IJCRR - 3(11), November, 2011
Pages: 97-105
Print Article
Download XML Download PDF
SCREENING OXYLIPINS FOR INHIBITORY ACTIVITY AGAINST BACTERIAL PATHOGENS OF PAPAYA
Author: B. Sujatha, P. Uma Maheswari Devi
Category: General Sciences
Abstract:Oxylipins are metabolites produced by the oxidative transformation of Poly unsaturated fatty
acids. In the present study, the plant oxylipins are screened for their antibacterial activity
against Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa and xanthomonas
axonopodis, the bacterial plant pathogens interfering with papaya cultivation. In vitro growth
inhibition studies showed that 13-LOX metabolites are potential bactericidal agents compared
to 9-LOX metabolites.
Keywords: Lipoxygenase, Oxylipins, Linoleic acid, Linolenic acid, Antibacterial activity.
Full Text:
INTRODUCTION:
Oxylipins are secondary metabolites derived from polyunsaturated fatty acids (PUFAs) and are central players in a variety of physiological processes in animals and plants Reinbothe et al [1]. In mammals, oxylipins are derived mainly from Arachidonic acid (20:4) and have pivotal function in the inflammatory process De wild et al [2]. In contrast, Phytooxylipins are formed from the oxygenation of linoleic (18:2) and linolenic acid (18:3). Oxylipins are not preformed but are synthesized denovo in response to mechanical injury, herbivore or pathogen attack. The biosynthesis of most of plant oxylipins are initiated by the action of Lipoxygenases (LOX). LOX (EC1.13.11.12), a non heme iron containing dioxygenase that catalyzes the
addition of molecular oxygen at either the C9 or C13 position of the C18 fatty acids such as Linoleic (LA) and α-Linolenic acids (ALA) and leading to the formation of corresponding hydroperoxy octadecadienoic acid (13-HPOD/9-HPOD) and hydroperoxy octadecatrienoic acid (13- HPOT/9-HPOT) respectively Feussner et al [3] .
LOX generated 9- and 13-hydroperoxides have been shown to affect plant cell viability and regulate localized cell death during the hypersensitive response Rusterucci et al [4]. The products of the LOX reaction are then converted to different oxylipins in at least six biosynthetic branches Porta et al [5]. The 13-hedroperoxides of ALA serves as the substrates for hydroperoxide lyase leading to the formation of Cis-3-hexenol and Trans-2-hexenal Noordermee et al [6]. The formation of Jasmonic acid is initiated by an enzyme, hydroperoxide cyclase, which reacts with 13-hydroperoxide linolenic acid to give 12-oxophytodienoic acid the precursor of jasmonic acid Vick and Zimmerman et al [7] .
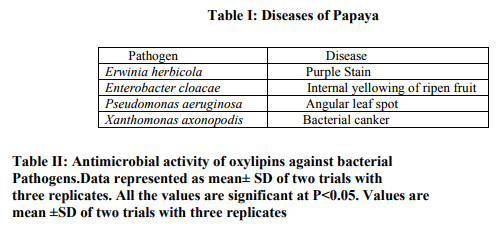
To get better insight into the biological activities of oxylipins, the present study is designed to asses the antimicrobial activities of oxylipins against papaya bacterial pathogens. Carica papaya has been infested with a number of bacterial diseases that cause serious damage to the papaya crop. Among them the most important bacterial pathogens are Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa and xanthomonas axonopodis. Erwinia sp cause chlorosis of the foliage with necrotic lesions, especially long midveins. This was followed by infection of the petioles and stem, and a rapid rot of the apical shoot, leading to the death of the tree. Petiole and stem lesions initially appeared as irregular, water soaked spots typical of those caused by leaf and stem blighting of bacteria. Another symptom called Purple stain is caused by Erwinia herbicola. Enterobacter cloacae causes internal yellowing of ripening fruit without displaying external symptoms. The disease is characterized by soft, yellow, disclosed flesh which diffuses spreading margins and an offensive, rotting odor in ripening papaya fruit. Some of the Pseudomonas aeruginosa species cause water soaked lesions and angular spots on Papaya leaves. Xanthomonas axonopodis causes tiny raised blister like lesions which later tan to brown and often surrounded by chlorotic halo.
MATERIALS AND METHODS
Materials: Cultures are obtained from Indian Institute of Microbial technology. HPLC solvents, n-hexane, Prop-2-ol, acetic acid, methanol were of high quality HPLC grade chemicals obtained from Merck, Sd-fine and SRL chemicals India. Cis-2-hexenol, Trans-2-hexenal, Jasmonic acid were obtained from Sigma chemical company, St.Louis, USA. Other chemicals used in this study were of high quality grade procured from Standard Indian Chemical companies.
METHODOLOGY
Preparation of hydroperoxides and hydroxides:
Partially purified papaya LOX was employed for the preparation of Hydroperoxides as per the method described by Reddanna et al [8]. Partially purified papaya LOX was incubated with 200µM linoleic or linolenic acid in 0.1M Tris HCl buffer pH 8.0 for 2 minutes at room temperature with constant shaking. The reaction was terminated by acidifying the reaction mixture to pH 2.5 with 6NHcl. The products were extracted with equal volumes of hexane:ether (1:1). The organic solvent was separated from the aqueous layer in a separating flask, was passed through the anhydrous granular sodium sulfate and subjected to evaporation in rotary evaporator to total dryness. The dried products were redissolved in methanol. The hydroperoxy compounds obtained in the reaction of papaya LOX with LA were reduced with sodium borohydrate and the products were extracted with equal volumes of hexane: ether (1:1). The organic solvent was separated from the aqueous layer in a separating flask, was passed through the anhydrous granular sodium sulfate and subjected to evaporation in rotary evaporator to total dryness. The dried product was re dissolved in methanol and was taken as 13-HOD (Hydroxy dienoic acid). Similarly the hydroperoxy compounds obtained in the reaction of papaya lipoxygenase with ALA were reduced with sodium borohydrate and the products were extracted with equal volumes of hexane: ether (1:1). The organic solvent was separated from the aqueous layer in a separating flask, was passed through the anhydrous granular sodium sulfate and subjected to evaporation in rotary evaporator to total dryness. The dried product was re dissolved in methanol grade and was taken as 13-HOT (Hydroxy trienoic acid)
Screening for antibacterial potential of oxylipins:
Antibacterial activity of oxylipins was determined by disc diffusion method as described by Langfeild et al [9]. One hundred microlitres of the diluted liquid cultures of Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa and Xanthomonas axonopodis were spread on sterile nutrient agar plates. The sterile filter paper discs (6mm diameter) impregnated with 6µg of different oxylipins were then placed on the surface of the medium and incubated at 370 c for 24hr. After the incubation period the diameter of inhibition zone around the oxylipin saturated discs were measured. Three replicates were maintained for each observation. 10% of ethanol was used as control for each observation.
Effect of Hydroperoxides and hydroxides on the growth of Bacteria:
To test the effect of oxylipins on the growth of Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa, Xanthomonas axonopodis, sterile discs were placed centrally on the surface of previously seeded agar plates. Various concentrations of Hydroperoxides and hydroxides (1.5µg, 3µg, 4.5µg and 6µg) dissolved in 10% ethanol were added to each filter disc and three replicates were maintained for each observation. Plates were incubated at 370 c and examined for zones of inhibition around each disc after 24hr. The lowest concentration of hydroperoxides and hydroxide that produced a detectable zone of inhibition was considered as a minimum inhibitory concentration (MIC). 10% of ethanol was used as control.
Determination of Minimum Bactericidal Concentration (MBC):
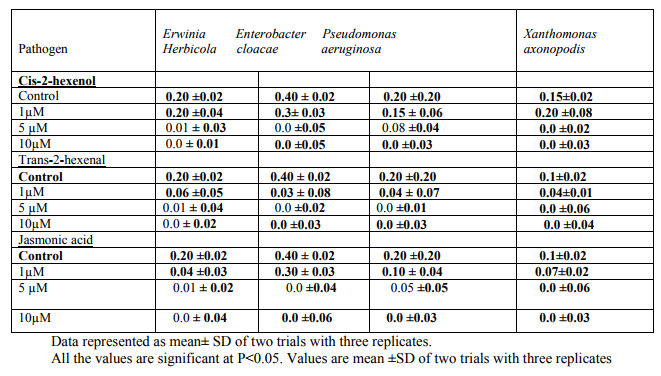
To determine the minimum Bactericidal concentrations, the graded levels of cis-3- hexenol, trans-2-hexenal and jasmonic acid were added to the sterile 150ml conical flasks containing of 50ml nutrient broth. The flasks were inoculated with an overnight culture of Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa, and Xanthomonas axonopodis and incubated at 370 c for 24hr.Growth was indicated by turbidity and measured at 660m in a colorimeter. Control was maintained by adding 10% ethanol. The experiment was repeated thrice. When growth was observed, the oxylipins under study was considered as bacteriostatic but when no growth appeared in the liquid culture the oxylipins was treated as bactericidal.
RESULT AND DISCUSSION
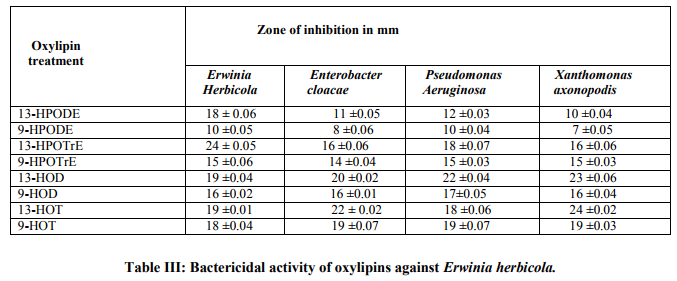
The oxylipins generated from LA and ALA were screened for their antibacterial activity against pathogens of Papaya (Table I). Results obtained in the present study revealed that the oxylipins possess potential antibacterial activity against Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa and Xanthomonas axonopodis. When tested with disc diffusion method, 13-LOX metabolites showed significant antibacterial activity on comparison with 9- LOX metabolites (Table II). Lox metabolites have been shown to play a role in plant response to biotic stress both as antimicrobial compounds Prost et al [10] and signal molecules that led to the activation specific defense genes Roshal et al [11]. All the bacterial pathogens of Papaya were found to be sensitive to the tested oxylipins.
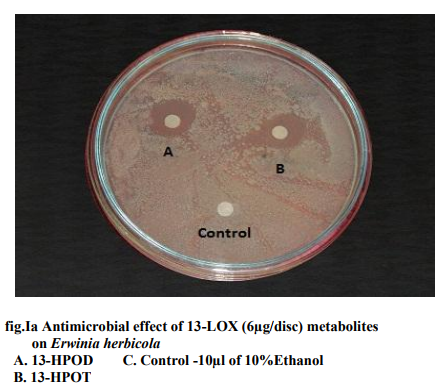
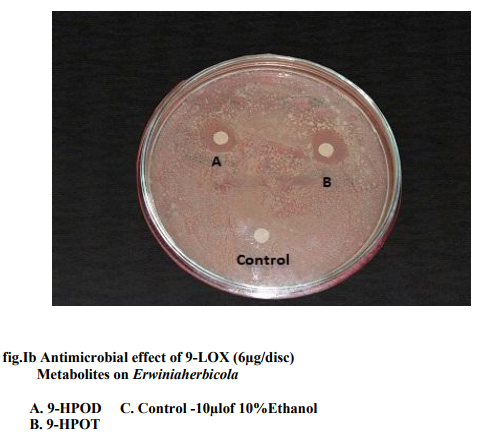
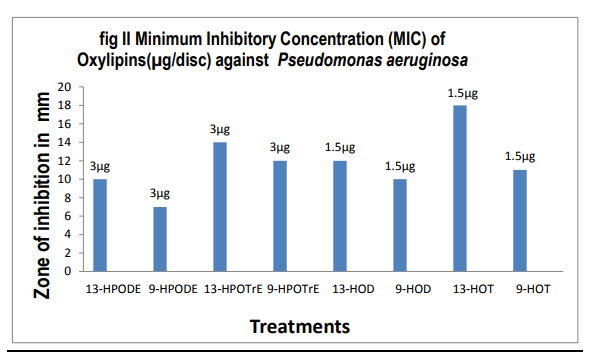
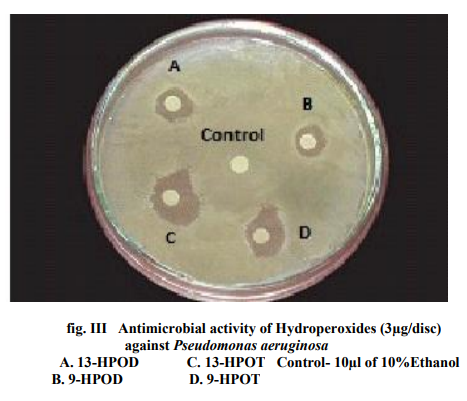
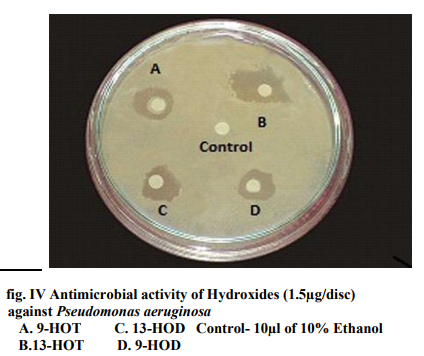
Of all the compounds tested maximum inhibition of bacterial growth was observed with hydroxides compared to hydroperoxides. ALA hydroperoxides (13- HPOT & 9-HPOT) are potent bactericidal agents (fig.I) compared to LA hydroperoxides (13-HPOD & 9-HPOD). In Control filter discs however, there was no inhibition of the bacterial growth. The antibacterial activity of LOX metabolites was enhanced by increase in the concentration of oxylipins. The concentration of both 13- and 9- LOX products required to inhibit the invitro bacterial growth was calculated and indicated in figure II. The MIC of hydroperoxides of LA and ALA were found to be 3µg/disc (fig.III) and 1.5µg/disc for hydroxides (fig.IV). The growth inhibiting activity of Cis-3- Hexenol, Trans-2-Hexenal and Jasmonic acid was measured on Erwinia herbicola, Enterobacter cloacae, Pseudomonas aeruginosa and Xanthomonas axonopodis in liquid medium. Trans-2-Hexenal, the high antibacterial oxylipin prevented the growth of all bacteria at low concentration. A direct relationship between amount of oxylipins (Cis- 3-Hexenol, Trans-2- Hexenal and Jasmonic acid) and retardation of bacterial growth was observed in liquid culture as shown in Table III. In vitro growth effects were measured at 660nm and compared to controls, grown in the presence of 10% ethanol (fig.V). Cis-2- hexenol and Trans-2-hexenal were able to strongly reduce growth of Pseudomonas invitro Croft et al [12]. Contribution of the oxylipin pathway to plant defense can proceed from production of signal molecule inducing defense gene expression or hypersensitive response Lacemera et al [13] .
CONCLUSION
The present study clearly emphasizes the role played by Lipoxygenase in plant defense mechanism. The plant oxylipin metabolome constitutes a large number of structurally diverse compounds formed by the oxygenation of fatty acids. The Present study demonstrates that the oxylipins are potential antimicrobial agents which help in eliminating different bacterial pathogens that cause serious damage to Carica papaya. The 13-LOX metabolites exerted pronounced antimicrobial activity, on comparison with 9-LOX metabolites, demonstrating the predominance of 13- LOX pathway in Carica papaya belongs to the family Caricaceae. Among all oxylipins tested, Trans-2-hexenal is the most efficient antimicrobial agent. In general it can be concluded that the screening provides a better insight in to the antibacterial activity of oxylipins and the role of oxylipins in combating the bacterial pathogens of Papaya.
ACKNOWLEDGEMENTS
The authors are grateful to the University Grants Commission, New Delhi, India for financial support to carry out the Major Research Project (F.No.34-241/2008 (SR-).

References:
1. Reinbothe S, Mollenhauer B, Reinbothe C. JIPs and RIPs: the regulation of plant gene expression by jasmonates in response to environmental cues and pathogens. The Plant Cell 6, 1994; 1197-1209 101 International Journal of Current Research and Review www.ijcrr.com Vol. 03 issue 11 November 2011
2. De Wild HPJ, Otma EC, Peppelenbos HW. Carbon dioxide actionon ethylene biosynthesis of pre climacteric and climacteric pear fruit. Journal of Experimental Botany, 2003; 54: 1537– 1544.
3. Feussner I, Wasternack C. The lipoxygenase pathway. Annu. Rev. Plant Biol, 2002; 53: 275-297.
4. Rustérucci C, Montillet J.L, Angel J.P, Battesti C, Alonso B, Knoll A, Bessoule J.J, Etienne P, Suty L, Blein J.P.. Involvement of lipoxygenasedependent production of fatty acid hydroperoxides in the development of the hypersensitive cell death induced by cryptogenic of tobacco leaves. J. Biol. Chem., 1999; 274: 36446-36455.
5. Porta, H. and Rocha Sosa M. Plant Lipoxygenase, Physiological and Molecular Features, Plant Physiol. 2002, vol. 130, pp. 15–21.
6. Noordermeer MA, Veldink GA, Vilegenthart JFG. Fatty acid hydroperoxide lyase: a plant cytochrome P450 enzyme involved in wound healing and pest resistance. Chembiochemistry, 2001; 2, 495-504.
7. Vick B A and Zimmerman DC. Oxidative systems for modification of fatty acids: the lipoxygenase pathway. In the biochemistry of plants: a Comprehensive treatise. Edited bt stumpf, P.K.Vol9 PP: 53-90. Academic Press, Orlando FL.
8. Reddanna P, Whelan J, Maddipat K.R and Reddy C. C. Purification of arachidinate 5- lipoxygenase from potato tubers. Methods Enzymol, 1990; 187: 268-277.
9. Langfield R.D, Scarno F.J, Heitzman M.E, Kondo M, Hammond G.B, and Neto C.C. Use of modified microtitre plate bioassay method to investigate antibacterial activity in the Peruvian medicinal plant Pereromia galioides. J. Ethanopharmacol, 2004; 94:279-281
10. Prost I, Dhont S, Rothe G, Vicente J, Rodriguez MJ, Kift N, Carbonne F, Griffiths G, Esquerre-Tugaye MT, Rosahl S, et al. Evaluation of the antimicrobial activities of plant oxylipins supports their involvement in defense against pathogens. Plant Physiol, 2005; 139:1902-1913.
11. Rosahl S. Lipoxygenases in plants-their role in development and stress response. Verlag der zeitschrift fur Naturforschung, 1996; 51:123-138
12. Croft K.P.C, Juttner F, Slusarenko A. J. Volatile products of the Lipoxygenase pathway from Phaseolus vulgaris (L.) leaves inoculated with Pseudomonas syringae pv phaseolicola. Plant physiology, 1993; 101: 1324.
13. Lacemera S, Balague C, Goble C, Geoffroy P, Legrand M, Feussner I, Roby D and Heitz T. The Arabidopsis Patatin-Like Protein 2 (PLP2) plays an essential role in cell death execution and differentially affects biosynthesis of oxylipins and resistance to pathogens. The American Phytopathological Society, 2009; MPM Vol.22, No.4, Pp.469- 481. doi: 101094/MPM1-22- 4-0469.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License