No such alterations were found in Group 3 (0.4 mg /g). Lipid peroxidation was observed in group 2 (0.75 mg /g) by decreased levels of lipid peroxides and the Pseudomonas treated groups (Group 4) showed significant increase (0.8 mg /g) in lipid peroxidation levels. An imbalance encountered between oxidant antioxidant ratio, reduced glutathione showed significant reduction in group 2 (0.52 mg /g) than control fishes (Group 1 (0.78 mg /g)). Pseudomonas treated groups (Group 4 (0.6 mg /g)) showed significant reduction. Copper was stagnant in group 2, and the Pseudomonas treated groups showed reduction in copper levels due to Pseudomonas.
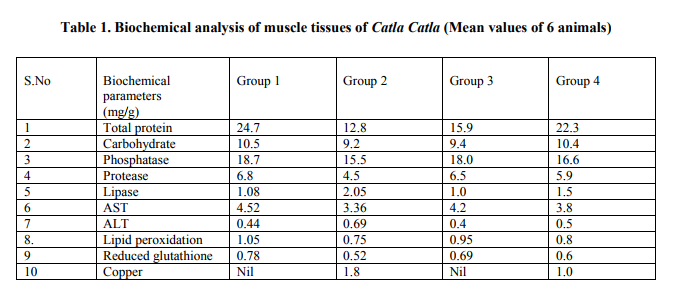
Figure 3 (a,b,c,d) represented the histopathological observation of liver tissue. Control (Group1) individuals showed normal architecture. Group 2 showed irregular shaped nuclei, nuclear hypertrophy, nuclear vacuolation and the presence of eosinophilic granules in the cytoplasm. Since Pseudomonas putida is very similar to strains of Pseudomonas aeruginosa, an opportunistic human pathogen, liver tissue of Group 3 was slightly to moderately damaged but Group 4 showed regeneration of liver cells
DISCUSSION
The fishes showed marked change in their behavior when exposed to different concentrations of copper. In lower concentration of copper (2& 4ppm/l), the fishes showed rapid swimming than control. At highest concentration (6,8 &10 ppm/l) showed lethargic and erratic swimming due to loss of equilibrium. At the time of death transient hyperactivity was also observed23. Mortality percentage was maximum at highest concentration of copper (5ppm/l) at highest exposure time (96 h). It might be due to cumulative action of copper at various metabolic sites. 24observed the alterations in protein content , ALT and AST levels, and explained that the fishes were responding to various stressors by a series of biochemical and physiological stress reactions. Enzymes responsible for proteincarbohydrate metabolism (ALT and AST) showed low levels than control group due to its sharing in transforming proteins to glycogen. 25explained the cytotoxicity of excessive copper accumulation in the liver of sheep and showed high copper status might have induced lowest levels of GSH.A wide variety of xenobiotics including aromatic carcinogens, heavy metal ions were sequestered by enzymes. When the load of foreign material exceeds the efficiency of detoxication, the integrity of cell membrane could be affected adversely cause leakage of enzymes. In copper toxicity, the copper subsequently transformed the enzymes to biologically inactive state. The present study was in agreement with this report. 26Histopathological report showed structural dearrangement in group 2 due to copper toxicity (exposed to copper) and Group 3 (Pseudomonas treated) due to Pseudomonas putida since it was an opportunistic human pathogen, Group 4 showed regeneration of liver tissues due to Pseudomonas putida has the unique ability to transform a toxic pollutant into a biodegradable product. The metal ions as transition metals cause cellular damages which in turn leads to the production of lipid peroxide radicals27. To counteract the deleterious effects produced by the free radicals antioxidant are utilized and leads to cause oxidant antioxidant imbalance. Pseudomonas putida has been a pivotal instrument for studies on the bioremediation of soils contaminated with xenobiotic compounds. The findings of the present study suggested that P. putida is also adapted to thrive in environments with copper toxicity28 .
CONCLUSION
Heavy metal toxicity resulted in adverse effect on muscle tissue components and altered metabolic changes served as a marker for the toxicity of sublethal concentrations of metals as well as pollutants. However, complementary studies are necessary for better understanding of the deleterious effects.
References:
1. Shakoori. A. R, and Muneer.B,[2011] Copper-resistant bacteria from industrial effluents and their role in remediation of heavy metals in wastewater.Folia Microbiologica, 47, (1): 43-50.
2. Saxena. D, Joshi. N and Srivastava. S[ 2002] Mechanism of Copper Resistance in a Copper Mine Isolate Pseudomonas putida Strain S4. Current Microbiol,45,( 6).
3. WHO[1998] Copper: Environmental Health Criteria 200. IPCS- International Programme on Chemical safety, World Health Organisation, Geneva.
4. Pelgrom,S.M.G.J, Lock.R.A.C, M.Balm and S.E.Wendelaar Bonga[1995]Integrated physiological response of tilapia, Orechromis mossambicus to sublethal copper exposure. Aquat. Toxicol,32:303- 320
5. Olojo. E.A, Olurin. K.B, Mbaka.G and Oluwemimo. A.D[2005] Histopathology of the gill and liver tissues of the African catfish Clarias gariepinus exposed to lead, Afr. J. Biotech., 4: 117-122.
6. Kock,G, Triendl.M and Hofer.R[1996]seasonal patterns of metal accumulation in arctic char (Salvelinus alpinus) from an oligotropic alpine lake related to temperature. Can.J. fish.Aquat.Sci.53:780-786
7. Li. X, Liu.Y, Song. L, Liu. J[ 2003] Responses of antioxidant systems in the hepatocytes of common carp (Cyprinus carpio L.) to the toxicity of microcystin– LR. Toxicon,; 42: 85-89
8. Smith.S.R[2009] A critical review of the bioavailability and impacts of heavy metals in municipal solid waste composts compared to sewage sludge. Environ Int. Jan;35(1):142-56
. 9. Chen X, Shi J, Chen Y, Xu X, Xu S, Wang Y[2006] Tolerance and biosorption of copper and zinc by Pseudomonas putida CZ1 isolated from metal-polluted soil. Can J Microbiol. 52(4):308-316.
10. Espinosa-Urgel,M, Salido.A, Ramos.J[2000]Genetic Analysis of Functions Involved in Adhesion of Pseudomonas putida to Seeds. J Bacteriol. 182: 2363-2369.
11. Doenmez. G and Aksu. Z [2001] Bioaccumulation of copper(II) and Nickel (II) by the non adapted and adapted growing Candida sp. Wat. Res, 35: 1425- 1434.
12. Carhart. G and Hegeman.,G[1975] Improved method of selection for mutants of Pseudomonas putida, Appl Microbiol, 30:1046-1047.
13. Lowry. O.H, Rosebrough. N.J, Farr. A.L and Randall. R.J[1951] Protein measurement with the Folin‘s phenol reagent, J Biol Chem, 19: 265-275
14. Black. D and Malcolm Love R,[1988] Estimating the carbohydrate reserves in fish, J Fish Biol 32: 335–340
15. Reis. J.L[1961] Phosphatase activity in the ocular tissues, Brit. J. Opththal., 35: 149- 152
16. Blanchard. P, Ellis. M, Maltin. C, Falkous. G, Harris. J.B and Mantle. D[1993] Effect of growth promoters on pig muscle structural protein and proteolytic enzyme levels in vivo and in vitro, Biochimie,75: 839-847.
17. Oser. B.L[1965] Hawk's Physiological Chemistry, 14th Edn. McGraw Hill Book Company,NewYork, 1113.
18. Hood.J and Middleton.J.E[1970] False elevation of serum aspartate aminotransferase in patients with pulmonary tuberculosis during administration of sodium aminosalicylate, Tubercle 51 :313-315
19. Matsuzawa. T and Katunuma. N[1966] Colorimetric assays for serum alanine transaminase and lactic dehydrogenase using diazonium zinc salt , Anal Biochem 17: 143-153.
20. Yagi. K,[1984] Assay for blood, plasma or serum, Meth Enzymol, 105: 328.
21. Moran. M.S, Depierre J.W and Mannervik B[1979] Levels of glutathione, glutathione reductase and glutathione –S- transferase activities in rat lung and liver, Biochim Biophys Acta, 582: 67-78.
22. Balwant Singh, S. S, Sahota and Narang, A.S,[1961] Complexometric estimation of copper, J Anal Chem, 182: 241-244.
23. Taylor. D, Maddock. B.G and Manace.G[1985] The acute toxicity of nine ?grey-list? metals to two marine fish species, Limanda limanda and Chelon labrosus. Aquatic.Toxicol., 7:135-144.
24. Abou EL-Naga,E.H, EL-Moselhy, K.M and Hamed,M.A,[2005] Toxicity of cadmium and copper and their effect on some biochemical parameters of marine fish mugil seheli, Egyptian J Aqua Res. 31:2 ; 60-71.
25. Sansinanea.A.S, Cerone. S.I, Streitenberger.S.A, García, C and Auza,N[1997] Superoxide dismutase activity and reduced glutathione levels in cu-overload liver from sheep. Nutrition Res, 17 :1; 115-123.
26. Christensen.G.M[1971] Effects of metal cations and other chemicals upon the in vitro activity of two enzymes in plasma of the white sucker Catostomus commerson, Che. Biol Interact.4:351-359.
27. Halliwell,B and Gutteridge.J.M.C[1985] Lipid Peroxidation:A Radical Chain Reaction. Free Radicals in Biology and Medicine. Clarendon Press, Oxford. 139- 189.
28. Canovas.D, Cases.I And Víctor de Lorenzo[2003] Heavy metal tolerance and metal homeostasis in Pseudomonas putidaas revealed by complete genome analysis Environmental Microbiology. Environ Microbiol , 5(12), 1242–1256.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License