IJCRR - 3(11), November, 2011
Pages: 184-189
Print Article
Download XML Download PDF
STUDYING IN-VITRO APPLICABILITY OF HOSTINDEPENDENT STRAIN OF BDELLOVIBRIO
BACTERIOVORUS AS A BIOFILM REDUCING AGENT
Author: Jadhav Manjiri B., Nair Rohan N., Purkar Anjali B., Kininge Pallavi T., Kulkarni Chitrarekha G.
Category: Healthcare
Abstract:Biofilms are surface-attached microbial communities with phenotypic and biochemical properties distinct from free swimming planktonic cells. The capability of the gram-negative predatory bacterium Bdellovibrio bacteriovorus to control and reduce an existing Serratia marcescens biofilm was evaluated by colony biofilm assay. A reduction in biofilm biomass was observed as early as 2 h after exposure to the predator, and as much as 75% reduction after 6hrs of exposure. The ability of B. bacteriovorus to reduce an existing biofilm was confirmed by reduction in CFUs after exposure of predator.
Keywords: Biofilm, Biofilm reduction, Bdellovibrio bacteriovorus
Full Text:
INTRODUTION
Biofilms are surface-attached microbial communities with phenotypic and biochemical properties distinct from free swimming planktonic cells. Biofilm formation is thought to begin when bacteria sense environmental conditions that trigger the transition to life on a surface, followed by a multi step process leading to the formation of a mature biofilm [1] . Biofilms are implicated in a significant amount of human bacterial infections. Bacterial biofilms also cause fouling, product contamination, equipment failure, and decreased productivity due to downtime for system cleaning and replacement. Antibiotic doses which kill suspended cells, for example, need to be increased as much as 1,000 x to kill biofilm cells (and these amounts would kill the patient first!).
Disinfection rates for biofilm cells are also far below planktonic kills by antimicrobials. Biofilms are a concern in the food industry, in that they can arise from raw materials, surfaces, people, animals, and the air. Once food or a surface in a food processing plant is contaminated, the bacteria can form colonies, and eventually biofilms. Other microorganisms may attach to the initially adhered microorganism, and a biofilm could form. Cleansers used to wipe the counter will kill planktonic or single cells of bacteria, but they may not be able to penetrate biofilms. Foods that come into contact with the counter are then susceptible to contamination. Various techniques have been evaluated for their capability to manage and control biofilms, among them are the use of different materials and coatings to reduce initial cell adhesion to surfaces and a variety of treatments aimed at decreasing or destroying already existing biofilms, such as heat, cleaning regimens, low-power laser, sonication, chemical treatments, antibiotics, quorum-sensing analogs, and lectins. Recently, there has been a renewed interest in the use of biological control agents against biofilms. These agents include the use of invertebrates and protozoa to reduce biofilms by means of grazing and the use of bacteriophages. Bdellovibrio can be used potentially against the biofilm formation as it is predatory in nature. [2]
Bdellovibrio spp. is Gram negative, 0.20– 0.4 to 0.5–1.4 motile by means of single, polar, sheathed flagellum. [3] What characterises this bacterial genus as unique is its predatory behaviour. Bdellovibrios attack other Gram negative cells, penetrate their periplasm, multiply in their cytoplasm, and finally burst their cell envelopes to start once again. [4] Bdellovibrios are largely found in wet, aerobic environments and were first isolated from soil, where they are commonly encountered [5]. However, they can also be found in fresh and brackish water, sewage, water reservoirs, and seawater. Another environmental niche with which bdellovibrios have been associated are biofilms. It is believed that biofilms might offer good conditions for bdellovibrios‘ survival since these organisms have been found in natural marine biofilms but are not always recovered from the surrounding water. It is suggested that in a biofilm bdellovibrios can benefit from higher prey density, which has been shown to be necessary for Bdellovibrio survival [6] .








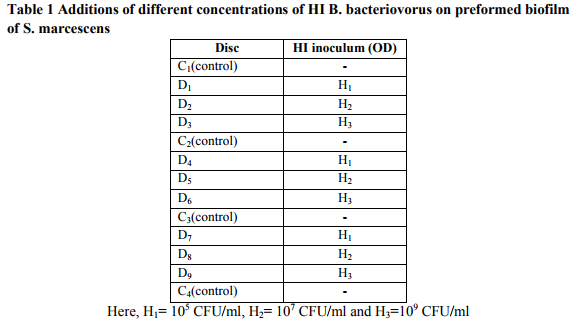
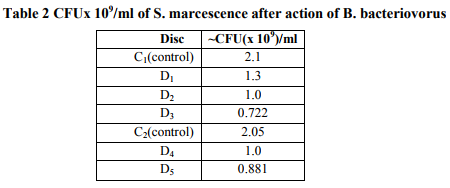

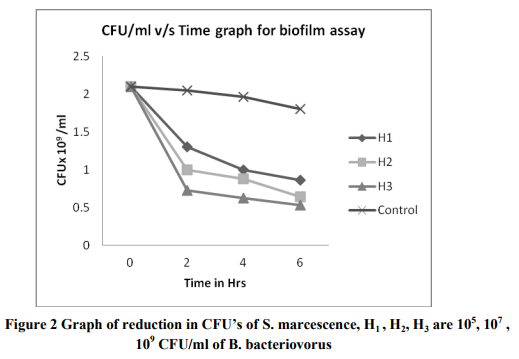
References:
1. ?Biofilm?, From Wikipedia, The Free Encyclopedia http://en.wikipedia.org/wiki/Biofilm
2. Kadouri, D. and O‘Toole, G. A. (July 2005), ?Susceptibility of Biofilms to Bdellovibrio bacteriovorus Attack?, Applied And Environmental Microbiology, Vol. 71, No. 7, pg. 4044–4051.
3. Brenner, D. J. , Krieg, N. R. , Staley, J. T. “Bergey’s manual of Systematic Bacteriology, Second Edition, The Proteobacteria, Volume Two, Part C, The Alpha-, Beta-, Delta- and Epsilon Proteobacteria? pg. 1041-1054.
4. Dworkin, M., Falkow, S. ?The Prokaryotes: Proteobacteria: delta and epsilon subclasses‘, pg. 15-23.
5. Stolp, H., and M. P. Starr. 1963. Bdellovibrio bacteriovorus Gen. Et Sp. N., a predatory, ectoparasitic, and bacteriolytic microorganism. Antonie Leeuwenhoek 29:217–248.
6. Bdellovibrio, From Wikipedia, The Free Encyclopedia http://en.wikipedia.org/wiki/Bdellovibr io
7. Merritt, J. H., Kadouri, D. E., O‘Toole, G. A. (2005), ?Growing and Analyzing Static Biofilms?, Current Protocols in Microbiology, pg. 1B.1.1- 1B.1.17.
8. J. Lindquist, (June 2005), ?More Dilution Plating?, John L's Bacteriology Pages, 188 International Journal of Current Research and Review www.ijcrr.com Vol. 03 issue 11 November 2011 http://www.jlindquist.net/generalmicro/ 102dil2
9. Williams, Neal H., and Silvia, P. (November 2006), ?Use of Bdellovibrionaceae as an antimicrobial agent?, US Patent Application 20060257374.
10. Iyer, J. (July 1, 2009), ?A Living Antibiotic, Anyone?, This Bacterium Might Just Be Man's New Best Friend?
11. “Serratia marcescens”, From Wikipedia, The Free Encyclopedia http://en.wikipedia.org/wiki/Serratia_m arcescens
12. Fry, J. C. and Staples, D. G. (Apr. 1976), ?Distribution of Bdellovibrio bacteriovorus in Sewage Works, River Water, and Sediments?, Applied And Environmental Microbiology, Vol. 31, No. 4, pg. 469-474.
13. Varon, M. and Shilo, M. (Mar. 1968), ?Interaction of Bdellovibrio bacteriovorus and Host Bacteria?, Journal of Bacteriology, Vol. 95, No. 3, pg. 744-753
14. Shilo, M. and Bruff, B. (1965), ?Lysis of Gram-Negative Bacteria by Host Independent Ectoparasitic Bdellovibrio bacteriovorus Isolates?, J. gen. Mimobiol. 40, pg. 317-328.
15. Widdel, F. (May 2007), ?Theory and Measurement of Bacterial Growth?, Version 3 04.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License