IJCRR - 4(13), July, 2012
Pages: 85-93
Date of Publication: 18-Jul-2012
Print Article
Download XML Download PDF
EFFECT OF FERMENTATION ON THE ANTI NUTRITIONAL FACTORS, ANTIRADICAL ACTIVITY AND IN VITRO PROTEIN DIGESTIBILITY OF CICER ARIETINUM L.
Author: Yadav Neelam, Sharma Shruti, Singh Alka, Tiwari Sudha
Category: Technology
Abstract:Objective: The aim of the present study was to find out the effect of fermentation on the physicochemical
properties (pH, total titratable acidity and protein content), antinutritional factors (phytate, total polyphenolic content (TPC) and tannin), antiradical activity and in vitro protein digestibility of desi Cicer arietinum seeds. Methods: Physicochemical parameters were assessed by using pH meter and titrimetric
method. Antinutritional factors such as phytate, TPC and tannin were determined by spectrophotometric,
Folin-ciocalteu and Folin\?denis method respectively and protein content and digestibility by Lowry's method and Pepsin-HCl method respectively. Results: Results showed that fermentation significantly reduced the pH from 6% to 4% and total titratable acidity from 1% to 4%. TPC in raw lentil was found to be 1011.4 mg/100g which was reduced to 150.5 mg/100g after 40 h of fermentation. There was 99% and
93% reduction in phytate and tannin content respectively after fermentation Antiradical activity was reduced from 90% to 65% at different intervals of fermentation. Fermentation significantly increased the in vitro protein digestibility from 43% to 75%.Conclusion: The study revealed that fermentation significantly improved the nutritive value of desi chickpea.
Keywords: Fermentation, Antinutritional, Polyphenols, Protein digestibility, Cicer arietimum.
Full Text:
INTRODUCTION
Fermentation is regarded as one of the oldest and most economical methods of processing and preserving foods aimed at prolonging shelf-life and improving palatability. It may also improve digestibility and nutritional value of food. Fermentation consists of modifying food by microorganisms that grow and reproduce and consume part of the substrate and enrich it with the products of their metabolism. It is an ancient technology that remains one of the most practical methods for preserving foods and enhancing their nutritional and organoleptic qualities. It is a desirable method for processing and preserving food because of its low cost, low energy requirements, and high yield, with acceptable and diversified flavors for human consumption (1) . Today, a variety of food products are derived from this technology in households, small-scale food industries as well as in large enterprises. Furthermore, fermentation is an affordable food preservation technology and of economic importance to developing countries. It enhances the nutritional quality of foods and contributes to food safety particularly under conditions where refrigeration or other foods processing facilities are not available (2).
Fermentation has been reported to cause a general improvement in the nutritional value of legumes. Whole or ground seeds, either raw or cooked, can act as substrate for fermentation. The fermented legumes are popular due to improved sensory characteristics, protein quality and digestibility and contents of some minerals and vitamins, as well as partial or complete elimination of antinutritional factors (3). Chickpea (Cicer arietinum) is a crop of economic importance and also an important source of protein in the diet of people in India as well as other. It is the third most important pulse crop in the world and is mainly grown in the semi-arid regions particularly in the Indian subcontinent and dry areas of the Middle East. The chickpea seeds are relatively higher in protein content (25 to 30 %), carbohydrate and calories than other legumes. Its seeds are also a good source of essential minerals like calcium, phosphorus, iron and vitamin B (4). Grain legumes or pulses, although are rich and low-cost sources of dietary proteins and nutrients for a large part of the world‘s population, their nutritive value is limited by the presence of several antinutritional and toxic substances and poor digestibility (5). Fermentation is a method that seems to enhance the nutritive value of legumes by increasing the levels of essential nutrients and reducing the level of antinutrients in foods. It ultimately contributes towards the improvement in the in-vitro digestibility of legumes (6). The aim of this work was to study the efficiency of fermentation on the reduction or elimination of antinutritional factors, improvement in the protein digestibility and changes in the physico-chemical properties of desi cicer arietinum seeds.
MATERIAL AND METHODS
Procurement of Raw Materials
Chickpea (Cicer arietinum) used for this investigation has been purchased from the local market of Allahabad city. All required chemicals and apparatus have been obtained from Centre of Food Technology, University of Allahabad. All the chemicals used in analysis were of AR (Analytical Reagent) grade.
Fermentation
The standard fermentation procedure was followed (7). The sample flour was mixed with distilled water (1:2 w/v) and incubated at 37ºC for 16, 24, 32 and 40 hours time intervals. The fermented samples were dried in hot air oven at 70ºC for 4 to 6 hours and the dried samples were grounded and stored in polyethylene bags at 4ºC for subsequent analysis.
Proximate, vitamin and mineral content
The AOAC methods (8) were used to determine proximate composition: drying at 105 ºC for 24 h for moisture (method 925.098); incineration at 550 ºC for ash (method 923.03); defatting in a Soxhlet apparatus with petroleum ether for lipids (method 920.39C with minor modifications). Carbohydrate content was estimated by difference method and the calorific value was estimated as given in Swaminathan (9).
pH and Total titratable acidity
The pH of the samples was determined according to the method of AOAC (10). The titrable acidity was estimated by titrating against 0.1 N NaOH to phenolphthalein end-point and the acidity was calculated as g lactic acid/100g (11).
Protein
Protein estimation was estimated by Lowry‘s method (12). The blue colour developed by the reduction of the phosphomolybdicphosphotungstic components in the FolinCiocalteau reagent by the amino acids tyrosine and tryptophan present in the protein plus the colour developed by the biuret reaction of the protein with the alkaline cupric tartrate was measured.
In vitro protein digestibility
The in vitro protein digestibility of the samples was determined by enzymatic method (13). A known weight of the sample containing 16 mg nitrogen was taken in triplicate and digested with 1 mg pepsin in 15 ml of 0.1 M HCl at 37ºC for 2 hours. The reaction was stopped by the addition of 15 ml 10% trichloro-acetic acid (TCA). The mixture was then filtered quantitatively through Whatman No. 1 filter paper. The TCA soluble fraction was assayed for nitrogen using the microkjeldahl method. Protein digestibility of the sample was calculated by the following formula:
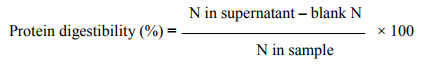
Total polyphenolic content
Polyphenols are extracted with 70% methanol from a test portion of finely ground sample at 70? C. The polyphenols in the extract was determined colorimetrically using Folin-Ciocalteu phenol reagent and gallic acid was used as a calibration standard (14). Phytate The phytate was extracted with trichloroacetic acid and precipitated as ferric salt. The iron content of the precipitate is determined colorimetrically and the phytate phosphorus content calculated from this value assuming a constant 4 Fe: 6 P molecular ratio in the precipitate (15). Tannin Estimation of tannins was done by Schanderel et al., (16) Tannin-like compounds reduce phosphotungstomolybdic acid in alkaline solution to produce a highly colored blue solution, the intensity of which is proportional to the amount of tannins. The intensity is measured in a spectrophotometer at 700 nm. Percent antiradical activity DPPH is a stable free radical that accepts an electron or hydrogen radical to become a stable diamagnetic molecule. The reduction capability of the DPPH radical is determined by the decrease in its absorbance at 516 nm induced by antioxidants. The absorption maximum of a stable DPPH radical in ethanol was at 516 nm (17).
Statistical Analysis
The results of fermentation at different hours are given as means ±standard error. Statistical analysis of variance (One-Way ANOVA) was done by using SPSS software 12 version, to determine differences among means. The statistically significant difference was defined as p≤0.01.
RESULTS
Proximate, Vitamin and Mineral Composition of chickpea
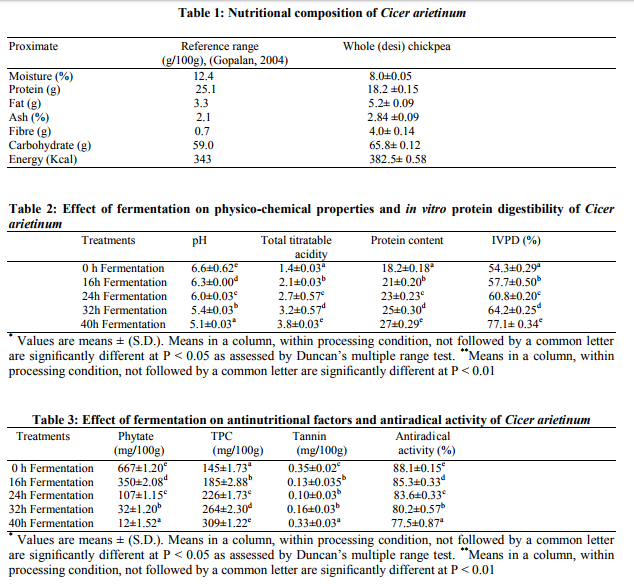
Table 1 shows the proximate, vitamin C and mineral composition of chickpea. These values were compared with the values given by Gopalan (18) in Food Composition table.
Effect of fermentation on the pH and total titratable acidity of chickpea
Unfermented flour of chickpea had a pH value of 6.6 (Table 2). Fermentation gradually reduced the pH with time. Fermentation of the flour for 16, 24, 32 and 40 hours had significantly (p≤0.05) dropped the pH to 5.2. Unfermented flour of chickpea had a Total Titratable Acidity (TTA) of 1.4% (Table 2). Fermentation gradually increased the TTA with time. Fermentation of the flour for 16, 24, 32 and 48 hours had significantly (p≤0.01) increased the TTA to 3.8%.
Effect of fermentation on the phytate content of chickpea
Phytate (mg/100g) content of chickpea is shown in Table 3 as affected by different periods of fermentation (16, 24, 32, 40 hours). Phytate content of the unfermented flour was 667 mg/100g. Fermentation of the chickpea significantly (p≤0.01) reduced the phytate content to about 98%.
Effect of fermentation on the total polyphenolic content of chickpea
The mean values for the effect of fermentation on total polyphenolic content are presented in Table 4.2. There is significant difference (p<0.01) among means. Fermentation at 16, 24, 32 and 40 h increased TPC by 27%, 22%, 16% and 17%, respectively (Table 3).
Effect of fermentation on the tannin content of chickpea
The tannin content of unfermented flour was 0.35%. Fermentation significantly (p≤0.05) reduced the tannin content by about 57% at the end of 40 hours. The values for tannin at different fermentation time periods are given in Table 3.
Effect of fermentation on the percent (%) antiradical activity of chickpea
The mean values for the effect of fermentation on antiradical activity are presented in Table 3. The analysis of variance of the data showed significant differences (p<0.05) among different time intervals. The percent antiradical activity of raw chickpea was 88%. During fermentation for 16, 24, 32 and 40 hr of chickpea, antiradical activity decreased by 3.4%, 2%, 3.6 % and 3.4% respectively.
Effect of fermentation on the protein content of chickpea
The protein content of raw chickpea was 18.2g/100g (Table 2). After 32 hours of fermentation period, the protein content was found to be 25.4g/100g. However, at the end of 40 hours of fermentation the protein content was observed to be 27.5g/100g. During fermentation for16, 24, 32 and 40 hr of chickpea, protein increased by 15.3%, 9.5%, 8.6 % and 8%, respectively.
Effect of fermentation on the in vitro protein digestibility of chickpea
The in vitro protein digestibility (IVPD) of unfermented chickpea flour was 54.3%. At the end of 40 hours of fermentation, the IVPD was found to be 77%. This increased value was observed to be significant (p≤0.01).
DISCUSSION
This decline in pH indicates the production of lactic acid. The pH drop was probably the result of microbial activity on the flour converting some of the carbohydrates into organic acids such as lactic acid, citric acid and acetic acids (19). According to these authors, the production of lactic acid bacteria during fermentation has attributed to the decrease in pH. Organic acids produced during fermentation also can potentially enhance Fe and Zn absorption via the formation of soluble ligands (20). The increased acidity and low pH as a result of fermentation enhances the keeping quality of fermented foods, by inhibiting microbial growth and also contributing to the flavor of the processed food (21) . The decrease in phytate content during fermentation might be due to activity of enzyme phytase naturally present in legumes and microorganisms in the dough. It has been suggested that the loss of phytate during fermentation could be a result of the activity of native phytase and/or the fermentative microflora by different workers (21; 19). Reduction in phytic acid contents of cereal and legume seeds with such processing treatments has been frequently reported (22). This has been attributed to an increase of phytase activities in fact; this enzyme makes the phytates soluble and released soluble protein and minerals. Fermentation also provides optimum pH conditions for enzymatic degradation of phytic acid which is present in cereals in the form of complexes with polyvalent cations such as iron, zinc, calcium, magnesium and proteins. With regard to the TPC content after fermentation, our results are in agreement with those reported in fermented Lens culinaris, V. sinensis and Glycine max (23) and others(24). Studies suggested that the fermentation process is an adequate and effective process for increasing the concentration of phenolic compounds and, hence, their nutritional and biological quality (25). Few workers have also reported an increase in polyphenol content of fermented flour with increase in temperature in pearl millet (26). Some authors have reported that polyphenols increase significantly or remained constant in the fermented food also natural fermentation was found to increase certain phenolic monomer like catechin (23). An increase in the concentration of total phenolic content during fermentation of millet and explained this by the hydrolytic activity of microorganism that degrade tannin phenolic compounds of low molecular weight (27). The decrease in tannin content after certain processing treatments in legumes was also observed by various authors (28). The decreases in the tannin content were attributed to the hydrolysis of polyphenolic compounds or tannin complexes during fermentation. It may also be due to the activities of microorganisms during fermentation which decreases the tannin content in the fermented product because microorganisms play a vital role in the reduction of tannins. These results were in agreement with earlier findings (29) who observed that optimum fermentation time is quite essential to obtain maximum tannin reduction in chickpea. Tannin reduction during fermentation might have been caused by the activity of enzyme tannase of fermenting microflora on tannin.This enzyme hydrolyses the deep side ester bond of hydroxylable tannin releasing gallic acid and glucose (5). Some studies have been also witnessed a decrease in percentage DPPH radical inhibition, amount of total phenolic compounds and super oxide anion radical inhibition as a result of fermentation (30). Few authors also stated that the presence of phytase produces flours with lower antioxidant activity might be due to the phytase acting on phytic acid, decreasing its concentration and liberating into the medium the phosphate groups and cations. Phytic acid is considered to be an antioxidant compound because it is a potent inhibitor of iron-catalyzed hydroxyl radical formation by chelating free iron (24). On the other hand, the liberated cations might favor the oxidation of different components of the medium. These two facts could be the cause of lower antioxidant activity. The increment in the per cent protein content during fermentation is also quantitative increment attributed to the utilization of carbohydrates by microorganisms (31;32). It can also be due to microbial synthesis from metabolic intermediates during fermentation an increment of raw protein during the fermentative process of ?Tape Ketan‘ and Pentaclethra macrophylla Benth variety (30). Increment of raw protein in the fermented product could be due to the protein hydrolysis product by extra cellular enzyme of fermented microorganism, such as proteases, promoting an increase in the total nitrogen content caused by the release of amino acid and short chain peptides. On the other side in cowpea no change in protein level has been reported (33). Antinutrients have the capacity of decreasing the content and increasing the palatability of the product because they form insoluble complexes with them (34). The antinutrients level are decreased in the fermented product and thereby increasing the protein content. The fermentation significantly improves the protein quality as well as the level of lysine in millet and other cereals (35). In the same way, during the fermentation of corn meal the concentrations of available lysine, methionine and tryptophan increase had been found to be increased (35). The poor protein digestibility of legumes is caused by phytic acid and polyphenols that bind to enzymes in the digestive tract and thus inhibit utilization of proteins (36). This adverse effect can be overcome by fermentation. In vitro protein digestibility is increased after fermentation because of partial degradation of complex storage proteins by endogenous and microbial proteolytic enzymes into soluble products (37). The increment in in vitro protein digestibility (IVPD) could be attributed to antinutrients degradation by microorganisms and to partial degradation of complex storage proteins into more simple and soluble products. It was suggested that fermentation causes structural changes in the storage proteins (prolamins and glutelins) making them more accessible to enzymatic attack. Studies have been also demonstrated that during fermentation, insoluble protein (prolamine and glutelin) under structural changes which makes them more accessible to pepsin attack, rather than being broken down into smaller sub-units. These changes are likely to have a marked effect on protein digestibility of seed protein and may be responsible for the increased protein digestibility. As a result of lactic acid fermentation, the protein digestibility can be elevated. Fermentation of food grains is known to be an effective method of improving the starch and protein digestibility and bioavailability of minerals.
CONCLUSION
On the basis of the above findings it is concluded that Fermented legumes are an integral and significant part of the diet of many people in developing countries, being one of the oldest and most economical methods of processing and preserving foods. Fermentation led to desirable changes in the legume, including an improvement in protein digestibility and enhancing of storage quality of the product, providing adequate amounts of food energy and a partial or complete elimination of anti-nutritional compounds (tannins, phytates and polyphenolc etc.). Therefore, natural fermentation of legumes has been a very effective process for increasing functionality of Cicer arietinum. Within the studied fermentation processes, whole grain fermentation is very promising, due to the obtained results as well as its lower cost.
ACKNOWLEDGEMENT
We are thankful to Prof G K Rai, Director IPS, University of Allahabad for providing all necessary facilities for this research work. I also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Deshpande SS and Salunkhe DK. (2000). Grain legumes, seeds and nuts: rationale for fermentation. Fermented grains legumes, seeds and nuts: a global perspective. FAO Agricultural Services Bulletin. 142:1-32.
2. Motarjemi Y. (2002). Impact of small scale fermentation technology on food safety in developing countries. International Journal of Food Microbiology. 75:213-229.
3. Tarar OM. (2009). Development, Characterization and Shelf Life Optimization of a Prototype Nutrient Dense Food Bar. M.Sc. thesis, University of Agriculture, Faisalabad, Pakistan.
4. Muehlbauer FJ, Cubero JI and Summerfield RJ. (1985). pp 266-311. Grain Legume Crops. pp 266-311. Collins, 8 Grafton Street, London, U.K.
5. Adewumi GA and Odunfa SA. (2009). Effect of controlled fermentation on the oligosaccharides content of two common Nigerian Vigna unguiculata beans (drum and oloyin). African Journal of Biotechnology. 8(11):2626-2630.
6. Granito MA, Torres JF, Guerra M and Vidal-Valverde C. (2005). Influence of fermentation on the nutritional value of two varieties of Vigna sinensis. Journal of European Food Research Technology. 220:176-181.
7. Omima EF, Abdullahi HET and Elfadil EB. (2010). Effect of fermentation on Biochemical Characteristics of Sorghum Flour Supplemented with Chickpea Flour. Journal of Applied Sciences Research. 6(7):860-865.
8. A.O.A.C. (1990). In Helrich K. (Ed.), Official methods of analysis. (15th ed.). Arlington, VA, USA.
9. Swaminathan (2003) Essentials of food and nutrition. Vol, I, 2nd Ed. Bangalore Printing and Publ Co. Ltd., Bangalore, p 101.
10. AOAC (1998) Official methods of Analysis. (16th ed.) Arlinghton, VA: Association of Official Analytical Chemists.
11. Mbata TI, Ikenebomeh MJ and Alaneme JC. (2009) Studies on the microbiological, nutrient composition and antinutritional contents of fermented maize flour fortified with bambara groundnut (Vigna subterranean L). African Journal of Food Science. 3(6):165-171
12. Lowry OH, Rosebrough NJ, Farr AL and Randall RJ. (1951) Protein measurement with Folin phenol reagent. Journal of Biological Chemistry. 103:265-275.
13. Monjula S and John E. (1991) Biochemical changes and in vitro protein digestibility of endosperm of germinating Dolichos lablab. Journal of the Science of Food and Agricuture. 55: 429-438.
14. ISO 14502-1:2005. Determination of substances characteristic of green and black tea -- Part 1: Content of total polyphenols in tea.
15. Wheeler EI and Ferrel RE. (1971). Methods for phytic acid determination in wheat and wheat fractions. Journal of Cereal Chemistry. 48:312-320.
16. Schanderl SH. (1970). Method in Food Analysis pp 709. Academic Press, New York.
17. Sanjha SD, Sheth NR, Patel NK, Patel D and Patel B. (2009). Characterization and evaluation of antioxidant activity of Portulaca oleracea. International Journal of Pharmacy and Pharmaceutical Sciences. 1:74-84.
18. Gopalan C, Sastri BVR and Balasubramanian SC (1989). Nutritive value of Indian foods, National Institute of Nutrition, Indian Council for Medical Research, Hyderabad, India.
19. Shimelis EA and Rakshit SK. (2008). Influence of natural and controlled fermentations on-galactosides, antinutrients and protein digestibility of beans (Phaseolus vulgaris L.) International Journal of Food Science and Technology. 43:658–665.
20. Gibson RS, Perlas L and Hotz C. (2006) Improving the bioavailability of nutrients in plant foods at the household level. Proceedings of the Nutrition Society. 65:160–168. Ibrahim, S. S., Habiba, R. A., Shatta, A. A., and Embaby, H. E. (2002). Effect of soaking, germination, cooking and fermentation on antinutritional factors in Cowpeas. Nahrung, 46; 92–95.
21. Elyas HAS, Tinay HA, Yousif EN and Sheikh AEE. (2002). Effect of natural fermentation on nutritive value and in vitro protein digestibility of pearl millet. Food Chemistry. 78:75-79.
22. Ibrahim, S. S., Habiba, R. A., Shatta, A. A., and Embaby, H. E. (2002). Effect of soaking, germination, cooking and fermentation on antinutritional factors in Cowpeas. Nahrung, 46; 92–95.
23. Bartolome B, Estrella I and Hernaandez T. (1997). Changes in phenolic compounds in lentils (Lens culinaris) during germination and fermentation. Z. Lebensmitlel Unters. 205:290-294.
24. Dueñasa M, Hernándeza T and Estrella I. (2007). Changes in the content of bioactive polyphenolic compounds of lentils by the action of exogenous enzymes. Effect on their antioxidant activity. Food Chemistry. 101:90-97.
25. Sullivan, T. W., Douglass, J. H., Andrews, D. J., Bond, P. L., HemcockJ. O., Bramelcox, P. J., Stegmeir, W. D., and Brethour, X. (1990). Nutritional value of pearl millet for food and feed. Proc. Int. Conf. on Sorghum Nutritional Quality, pp. 83–94.
26. Khetarpaul, N., and Chauhan, B. M. (1991). Effect of natural fermentation on phytate and polyphenolic content and in vitro digestibility of starch and protein of pearl millet (Pennisetum typhoideum). Journal of the Science of Food and Agriculture, 55(2), 189–195.
27. Hadimani, N. A., Ali, S. Z., and Malleshi, N. G. (1995). Physicochemical composition and processing characteristics of millet varieties. Journal of Food Science and Technoogy. Mysore, 32(3), 193–198.
28. Saxena, A. K., Sharma, A. K., Sehgal, K. L., and Bakhshi, A. K. (1992). Proximate composition and fatty acid make-up of some improved varieties of pearl millet in Punjab. Indian Journal of Nutrition and Dietetics, 29(5), 177–180.
29. Moreno, C.R., E.O.C. Rodríguez, J.M. Carrillo, O.G.C. Valenzuela and J.B. Hoyos. (2004). Solid state fermentation process for producing chickpea(Cicer arietinum L)tempeh flour. Physicochemical and nutritional characteristics of the product. Journal of Science Food Agricultural 84(3):271-278.
30. Oseni OA and Akindahunsi AA. (2010). Phytochemical Properties and Effect of Fermentation on the Seed of Jatropha curcas. American Journal of Food Technology. 6:158-165.
31. Awada SH, Hady A, Hassan AB, Ali MI and Babiker EE. (2005). Antinutritional factors content and availability of protein, starch and mineral of maize and lentil as influenced by domestic processing. Journal of Food Technology. 3(4):523-528.
32. Fadlallah E, Abdullahi H, Tinay E1 and Babiker E. (2010). Effect of Fermentation on Biochemical Characteristics of Sorghum Flour Supplemented with Chickpea Flour. Journal of Applied Sciences and Research. 6(7):860-865.
33. Akinyele IO and Akinlosotu A. (1999). Effect of soaking, dehulling and fermentation on the oligosaccharides and nutrient content of frijol (Vigna unguiculata). Food Chemistry 41:43-53.
34. Usha, A., Sripriya, G., and Chandra, T. S. (1996). Effect of fermentation on primary nutrients in finger millet (Eleusine coracane). Journal of Agricultural and Food Chemistry, 44, 2616–2818.
35. Hamad, A. M. and M. L. Fields (1979). Evaluation of the protein quality and available lysine of germinated and fermented cereal. Journal of Food Science. 44: 456.
36. Abdelhaleem WH, Tinay AH, Mustafa AI and Babiker EE. (2008). Effect of fermentation, malt-pretreatment and cooking on antinutritional factors and protein digestibility of sorghum cultivars. Pakistan Journal of Nutrition.7:335-341.
37. Calvin O, Noetzold H, Bley T and Henle T. (2004). Proximate composition and digestibility of fermented and extruded uji from maize–finger millet blend. Journal of the Swiss Society of Food Science and Technology. 8:827-832.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License