IJCRR - 4(8), April, 2012
Pages: 155-167
Date of Publication: 25-Apr-2012
Print Article
Download XML Download PDF
OIL-WATER SEPARATION USING FLY ASH ZEOLITE TREATMENT
Author: Chintan Pathak, V. K. Srivastava
Category: Technology
Abstract:Introduction: A large amount of water is used in the Upstream, Downstream, Petroleum and Automobile industrial processes and a huge fraction of it comes out as waste after getting polluted by oil and other toxic substances. Liquid wastes from the Petroleum Industries are relatively less toxic in nature and can be easily treated by conventional processes. However, solid wastes, especially oily Waste still remains as major environmental hazards, demanding safer disposal practices. Methodology: Oil contaminated wastewater is an extremely complex and variable waste of organic compounds ranging up to high molecular weight tars and bitumen's. Oily waste disposal is a major threat to the environment since they ultimately deplete the natural capital and degrade the prisnity of the eco system. This is the challenging area for petroleum scientist to establish the method by means of which maximum proportion of oil from
the waste can be recovered. Experimental Set-up: There are different Chemical and Biological Treatment methods available. The Need is to optimize different treatment technologies in a cost effective method. The purpose of this work is to increase the oil-water separation by using zeolite treatment along heat treatment. Result and Conclusion: Results show the oil-water phase separation with zeolite treatment, as compared to fly ash. Heat treatment may be effectively used to treat oily waste as it gives more separation in shorter duration of time without creating any environmental as well as human health hazard. Increase in temperature and duration of heating gives more separation and more reduction in
phase separation.
Keywords: Oil, Oily Waste, Oil Spill, Phase Separation, Environmental Pollution control
Full Text:
INTRODUCTION
The increasing requirements in the sphere of environmental protection have induced search for more effective, inexpensive and ecologically safe solutions. Given that, the zeolite is aluminosilicate members of the family - microporous solids known as ?molecular sieves? and have sorptive and ion-exchange properties; the most important pollution of hydrocarbon (HC) (viz. petrochemical spills), may find applications in the removal of oil spill, using hydrophobic – oleophilic - high-silica content - hydro thermally stable – zeolite; particularly those, prepared cheaply from fly ash; while, simultaneously providing a solution to other environmental problems. The research will be applicable in wide variety of environmental pollution control applications. Since cleanup after an oil spill is so ineffective and so difficult, and does not always fully rehabilitate affected areas. As a challenging task, the author has experimentally investigated the strategy for synthesis and application of flyash zeolite for oil spill cleanup. When we think of oil spills, we habitually think of oil tankers spilling their cargo in oceans or seas. However, oil spilled on land often reaches lakes, rivers, and wetlands, where it can also cause damage. Oceans and other saltwater bodies are referred to as marine environments. Lakes, rivers, and other inland bodies of water are called freshwater environments. The term aquatic refers to both marine and freshwater environments. When oil is spilled into an aquatic environment, it can harm organisms that live on or around the water surface and those that live under water. Spilled oil can also damage parts of the food chain, including human food resources. The severity of the impact of an oil spill depends on a variety of factors, including characteristics of the oil itself. Natural conditions, such as water temperature and weather, also influence the behavior of oil in aquatic environments. Various types of habitats have differing sensitivities to oil spills as well. The most interesting results among those described in literature [1-5] on the development and application of the materials for adsorption purification of petroleum and oil products from water are considered and generalized [6].The purpose of this work is to provide a logical overview of fly ash synthesized zeolite for oil spill cleanup. Oily waste disposal is a major threat to the environment since they ultimately deplete the natural capital and degrade the prisnity of the eco system.
- Presence of oily waste in water bodies affects the potability of water and also its use for agricultural as well as recreational purposes. Even traces of oily waste can form a thin film on the surface of water body and render it unfit aesthetically. This can also prevent the natural oxygenation of water bodies and in turn affect the ecosystem associated with it.
- Layer of oil on aquatic living bodies affects the metabolic activities.
- Oily waste provides a hostile environment to the soil bacteria and prevents the natural nutrient transformation cycle in plants and microorganisms.
- Oily waste, when exposed to open atmosphere will disintegrate due to UV rays and will release volatile organic compounds into atmosphere which are carcinogenic compounds in nature. These obnoxious gases create odour nuisance to the nearby habitat.
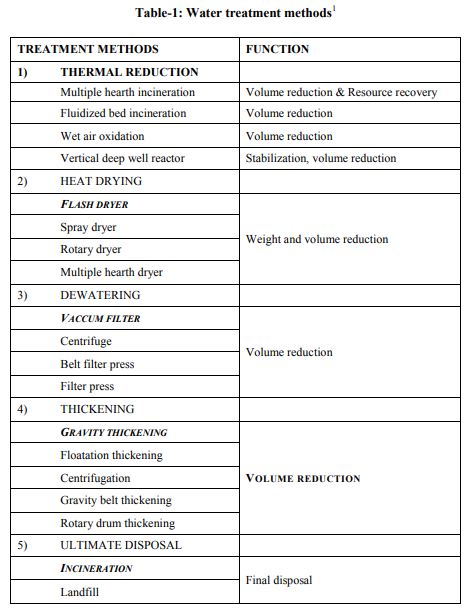
There are some treatments given to waste oily water, shown in Table-1. Sorbents are materials that soak up liquids. They can be used to recover oil through the mechanisms of absorption, adsorption, or both. Absorbents allow oil to penetrate into pore spaces in the material they are made of, while adsorbents attract oil to their surfaces but do not allow it to penetrate into the material. To be useful in combating oil spills, sorbent need to be both oleophilic and hydrophobic (water-repellant). Although they may be used as the sole cleanup method in small spills, sorbent are most often used to remove final traces of oil, or in areas that cannot be reached by skimmers. Once sorbent have been used to recover oil, they must be removed from the water and properly disposed of on land or cleaned for re-use. Any oil that is removed from sorbent materials must also be properly disposed of or recycled. Sorbents can be divided into three basic categories: natural organic, natural inorganic, and synthetic. Natural organic sorbent include peat moss, straw, hay, sawdust, ground corncobs, feathers, and other carbon-based products. They are relatively inexpensive and usually readily available. Organic sorbent can soak up from 3 to 15 times their weight in oil, but they do present some disadvantages [6]. Some organic sorbent tend to soak up water as well as oil, causing them to sink. Many organic sorbent are loose particles, such as sawdust, and are difficult to collect after they are spread on the water. Adding flotation devices, such as empty drums attached to sorbent bales of hay, can help to overcome the sinking problem, and wrapping loose particles in mesh will aid in collection. Natural inorganic sorbent include clay, vermiculite, glass, wool, sand, and volcanic ash. They can absorb from 4 to 20 times their weight in oil [7]. Inorganic substances, like organic substances, are inexpensive and readily available in large quantities. Synthetic sorbent include man-made materials that are similar to plastics, such as polyurethane, polyethylene, and nylon fibers. Most synthetic sorbent can absorb as much as 70 times their weight in oil, and some types can be cleaned and reused several times [8]. Synthetic sorbent that cannot be cleaned after they are used can present difficulties because they must be stored temporarily until they can be disposed of properly. Adsorbent materials are attractive for some applications because of the possibility of collection and complete removal of the oil from the oilspill site. The further advantage is, it can be, in some case, be recycled. Some important propertise of good adsorbent includes, hydrophobicity and oleophilicity, high updatake capacity, high rate of uptake, retention over time, oil recovery from it and reusability. It is interesting to mention that high-quality zeolite with high water- and oiladsorpotion and CEC may be readily produced from inexpensive flyash (a by-product of thermal power stations) and other solid waste materials containing silica and alumina [9-14], thus providing a solution to other environmental problems in addition to application in the removal of oil spills. Zeolite, which are synthesized, show some of these above mentioned properties. It has been suggested [10] that zeolite can be useful for oil spill cleanup due to their oleophilic and hydrophobic characteristics. In addition, there are 3 properties of zeolite that make them technologically important: they are selective and strong adsorbents, they are selective ion exchangers and they are catalytically active [7]. Researches are increasingly focusing on hydrophobic pure-silica (or high silica) zeolite as alternative sorbents for activated charcoal for sorpotion of organic pollutants (such as volatile organic compounds) [14].
Hydrophobic zeolite have a small percentage of aluminum atoms in their crystal structure thereby shifting their adsorption affinity away from polar molecules, like water, towards non-polar substances, like organic solvents (i.e. they are highly organophillic). These zeolite are thermally and hydrothermally stable (upto about 1300 oC) and like other aluminosilicates, they have a unit structure with a defined pore size of 0.2-0.9 nm, resulting in a high specific surface ares. Hydrophobic zeolite also have the advantages like, little need for safety with regards to fire risk (since zeolite are inflammable), co-adsorption with water possible only when the relative humidity is higher than 70 %, can be regenerated with steam [15-16] or by calcination at high temperature [17-18]. Thus the possibility of application of flyash synthesised zeolite with desired shapes and sizes of are of great technological value in the oil spill clean-up.
MATERIAL AND METHODS
In order to insure the maximum digestion of silica from fly ash, it has been opined by the previous researchers that the microwave can be employed for complete heating of fly ash samples as compared to conventional hydrothermal method. Based on this, the main objectives (viz., less activation and digestion time, maximum silica digestion, more nucleation and better crystal growth of zeolite) of the present research has been planned. Fly ash samples from Bhusaval Thermal Power Station (BTPS), Bhusaval, Maharashtra, India; were collected for the present study. The ash sampling was done from hoppers and representative samples were prepared for three different fields or hoppers. The 40% pure hydrofluoric acid (HF) from Merck Ltd. was used for digestion of fly ash by employing microwave. The most general physical properties of zeolite are its bulk density and specific gravity (i.e., somewhere in between 2 to 2.4) which can correlate with its porosity (i.e., the measure of the pore volume in zeolite) and its most dependent parameter i.e., cation exchange capacity. The type of zeolite formed is a function of the temperature, pressure, concentration of the reagent solutions, pH, process of activation and ageing period, SiO2 and Al2O3 contents of the raw materials.
EXPERIMENTAL SET UP
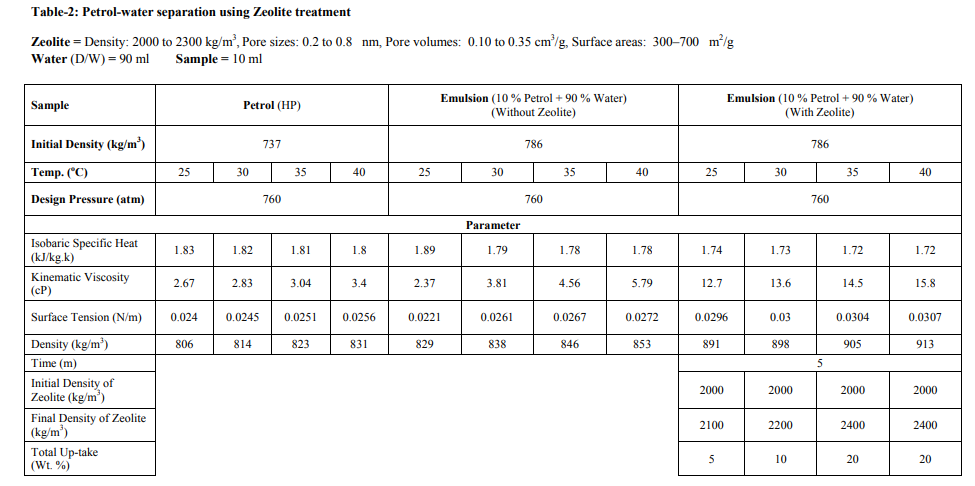
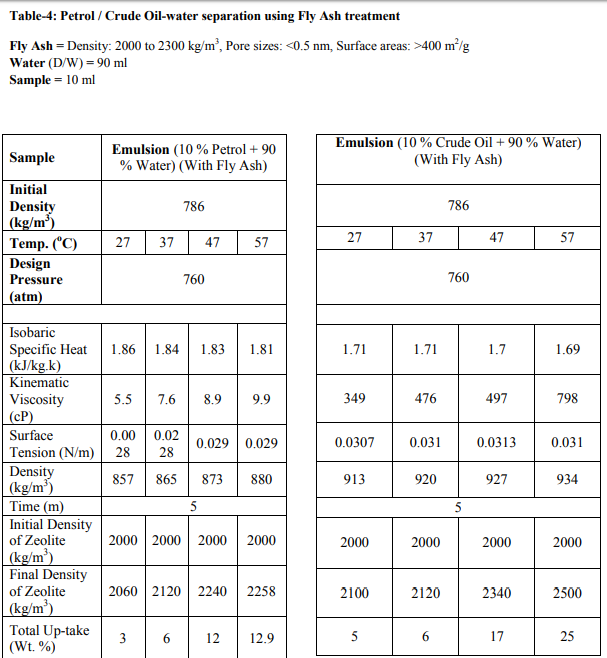
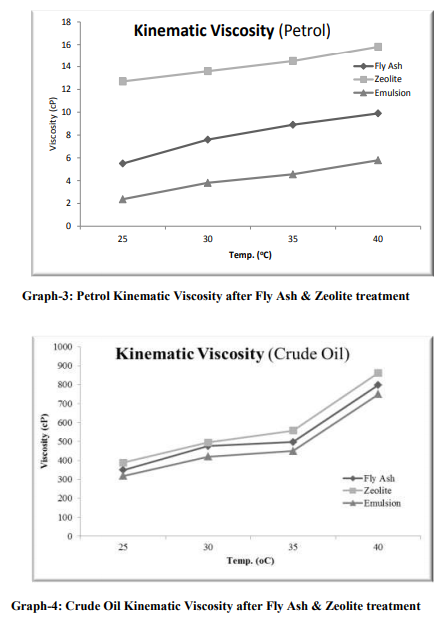
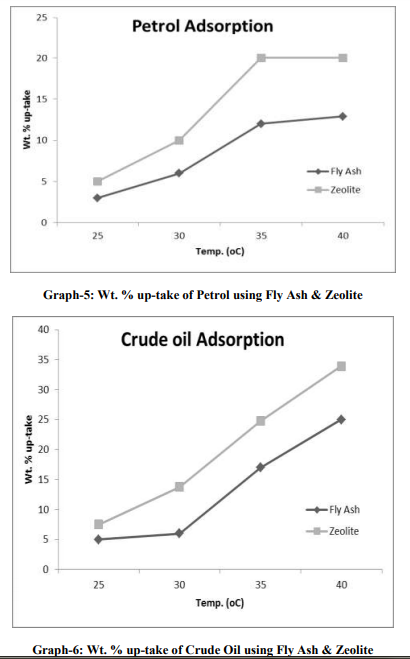
The samples were collected from the 2 major sources: [1] Petrol from Petrol Pump (HP) and [2] Crude Oil from Exploration site. The density which we have used for sample-1, is 737 kg/m3 and for sample-2, is 825 kg/m3 . Fly Ash samples were collected from Thermal Power Station, Bhusaval having Density 2-2.3 gm/cc. The Pore size and surface area of collected fly ash was well characterized, which is 400 m2 /g, respectively. Fly Ash was then treated with 40 % pure Hydrofluoric Acid (HF) along with Microwave Irradiation. The range of temperature was from 45-110 oC. The time range was 5-15 min. The physical characteristics of these synthesized fly ash zeolites are as follows: Density: 0.2 to 2.3 gm/cc Pore sizes: 0.2 to 0.8 nm Pore volumes: 0.10 to 0.35 cm3 /g Surface areas: 300–700 m2 /g Oil-Water Emulsion was prepared by addition 90 ml of D/W water into 10 ml of Petrol and Crude Oil samples. It was then kept for 24 hrs. for formulation of micro-emulsion. The microemulsion which was formed between oil and water phase, is separated with the help of Seperatory funnel. After obtaining the micro-emulsion, 5 gm of fly ash was added and treated with the heat treatment at different temperature ranges from 25- 45 oC (Fig.- 1,2,3,4; Table-4) for Petrol and Crude Oil, simultaneously. Similarly, 5 gm of Fly Ash Synthesized Zeolite were also added into the micro-emulsion and heat treatment was given at different ranges from 25-45 oC (Fig.-1,2,3,4; Table-2 and 3) for Petrol and Crude Oil. Following parameters were set-up for taking getting result of Oil-Water Phase Separation using Microwave Assisted Zeolite Treatment: Isobaric Specific Heat (kJ/kg.k) Kinematic Viscosity (cP) Surface Tension (N/m) Density (kg/m3 ) Separated study of oil and water were also taken on volume basis and weight basis, after every 5 min.
RESULT
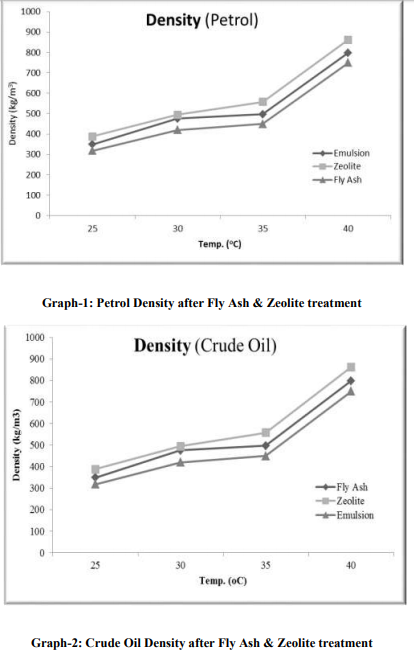
Separation study shows that the fly ash and zeolite, being the porous material, adsorbing the oil. It is resulted into the increasing density, from which, it can be determined that an Emulsion after zeolite treatment gives increasing in the viscosity and decreasing surface tension. From this it can be predicted that as the density increases more towards water density, the oil-water separation may take place. From this study of oil-water separation with the help of fly ash and zeolite, it has been observed that the separation rate between oil and water increases with increases in temperature. After the treatment of flyash with microwave irradiation into the HF acid, the adsorption capacity increases because of increase in pore size. At 25 oC, the emulsion density is around 829 kg/m3 , after zeolite separation the density is 891 kg/m3 and with flyash separation the density is 857 kg/m3 . The kinematic viscosity, at 25 oC, of emulsion is 2.37 cP, with zeolite separation is 12.7 cP and with flyash separation, 5.5 cP. From the graph-3,4,5,6; it is very clear that almost all the parameters (viz.,Kinamatic viscosity and Density), shows the oil-water separation is more favorable for zeolite as compared to flyash.
From the graph-1 and 2, it can be seen that the wt. % up-take of Petrol and Crude Oil, is more using zeolite compare to flyash. As temperature increases, it can also be concluded that the Total Wt. % up-take increases.
CONCLUSION
The microwave-assisted digestion method for fly ash provided comparable or better results to that of conventional hydrothermal digestion method. The variations in weight of sample, temperature, and time resulted in improved recoveries of several elements, but generally. In addition, preparation and time needed for digestion was significantly reduced with this method. The microwave-assisted method for fly ash did work better than hydrothermal digestion method. There is, however, scope for more improvement in future research. The use of hydrofluoric acid was absolutely necessary for digestion of fly ash because fly ash primarily consists of silicates and oxides. Most of the elements in ash were successfully extracted with hydrofluoric acid. The materials prepared by the method have a pore structure consisting of micropores as well as mesopores and macropores. The shape, composition, pore structure and mechanical properties of the zeolite microspheres make them interesting for application in areas such as adsorption for oil-spill cleanup.
References:
1. Metcalf and Eddy, Inc. (1999) ?WasteWater Engineering, 3 rd Edition. 765-915, TATA McGraw-Hill publishing company Limited, New Delhi.
2. Bhatia, S.C. (2002) ?Handbook of Industrial Pollution and Control?. 2:312, CBS publishers.
3. Cormack, d. (1983) Responses to oil and chemical Marine Pollution. Applied science publishers, New York.
4. Noyes, Robert (2005) ?Unit operations in Environmental Engineering. 307, Jaico publishing house.
5. Kiely uwe (2006) Environmental Engineering. 718, Irwin McGraw-hill.
6. Noyes, Robert (1993) Pollution Prevention Technology Handbook. 30, 39,105,206,502, Noyes publications, U.S.A.
7. Sharma, B. K. (2004) ?Industrial Chemistry, 14 th edition. Goel publishing house.
8. Willard, H. H. (1986)Industrial Methods of Analysis, 6 th edition. CBS publishers.
9. Gupta, v. (2006) Break through in oil-water separation. Environment science and Engineering, 57-58.
10. Rao, M. N. and Datta, A. K. (1978) ?Wastewater Treatment?, 2 nd edition. Oxford and IBP publishing Co. Pvt. Ltd.
11. Punmia, B. C. and Jain, A. K. (2005) Wastewater Engineering. Laxmi publications.
12. Gidde, M. R. and Lad, R. K. (2005) ?Environmental Engineering, 2nd volume. Nirali publication.
13. Chakravarty, R. N. and Bhaskaran, T. R. (1973) Treatment and Disposal of oil refinery wastes, IAWPC volume 10. 137- 153.
14. Haruna, J. and Meguro, M. (1992). JP Patent 04012015.
15. Kuntzel, J.; Ham, R. and Melin, T. (1999). Chem. Eng. Tech. 22(12), 991.
16. Kuntzel, J.; Ham, R. and Melin, T. (1999). Chem. Eng. Tech. 71, 508.
17. Otten, W.; Gail, E. and Frey, T. (1992). Chem. Ing. Tech. 64, 915.
18. Ruthven, D.M. (1988). Chem. Eng. Prog. 84, 42.


|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License