IJCRR - 4(22), November, 2012
Pages: 47-53
Date of Publication: 24-Nov-2012
Print Article
Download XML Download PDF
STUDY OF PHYTOHORMONES EFFECT ON MICROPROPAGATION AND SHOOT INDUCTION IN ASPARAGUS RACEMOSUS
Author: Anurag Jain, Krishan Kumar, Mukesh Kumar
Category: General Sciences
Abstract:The present investigation was undertaken to study the effect of different concentrations of BAP, Kin, Ads on shoot induction in Asparagus Racemosus. There was shoots induction after 3 weeks when Nodal explants segments were cultured on basal medium supplemented with BAP, Kin and Ads. Shoots induction on MS basal medium supplemented with BAP (1 mg/l) + kinetin (1 mg/1) were transferred to MS basal medium containing BAP (1.5mg/1) + Ads (100mg/1) and BAP (1.5mg/1), Kin (l mg/1) respectively to the Proliferation shoots. Stem segments cultured on MS basal medium supplemented with various concentrations of BAP (0.75, 1.5,1.0 mg/1), AdS (50,100 mg/l) and Kin (0.75,1.0,1.5 mg/l) induce shoot, But a combination of l mg/1 BAP + 1 mg/l Kin and 1.5 mg/1 BAP + 100.mg/1 Ads showed highest percentage of shoot proliferation after 7-14 days.
Keywords: Asparagus Racemosus, shoot induction, BAP, Kin and Ads
Full Text:
INTRODUCTION
Asparagus racemosus Wild is a multidimensional medicinal plant. Family Liliaceae English name Asparagus, Indian name shatmuli, Satavari, (Sanskrit) Satawar, satavari, (Hindi) , Its distribution Tropical and subtropical India. It is straggling or scan dent, much branched, spinous shrub. The plant is a climber growing to 1-2m in length. The leves are like pine needles, small and uniform. The inflorescence has tinny white flowers, in small spikes. The roots are finger like and clustered (Purohit and Vyas, 2004). It has thorny branches the ripen fruit is small, round and red. Seeds are black. Stem woody, whitish gray or brown armed with strong, straights or reduced spines, 5-13mm long; cladodes more or less acicular, 2-6 Nate, falcate; finely acuminate; leaves reduced to sub-erect or sub-recurred spine, fragrant, small profuse in simple or branched racemes up to 7m long; scarlet, tri-lobed, 4-6 in diameter (Dutta, 2007). Its phytoestrogen properties are extensively used in combating menopausal symptoms and increasing lactation (Sabin et al., 1968). It also has antioxidants (Kamat et al., 2000) and properties (Rage et al., 1989) and is widely used in Ayurveda for treating dyspepsia (Dalvi et al., 1990). The major active medicinal constituents are steroidal saponins shatavarins I-IV that is present in the roots of the plant (Sairam, 2003). It has been reported in the Indian and British Pharmacopoeias and in traditional systems of medicine such as Ayurveda, Unani, and Siddha. The multiple uses of this species have increased its commercial demand, resulting in overexploitation. Because of destructive harvesting, the natural population of it racemosus is rapidly disappearing, and it is recognized as vulnerable (Bopana and Saxena 2001). The development of an efficient micropropagation protocol will play significant role in meeting the requirements for commercial cultivation, thereby conserving the species in its natural habitat. According to ayurveda, asparagus is much useful in case of reproductive disease. Plant is reach source of plant derived estrogens. It is good source of folic acid, potassium, dietary fiber. It is cardio tonic, hypoglycemic, antioxytocis to uterine contraction, diuretic, antioxidant, insulin secreting an improving potency, many formulation based on Asparagus are in commerce used for bleeding disorders, gout, low sperm count (Goyal et al., 2003). The methanol extracts of roots of Asparagus wild is reported to show considerable in vitro antibacterial activity against various common pathogens (Mandal et al., 2007). There is nothing to hoist in speaking that A. racemosus is a doctor. It is suggested in nervous disorders, dyspepsia, diarrhoea, dysentery, neuropathy, cough, certain infectious disease (Goyal et al., 2003). Root paste which stimulates milk secretion, is used as an invigorating tonic to lactating women and live stock. It is taken to treat high fever. Root tubers are fed to get relief from milking disorder of cattle, which is regarded as appetizer. They are also useful in dysentery, tumors, inflammations, disease of blood and eyes, throat complains, tuberculosis leprosy, epilepsy and high blindness. Roots are also used in disease of Kidney and liver, scalding urine, gleets. Fruits are eaten to treat pimple (Dutta, 2007; Rajbhandari, 2001). The whole plant is used for treatment of diarrhoea, Diabetes, and Rheumatism. Seed are also used for blood purification.
MATERIALS AND METHODS
Preparation of culture media: Double distilled water was used for the preparation of medium. The amounts of macro and micro nutrients, organic salts, vitamins, growth regulators and sucrose were added to the double distilled water kept in distilled water. The final volume was made in a graduated cylinder/breaker by adding double distilled water. The pH of the solution was adjusted to 5.7-5.8 using either 0.1N HCl or 0.1N NaOH. For solidification of the medium, agar powder (Tissue culture grade, agar-agar type) @ 0.8% w/v was added to warm solution and then, boiled for proper dissolving and melting of agar powder. Then the medium was poured in glass vessel (i.e. culture tube or culture bottle). After that the vessel was covered by lid or with aluminium foil. Basal nutrient media used during this investigation are given in table was BM1- BM7 modification of Murashige and Skoog medium (1962) was used in present project.
Procedure
The medium was prepared in sterile flasks. Required quantities of stock solutions 50 ml of stock 1, 5ml of stock 2, 10ml of stock 3, , 10ml of stock 4, 10ml of stock 5 was added. The pH of the solution was adjusted 5.8 by adding NaOH and HCl drop by drop. Final volume of medium was made to 1 litter by adding distilled water. Adding of 8% agar, which was dissolved in warm water followed by constant stirring. The medium thus prepared was dispensed into different culture tubes or culture bottles each having 20 to 30 ml of the media. The medium was autoclaved at 15 lbs pressure and 121°C temperature for 20 minutes. The medium was finally allowed to cool and solidify. The tissue culture technique involves the isolation, sterilization, inoculation and cultivation of plant cells, tissues and organs under aseptic conditions in culture vials, containing nutrient medium. In tissue culture, the organized structures like shoot tips, root tip etc are culture in-vitro to obtain their development as organized structure under controlled environmental conditions (temperature, light, humidity, aeration) which effectively controls the expansion of any genotype or phenotypic potential in explants. Isolation of Explant Source of explants: The explant parts such as nodal segments shoot tip, and In vivo germinated seeds were taken from plants of Asparagus racemosus Wild in herbal garden of SINGHANIA UNIVERSITY. Pacheri Bari, Jhunjhunu.(Rajasthan) during February - March seasons and from in vitro germinated seeds. The specific differences in the regeneration potential of different organs and explants have various explanations. The significant factors include differences in the stage of the cells in the cell cycle, the availability of or ability to transport endogenous growth regulators, and the metabolic capabilities of the cells. The most commonly used tissue explants are the meristematic ends of the plants like the stem tip, auxiliary bud tip and root tip. These tissues have high rates of cell division and either concentrate or produce required growth regulating substances including auxins and cytokinins.
METHOD
Sterilization of explants: Young stem segments, young shoot tip and seeds were surface sterilized, by first washing them in Extran detergent in running tap water for 10- 15 min to remove soil particles and debris, after that were wash with Bavestin (100mg/ml) 2-3 drop in 100 ml water then wash were with distilled water, dipping in absolute alcohol for 2 min and also dipping in 20% (v/v) commercial sodium hypochlorite solution for 5- 6 min. wash with sterilized distilled water for one times , followed immediately, immersing them in 0.1% (w/v) HgC12 solution for 5 min treatment under Laminar Air Flow Unit, they were thoroughly washed 5-6 times in sterile double distilled water. This entire treatment was carried out at low temperature to prevent damage to the shoots. Inoculation Before inoculation, inside the laminar air flow chamber the ultraviolet lamp was put on carefully to avoid contamination for at least 20 minutes, the working table of laminar air flow chamber was wiped thoroughly with 70% ethanol before use. The material required for inoculation was steam sterilized. The hands were .cleaned with 70% ethanol. Then, the individual explants were inoculated in individual culture tubes or culture bottles having solidified culture medium. Forceps and scalpels were flame sterilized before each inoculation. The explants were then, cultured on MS medium supplemented with different concentration of BAP, Kn, AdS and NAA.

Incubation of Culture
After inoculation cultures were kept in the incubation room where the temperature and light period was maintained at 25 ± 20C and 16 hr. photoperiod with approximate 1500 lux intensity of light respectively. Source of the light was from the four florescent tube lights each of four feet and 40 W (Philips make) a total of 22 culture bottles for each treatment has been kept on the culture racks. After three week period callus developed in the medium or shoots/roots develop from explants.
RESULTS AND DISCUSSION
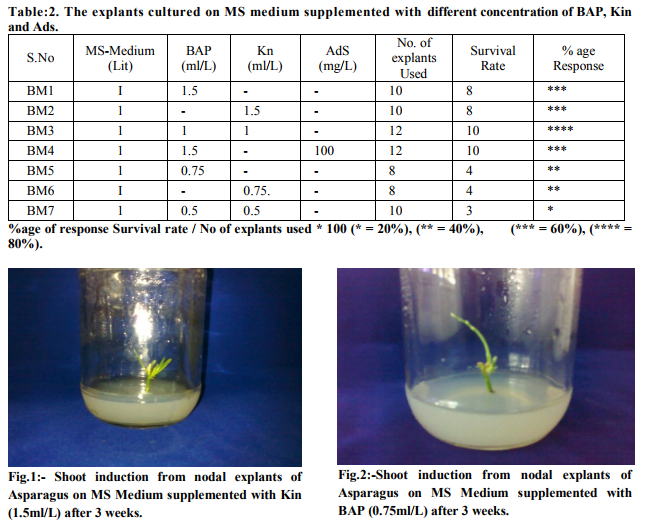
Shoot Initiation: Nodal explants were cultured on MS medium supplemented with different concentrations of BAP, Kin and Ads. The nodal segments get swollen after 3 days and the small originating shoot segment were observed as shown in table 2.When the hormone concentration is given as1.5ml BAP(fig.4) ,1.5ml Kn(fig.1) and 1.5mlBAP and 100mg Ads in 1l MS medium then 60% response was observed. When the hormone concentration is given as 1ml BAP and 1ml Kn in 1l MS medium then 80% (maximum) response was observed (fig.3). When the hormone concentration is given as respectively 0.75ml BAP (fig.2) and 0.75ml Kn(fig.5) in 1l MS medium then 40% response was observed. When the hormone concentration is given as respectively 0.5ml BAP and 0.5ml Kn in 1l MS medium then 20% response was observed (fig.6).Good response was observed in fig.7 and fig.8 when transferred to same medium. The explants showed shoot initiation after three weeks.


DISCUSSION
Culture Establishment (Stage-1): Asparagus racemosus has a variety of usages. The first step on initiating in vitro culture is to successfully control the fungal as well as bacterial contaminations. The general surface sterilization procedure i.e. mercuric chloride for 5 min. failed to control the microbial infection in the explants. Hence, two procedures of surface sterilization were tried in the present study. The treatment of nodal explants with mercuric chloride for 5 min followed by sodium hypo chloride for 5 min. (T2) was found to be the best in respect of explants survival (90%) and reduced explants contamination over the other treatments. Sodium hypochloride has bactericidal action and is generally used for surface sterilization and also helps in the controlling the microbial infection in the case of shoot proliferation. Shoots Proliferation (Stage-2): When the nodal explants inoculated on MS medium supplemented with different concentrations of BAP, Ads and Kinetin in different combinations. The best shoot induction was achieved when the cultures were transferred to MS medium supplemented with BAP (1 ml/lit.) + kinetin (1 ml/lit.) and BAP (1.5ml/lit.) + AdS (100 mg/lit.). In other experiment study reported in Asparagus racemosus in vitro shoot proliferation was obtained by culturing single node segments in Murashige and Skoog's (MS) medium supplemented with 3.69 μM 2- isopentyl adenine and 3% sucrose with a multiplication rate of 3.5 (Bopana and Saxena., 2008). Shoot proliferation (Stage-3): The shoot proliferation medium comprises of different concentration of BAP and Ads in different combinations. The best proliferation with well differentiated micro-shoots was achieved when the small nodal segments were transferred to MS medium supplemented with BAP (1.5 ml/L) and Ads (100mg/lit.).The treatments recorded 15-20 % proliferation after 1 week of first sub-culture. The shoot proliferation in tissue culture is largely due to the action of BAP. Optimum dose of BAP enhances the multiple shoot proliferation as well as length. In other experiment study reported high rate of multiple shoots was obtained from nodal explants of Asparagus racemosus Roxb. On MS supplemented with 0.27 µM NAA, 0.46 µM Kn and 0.6 agar (Mehta and Subramanian, 2005).
CONCLUSION
Study was conducted on Micropropagation of Medicinal Plant Asparagus racemosus Wild. The following conclusions are: Nodal segments and apical buds of 20-30 days old Asparagus racemosus wild Plants were inoculated on MS medium supplemented with BAP (1ppm/L) + Kinetin (1ppm/L) and BAP (1.5ppm/L) + Ads (100mg/L) and then transferred to same medium was found best shoot induction. Proliferated shoots were separated and transferred to NAA (0.5mg/l) + BAP (1mg/l) + kin(1mg/l) medium was found to best roots formation.
References:
1. Bopana, N. and Saxena, S. (2008). In vitro propagation of a high value medicinal plant In Vitro Cell, Dev, Biol.-Plant 44:525-532.
2. Dalvi, J., Hayes, P. Y. and Johidin, A. H. (2007). The biology of Australian weeds Asparagus asparagoides (L.) AustraliaPlant Protection Quarterly 21(2): 33-36.
3. Dutta, N. K., Roy, R. N. and Chavan, S.R. (2007). Steroid saponins from the roots of Asparagus racemosus Willd. N. 0. Liliaceae, JRIM. 6(2):I32-138.
4. Goyal, R. K., Singh, J. and Lai, H. (2003). immunomodulatory activity of Asparagus racemosus on systemic Th1/Th2 immunity: Implications for immunoadjuvant potential Indian jurnal Med Sci, 57(9): 408- 14.
5. Kamat, J. P., Boloor, K. K., Devasangaram, T. P. A. and Venkatachalain, S. R. (2000). Antioxidant properties of Asparagus racemosus against damage induced by gamma radiation in rat liver, mitochondria 71(3):425-435.
6. Mandal, S. C., Kumar, A., Lakshami, S.M., Sinha, S., Saha, B. P. and Pal, M. (2002) Antitumor effect of Asparagus racemosus root against sulfur dioxide induced cough in mice Fitoterpia j, science 71(6):686-689.
7. Mehta, S.R. and Subramanian, R.B. (2005). Direct In vitro Propagation of Asparagus adscendens Roxb, Plant Tissue Cult. 15(1): 25-32.
8. Purohit, S.S. and Vyas, S.P. (2004). A scientific approach including processing and tiniancial guidelines of Asparagus racemosus Jhodhpur Agrbios, 62 4p., tables, ISBN 81 7754-214 -1.
9. Rege, N. N., Thatte, U. M., Dahanukar, S. A. (1999). Adaptogenic properties of six. rasayana herb used in Ayurvedic medicine Phytother Res. 13(4):275-9L
10. Sabins, P.B., Gaitonde, B. B., Jatmalani, M. (1968). effect of alcoholic extract of Asparagus racemosus on mammary gland of rats. Indian]. Exp. Bid.6:55-7
11. Sairam, K., Priyambada, S., Aryya, N.C. and Goel, R.K. (2003). Gastroduodenal ulcer protective activity of Asparagus racemosus: an experimental, biochemical and histological study J Ethanopharmacol, 86: 1-10.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License