IJCRR - 4(22), November, 2012
Pages: 151-156
Date of Publication: 24-Nov-2012
Print Article
Download XML Download PDF
QSAR STUDIES OF PHTHALIMIDE DERIVATIVES FOR THEIR POTENT ANXIOLYTIC ACTIVITY
Author: Suvarna Prabhakar Gajare, Supriya S. Mahajan
Category: Healthcare
Abstract:Heterocyclic compounds represent an important class of biologically active molecules; specifically those containing the substituted imide nucleus have been shown to possess high biological activities. Phthalimide derivatives have been found to exhibit industrial, agricultural and biological applications. A series of phthalimide derivatives were synthesized and studied for their acute oral toxicity as per the OECD guidelines and anxiolytic activity using Elevated plus-maze animal model. The compounds were screened for anxiolytic activity using diazepam as the standard. Anxiolytic activity was calculated based on the per cent open arm entries and average time spent by mice on open arms. The QSAR studies were carried out by using molecular modeling software Maestro from Schrodinger, USA. The best QSAR model was obtained when anxiolytic activity was correlated with ionization potential (IP) values of phthalimide derivatives.
Keywords: Phthalimides, Acute oral toxicity, Anxiolytic activity, Elevated plus maze, QSAR, Ionization potential
Full Text:
INTRODUCTION
Phthalimide derivatives form an interesting group of compounds, many of which possess broad spectrum pharmacological properties such as analgesic1 , anticonvulsant2 , antitubercular3, 4 , hypolipidemic5 , anxiolytic6 , anti-inflammatory7 , antimicrobial8 and antipsychotic9 . Anxiety is an emotional state, unpleasant in nature, associated with uneasiness, discomfort and concern or fear about some defined or undefined future threat10 . During the last two decades, pharmacology with psychoactive drugs has been increasingly recognized as most effective in the management of anxiety, stress and psychosomatic disorders. The continuous usage of tranquilizers and psychotropic drugs has led to a variety of autonomic, endocrinal, allergic, hematopoietic and neurologic side effects. Elevation or depression of mood is another important side effect of such drugs. Tranquillizers have proved their efficacy in controlling anxiety and tension in many emotional as well as physical disorders. However, such agents primarily relieve the symptoms and offer relief for only a short duration10 . Elevated plus maze is the simplest apparatus used to study anxiolytic response of almost all the types of anxiolytic agents. Major advantages of this procedure are - a) it is simple, fast and less time consuming, (b) no noxious stimuli (sound or light) are required, and (c) it is a predictable and a reliable procedure for studying anxiety response as well as anxiolytic action of drugs11 . By QSAR models, the biological activity of a new or untested chemical can be inferred from the molecular structure of similar compounds, whose activities have already been assessed. The quantitative structure activity relationships study is essentially a computerized statistical method, which tries to explain the observed variance in the biological effect of certain classes of compounds as a function of molecular changes caused by the substituent 12, 13 .
MATERIALS AND METHODS
Chemistry
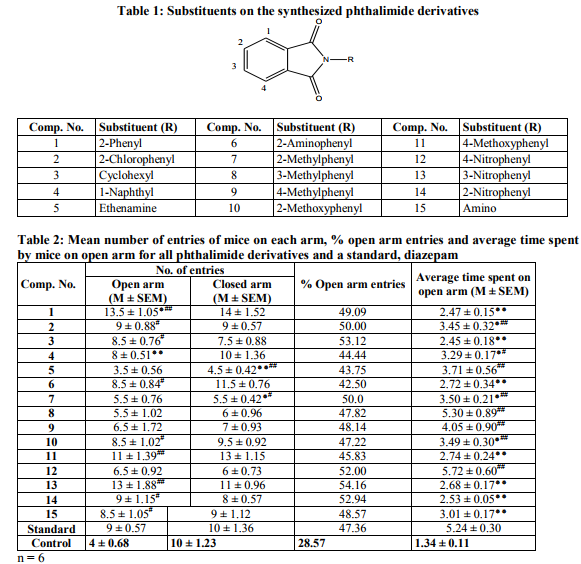
The chemicals required for the synthesis of phthalimide derivatives were purchased from Merck Specialties Pvt. Ltd., Spectrochem Laboratories, and Rankem Laboratories. All phthalimide derivatives were synthesized by ecofriendly method using microwave irradiation method14. Structural data of the synthesized derivatives is presented in Table 1.
Pharmacological Evaluation
The acute oral toxicity studies and anxiolytic activity were performed on Swiss albino mice of either sex, weighing between 25 and 30 g. All the animals were purchased from Haffkine Biopharmaceuticals Ltd., Mumbai, India. The animals were maintained at 25 ± 2 °C, 50 ± 5 % relative humidity and 12 h light/dark cycle. The animals were fasted for 24 h prior to the experiments and water provided ad libitum. The animal study protocols were approved by the Institutional Animal Ethics Committee of C. U. Shah College of Pharmacy, Mumbai, India. Acute oral toxicity studies15 Acute toxicity studies were performed as per the Organization for Economic Co-operation and Development (OECD) guidelines. Before experimentation, the animals were divided into the control group and the test groups, each group consisting of six animals. The control group received orally, a single dose of 10 ml/kg body weight of a control [1 % w/v sodium carboxymethyl cellulose (CMC) suspension]. The test compounds, at different dose levels of 500, 1000 and 2000 mg/kg body weight, were administered orally to the animals present in the test groups. After the administration of the test compounds, animals were observed for a period of 14 days for the changes in the skin, fur, eyes and behavioral pattern. Mortality of mice in each group was also observed. A dose leading to these changes or mortality was considered to be a toxic dose.
Anxiolytic activity16-18
Procedure:Elevated plus maze method Mice of either sex weighing between 20 and 25 g were used for determining anxiolytic activity of phthalimide derivatives. Six animals were used for the negative control and the positive control (standard) groups, each. The animals in the test groups were administered the test compounds orally at a dose of 200 mg/kg as a suspension in 0.5 % sodium CMC. The mice in the positive control group were treated with an oral dose of 1 mg/kg of diazepam in the form of a suspension in 0.5 % sodium CMC. The mice in the negative control group were administered orally 0.5 % sodium CMC (10 ml/kg). After an hour, the test animals were placed individually at the center of the maze, facing an enclosed arm. The anxiolytic activity was evaluated for 5 min as: 1) the number of entries by each mouse on the open arms, 2) the number of entries by each mouse on the closed arms, 3) the time spent by each mouse on the open arms, and 4) the time spent by each mouse on the closed arms. The anxiolytic activity was calculated as the per cent open arm entries and the average time spent on the open arms. The per cent open arm entries of mice was calculated by using the following formula.

Quantitative structure activity relationships (QSAR) studies
“Maestro” – the molecular modeling software from Schrodinger Inc, USA, was used to develop quantitative structure activity relationships models. The software LigPrep was used to get correct conformational structures of the synthesized phthalimides. The software QikProp provided different physicochemical parameters of phthalimides. The correlation between the biological activity and physiochemical properties of phthalimide derivatives was studied using the program Strike from Schrodinger. The QSAR studies of 15 phthalimide derivatives were performed by simple linear regression analysis, considering Log (% open arm entries) for anxiolytic activity as the dependent variable. The best QSAR model obtained for anxiolytic activity was validated by dividing the data set of 15 phthalimide derivatives into training set of 9 compounds and test set of 6 compounds. Distribution of compounds into two sets was done randomly. Internal validity of the best QSAR model was checked by correlating the observed and predicted biological activities of the training set compounds and external validity was checked by correlating the observed and predicted biological activities of the test set compounds. Statistical analysis The results of the anxiolytic activity were expressed as mean ± SEM (Standard Error of Mean) values. The statistical analysis for the anxiolytic activity of phthalimide derivatives was performed using one-way analysis of variance (ANOVA), followed by Dunnett?s test, for multiple comparison between the control group and the test groups, using the GraphPad software, USA. The „p? values less than 0.05 were considered to be significant.
RESULTS AND DISCUSSION
Acute oral toxicity studies
None of the synthesized compounds showed any significant changes in the skin, fur, eyes and other behavioral patterns in mice at any of the tested dose levels. No mortality was observed in the control and the test groups.
Anxiolytic activity of phthalimides
Anxiolytic activity of phthalimide derivatives is presented in Table 2. The well-known anxiolytic agent diazepam increases the per cent open arm entries and average time spent by mice on open arms. The per cent open arm entries given by diazepam was 47.36. Out of 15 compounds, 10 compounds showed better anxiolytic activity as compared to the standard, diazepam. Compounds 1, 8, 9 and 15 showed per cent open arm entries slightly lesser than 50, but it was more than that for diazepam, whereas compounds 2, 3, 7, 12, 13 and 14 showed per cent open arm entries equal to or more than 50. Remaining compounds showed per cent open arm entries in the range of 44-47. The average time spent on open arms by the mice treated with diazepam was 5.24 ± 0.30 sec. It was more than diazepam in case of compound 8 (5.30 ± 0.89 sec) and compound 12 (5.72 ± 0.60 sec). Thus, these compounds showed higher anxiolytic activity as compared to diazepam.
Development and validation of QSAR models
The best QSAR model (equation 1) obtained for the anxiolytic activity of the synthesized phthalimide derivatives is discussed below. Log (% open arm entries) = 1.2253 + 0.0041 IP……………………eq. 1 n = 9, r2 = 0.97, s = 0.0041, F = 26.5 The positive sign associated with ionization potential (IP) in equation 1 indicated that the compounds with high IP can show good anxiolytic activity. The correlation between the observed and predicted anxiolytic activities for the training and test set compounds is shown graphically in Figures 1 and 2, respectively. The high values of r 2 for the training set (r 2 = 0.982) and test set (r 2 = 0.923) indicated good internal predictivity and external predictivity of the best QSAR model.
CONCLUSIONS
Out of 15 phthalimide derivatives 10 were found to be good anxiolytic agents. Increase in the per cent open arm entries and average time spent by mice on open arms indicated reduction in fear in animals. Compounds with high IP showed good anxiolytic activity. New phthalimide derivatives showing higher anxiolytic activities can be designed and synthesized using the results obtained from the QSAR studies.
ACKNOWLEDGEMENT
Authors acknowledge the immense help received from the scholars, whose articles are cited and include in reference of this manuscript. The authors are also grateful to authors, editors and publishers of all those articles, journals and books from where the literature for this article has been received and discussed.
References:
1. Antunes R, Batista H, Srivastava R, Thomas G, Araujo CC, Longo RL. Synthesis, characterization and interaction mechanism of new oxadiazolo-phthalimides as peripheral analgesics. J Mol Str 2003; 660:1-13.
2. Bailleux V, Vallee L, Nuyts JP, Vamecq J. Anticonvulsant activity of some 4-amino-Nphenylphthalimides and N-(3-amino-2- methylphenyl) phthalimides. Biomed and Pharmacother 1994; 48:95-101.
3. Jean SL, Paulo YR, Chung CM, Celio TH, Fernando PR, Clarice LQ. Synthesis and in vitro anti mycobacterium tuberculosis activity of a series of phthalimide derivatives, Bioorg Med Chem 2009;17:3795–3799.
4. Babu AH, Ramana AV, Sinha R, Yahav JS, Arora SK, Antitubercular agents. Part 1: Synthesis of phthalimido and naphthalimido linked phenazines as a new prototype antitubercular agent. Bioorg Med Chem 2005;15:1923-1926.
5. Chapman JM, Cocolas GH, Hall IH, Hypolipidemic activity of phthalimide derivatives. A comparison of phthalimide and 1,2-benzisothiazolin-3-one 1,1-dioxide derivatives to phthalimidine and 1,2- benzisothiazoline 1,1-dioxide congeners. J Med Chem 1983;26 (2):243–246.
6. Hassanzadeh F, Rabbani M, Khodarahmi GA, Fasihi A, Hakimelahi GH, Mohajeri M, Synthesis of phthalimide derivative and evaluation of their anxiolytic activity, Res Pharm Sci 2007; 2: 35-41.
7. Collin X, Robert J, Wielgosz G, Le BG, Bobin-Dubigen C, Grimaud N. New antiinflammatory N-pyridinyl (alkyl) phthalimides acting as tumour necrosis factor-alpha production inhibitors. Eur J Med Chem 2001;36:639 -649.
8. Patel HS, Mistry HJ, Patel NK, Desai SN. Synthesis and antimicrobial activity of some new phthalimide derivatives. Bulgarian Chem Comm 2004;36:167 -172.
9. Al-Rashood KA, Mustafa AA, Alhaider AA, Ginawi OT, Madani AAE, El-Obeid HA. Antipsychotic properties of new N-(4- substituted-1-piperazinylethyl) and N-(4- substituted-l-piperidinylethyl)-phthalimides. J Pharm Sci 1988;77: 898-901.
10. Tripathi, KD. Essentials of medicinal pharmacology. 4th ed. New Delhi: Jayee brother publication; 1993. p.752.
11. Koslow SH, Murthy R, Coelho, GV. Plants and plant products for mental health, us department of health and human services, 4th edition, 1995, 163-171.
12. Kubinyi H. Burger?s medicinal chemistry and drug discovery: the quantitative analysis of structure activity relationships, 5th ed. New York: John Wiley and Sons; 1995(1) p. 497- 552.
13. Mure SA, Bologab CM, Mracecb A, Chiriacb B, Jastorffc ZS, Náray-Szabóe G. Comparative QSAR study with electronic and steric parameters for cAMP derivatives with large substituents in positions 2, 6 and 8, J Mol Str: Theochem 1995;342: 161-171.
14. Gajare SP, Mahajan SS. Eco-Friendly synthesis of phthalimide derivatives, their analgesic activitiy and QSAR studies. I J Pharm Phytopharmacological Res. In press
15. OECD (2008) OECD Guidelines for the Testing of Chemicals Test No. 425: Acute Oral Toxicity – Up and Down procedure.
16 Sharma K, Arora V, Rana AC, Bhatnagar M. Anxiolytic effect of convolvulus pluricaulis choisypetals on elevated plus maze model of anxiety in mice. J Herb Medi Toxi 2009;3: 41-46.
17 http://www.answers.com/topic/anxiolytic activity - animal model, visited on December 2010.
18 Kulkarni SK. Handbook of experimental pharmacology, 3rd ed. New Delhi: Vallabh Prakashan 1999; p. 27-32, 123-128.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License