IJCRR - 4(1), January, 2012
Pages: 51-55
Print Article
Download XML Download PDF
POLYANILINE COATED EXPANDED GRAPHITE ACTS AS A NEW ELECTRODE MATERIAL FOR ETHANOL
FUEL CELL
Author: Abhik Chatterjee, I Basumallick
Category: General Sciences
Abstract:An expanded graphite electrode coated with a thin layer of polyaniline and loaded with platinum has been used to study electro-oxidation of ethanol. The graphite plate electrode was expanded by doping with potassium (K)-vapour using vapour incorporation technique developed at our laboratory. The expanded graphite was platinised by electro-deposition
technique and its electrochemical behaviour was examined using a laboratory model fuel cell. The open-circuit potential (OCP) and shortcircuit current (SCC) values of laboratory model fuel cell in 1M H2SO4 comprising of normal and expanded graphite electrodes having same catalytic loading as working electrode were observed. The expanded graphite electrode was also coated with a thin film of polyaniline and then loaded with Pt catalyst as before. Cyclic voltammogram of polyaniline coated electrodes in H2SO4 solution was investigated. Electrocatalytic activity of the modified electrode is better than Pt deposited electrode due to the higher reaction area of Pt particles.
Keywords: Polyaniline, ethanol, electrocatalyst, expanded graphite
Full Text:
INTRODUCTION
Metal microparticles dispersed in polymer modified electrodes have been widely used as electro-catalyst. There are various polymeric films, such as polypyrrole (PPY), polyaniline (PANI), poly(3- methylethiophene) (PMT), poly(3,4- ethylenedioxythiophene) (PEDOT), and so forth, have been investigated as conducting catalyst supports 1,2 . These polymers are usually used as matrix to incorporate noble metal catalysts in the application for electro-oxidation of small molecules such as hydrogen, methanol and formic acid, etc 3,4,5 . Among the various conducting polymers, polyaniline (PANI) is one of them most interesting material because of its moderately high conductivity, well behave electrochemistry, easy preparation and possible applications as electro-catalyst towards various electro-oxidation reactions. A thin film of a conducting polymer (CP) improves the interfacial properties between the electrode and the electrolyte. Conducting polymer can allow a facile flow of electronic charge during the electrochemical oxidation of alcohol on Pt. Generally, an electrochemically deposited conducting polymer develops three dimensionally on a substrate 6 . Therefore, it introduces a high porosity and roughness; as a result it generates a large surface area for electrochemical reactions. The major problems of fuel cells are poisoning of the electro-catalyst even with Pt as the catalyst and its crossover from anode to cathode compartment. Besides elelectro-catalyst, the carbon material used as catalytic support also plays an important role in dictating fuel cell efficiency. In the present research an attempt has been made to improve catalytic activity of ethanol (EtOH) fuel cell using polyaniline coated expanded graphite as catalytic support material.
MATERIAL AND METHODS
H2SO4 (Merck), H2PtCl6,6H2O (Arora Matthey Limited) were used as supplied. Aniline (Merck) and EtOH (Bengal chemicals) were distilled before use. The distillate was collected rejecting head and tail fractions. The anode was a 1cm2 graphite plate of thickness 3.8 mm obtained from the R&D of BHEL, India. Graphite plates were expanded using a simple technique developed at our laboratory. The experimental arrangement for expansion is shown in figure 1 7 . The graphite plate was expanded to an extent of 20% by potassium vapour. Pt particles were deposited on the working electrode (2 cm2 ) under galvanostatic condition using +10 mA.cm-2 current for 30 minutes from chloroplatinic acid solution (0.01M) in 0.5M H2SO4. The air cathode was fabricated by loading a 1cm2 graphite plate with Pt. For the sake of comparison a Pt loaded anode without expansion was used. All experiments were carried out at 30oC. Polyaniline was prepared galvanostatically. Electrolyte solution was 0.5 M aniline in 1M H2SO4 solution. The solution was taken in a one compartment cell in which supporting material was used as working electrode and counter and reference electrode merged to Pt wire. -1 mA.cm-2 current under galvanostatic condition was passed for 100seconds to deposit a film of PANI. After polymerization the polymer coated electrodes were washed repeatedly with distilled water and used for electrochemical studies. Cyclic voltammogram of PANI coated electrodes in H2SO4 solution was investigated using a potentiostat-galvanostat (PAR Versastat TM II). Laboratory model fuel cell Laboratory model fuel cells were housed in two glass rectangular chambers (50ml capacity) connected by an inverted Ushaped bridge. Cathode and anode, as fabricated above, were inserted into the respective chambers through the openings of the lid. H2SO4(1M) were poured into cathode and anode chambers, respectively. Alcohol was added to the anode chamber to obtain the desired concentration. The U tube was plugged with foam, which had been soaked in the acid to avoid crossover of alcohol. Air was bubbled slowly through cathode chamber using an air pump.
RESULTS
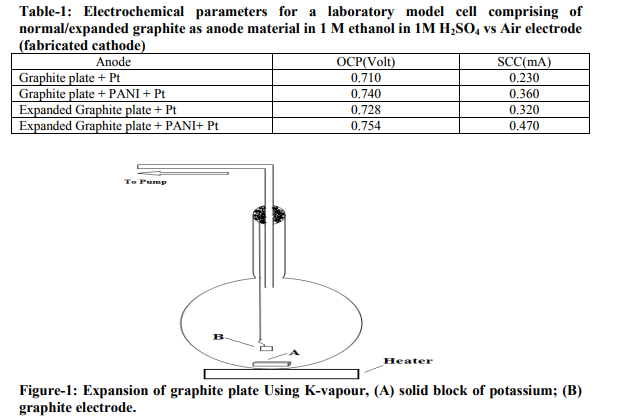
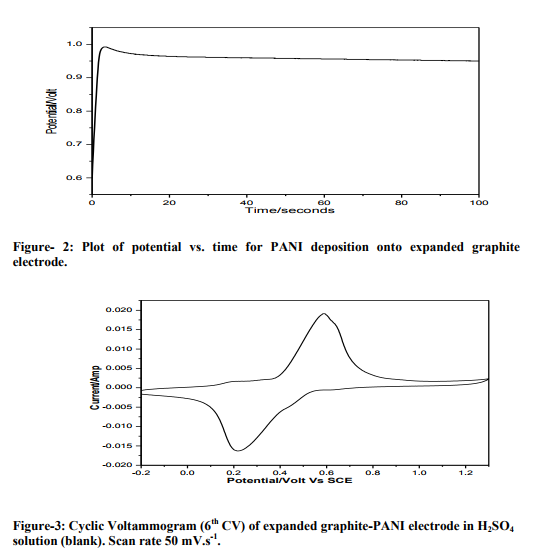
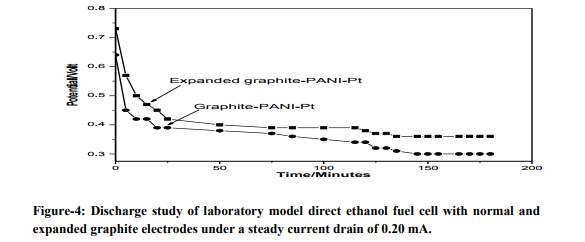
Figure 2 shows the galvanostatic polymerization curve of aniline on support material surface from a deposition bath of 0.5M aniline in 1M H2SO4. From the figure, it is clear that the polymerization starts at about potential of 0.95 volts and continues in the potential range of 0.9 to 1.0 V for a period of 100 seconds. Figure 3 shows the cyclic voltammogram of expanded graphite-PANI electrode in H2SO4 solution (blank) at a scan rate of 50 mV.s-1 . It is found that within the potential limits – 0.2 to 1.3 V vs. SCE, the response changed on cycling 8 . After about the five cycles, the PANI film shows a steady response and then it was used as an anode material for deposition of Pt. The open-circuit potential (OCP) and shortcircuit current (SCC) values of laboratory model fuel cell in 1M H2SO4 comprising of normal and expanded graphite electrodes having same catalytic loading as working electrode are shown in table1. Steady open-circuit potentials (OCP) were reached within 30 minutes. The potential remained undisturbed for an appreciable duration which indicates that these electrodes are stable in acidic medium. The shortcircuit current (SCC) values were measured after attainment of a steady OCP value. These data demonstrates the role of support material on the activity of catalyst. To compare the performances of laboratory model fuel cells with different anodes, cells were discharged under 0.20 mA current for 180 minutes and profiles are presented in figure 4. The observed discharge profiles indicate that the cell with expanded graphite electrode exhibits better performances.
DISCUSSION
The CV of PANI coated electrode shows two peaks (figure 3). In the anodic scan, the peaks at about 0.57 V is due to SO4 2- anion up taking. In the reverse scan, the peaks at about 0.23 V is due to SO4 2- anion expulsion 8 . Generally, graphite electrodes are utilised for providing support only, no electrocatalytic activity has been observed for alcohol fuel cell. Pt deposited on support material is known as an efficient electrocatalyst for alcohol oxidation. But the catalytic activity of the electrode can be enhanced if the support material is modified. It is seen from the table that OCP value is higher for Pt loaded expanded graphite material electrode. This indicates better catalytic effect of expanded graphite.
Electro-oxidation of EtOH takes place via an initial absorption onto the anode surface followed by deprotonation. For expanded graphite the nanographite channels act as better absorption sites 7 and the electrocatalytic activity of Pt within these channels is also enhanced as is reflectd in their SCC values (table1). Basically, potassium metal as dopant changes the interlayer distance between graphite layers. Table1 also indicates the relative improvement of catalytic effect of normal and expanded graphite when loaded with Polyaniline and Pt. It is very interesting that when Pt particles were deposited onto PANI coated electrode, catalytic effect is further improved. This is due to high dispersion of Pt particles. As a result the specific reaction area of these electrodes is increased and thus improves catalytic efficiency. The observed discharge profiles (figure 4) indicate that the cell with expanded graphite electrode exhibits better performance. The reason is the higher tolerance of the platinum particles to poisoning effect, in comparison with the serious problem of poisoning effect on bulk platinum electrodes. PANI may adsorb some of the poisonous intermediates and thus the adsorption prevents the dispersed Pt particles from becoming deactivated 9 . Thus this research introduces the use of polyaniline modified expanded graphite as an efficient electrode material in fuel cell.
ACKNOWLEDGEMENTAuthors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors /publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Patra S, Munichandraiah N. Electrooxidation of methanol on Ptmodified conductive polymer PEDOT. Langmuir 2009; 25 (3): 1732–1738.
2. Swathirajan S, Mikhali YM. Methanol oxidation on Platinum-Tin catalysts dispersed on Poly(3-methyl) thiophene conducting polymer. J Electrochem Soc 1992; 139( 8) :2105-2110.
3. Kitani A, Akashi T, Sugimoto K, Ito S. Electrocatalytic oxidation of methanol on platinum modified polyaniline electrodes. Synth Met 2001; 121(1- 3):1301-1302.
4. Liu F, Huang L, Wen T, Gopalan A. Large-area network of polyaniline nanowires supported platinum nanocatalysts for methanol oxidation. Synth Met 2007; 157(16-17):651-658.
5. Kost KM, Bartak DE, Kazee B, Kuwana T. Electrodeposition of platinum microparticles into polyaniline films with electrocatalytic applications. Anal Chem 1988; 60(21): 2379-2384.
6. Patra S, Barai K, Munichandraiah N. Scanning electron microscopy studies of PEDOT prepared by various electrochemical routes. Synth Met 2008; 158(10): 430–435.
7. Bhattacharya A, Hazra A, Chatterjee S, Sen P, Laha S, Basumallick I. Expanded graphite as an electrode material for an alcohol fuel cell. J Power sources 2004; 136: 208-210.
8. Hatchett DW, Josowicz M , Janata J. Acid Doping of Polyaniline: Spectroscopic and Electrochemical Studies. J Phys Chem B 1999; 103(50): 10992-10998.
9. Yano J, Shiraga T, Kitani A. Dispersed platinum and tin polyaniline film electrodes for the anodes of the direct methanol fuel cell. J Solid State Electrochem 2008; 12:1179-1182.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License