IJCRR - 4(22), November, 2012
Pages: 29-34
Date of Publication: 24-Nov-2012
Print Article
Download XML Download PDF
IN VITRO MICROPROPAGATION OF LAWSONIA INERMIS: AN IMPORTANT MEDICINAL PLANT
Author: Pooja Singh, Kirti Jain, Bharti Jain, Swati Khare
Category: General Sciences
Abstract:Lawsonia inermis (Lythraceae) is commonly called as Heena. Lawsonia inermis L. is a medicinal as well as commercially used plant. It has been found that conventional methods of propagation of L. inermis, sexual as well as vegetative, are beset with many problems that restrict their multiplication on a large scale. Propagation through seed is unreliable because of disease and pest problems, short viability and heavy rains during the seeding season in the natural habitat. Therefore, it is necessary to use in vitro propagation for rapid plantproduction. For shoot formation both apical shoot buds and meristems were used. This was achieved on MS medium containing different concentration of BAP alone or in combination with Kinetin was tested. Best shoot initiation response was achieved on MS Medium supplemented with 0.2 mg/l BAP + 0.2 mg/l kn on both apical shoot and axillary meristem explants.
Keywords: Lawsonia inermis, in vitro micropropagation, germplasm, aroma etc.
Full Text:
INTRODUCTION
Lawsonia inermis Linn (Lythraceae) is commonly called as Heena or Mehndi is an agricultural and economically important medicinal plant known for its cosmetic and traditional medicine properties, Lawsonia is monotypic genus, native of North Africa and south-west Asia widely cultivated. It is also cultivated in many tropical andwarm temperate regions as a hedge plant. The leaves that yield the dye confined to India, manly in Punjab and Gujarat and to a small extent in Rajasthan and Madhya Pradesh. Lawsonia inermis is a popular skin and hair coloring agent in many parts of the world. L. inermis is a glabrous, much branched shrub or small tree and grows wild. It needs around 5 years to mature and produce leaves with useful levels of tannins. The plant produces heavy white and yellow flowers. The flower of Lawsonia has a strong aroma with high commercial value. It is extensively used as a dye in silk and wool industry. Its root is considered a potent medicine for gonorrhea and herpes infection. In addition, it is a medicinal plant traditionally used by diverse groups of tribal/ethnic people Lawsonia inermis is used as an antirheumatic and antineuralgic agent (Marc, 2008), and also has potential as an antidiabetic drug(G.R.Rout, et.al, 2001).
MATERIALS AND METHODS
The technique involves the isolation, inoculation and regeneration of plant cells, tissues, and organ under aseptic culture vials, containing synthetic nutrient medium. Both the chemical compositions of the medium and the aseptic (light, temperature, humidity, aeration etc.) effectively control the expression of any genotype or phenotype in the explants. Actively growing young stems (4-5 cm) of L. inermis were collected from Sarojini Naidu Govt. Girls P.G. College Bhopal.
Surface sterilization procedure- Meristem were thoroughly washed under running tap water for 30 min then treated with 5% tween-20 for 5 minutes with constant stirring followed by 3-4 rinses in sterile distilled water and further treated with an antifungal agent (Bavistin) for 2 hours and were further with detergent for 10 min. and rinsed 4-5 times tap water. Further sterilization procedures were carried out inside laminar air flow chamber, where Meristem were surface sterilization through single dip in 70% (v/v) for 1 minute followed by three times rinses in sterile distilled water. There after mercuric chloride (0.1%) treatment was given to explants for 8 minutes followed by four times rinsed in sterile distilled water. Thereafter meristem were carefully transferred to be placed over sterile petridish and were then inoculated into the culture establishment medium inoculated into the culture establishment medium (MS Medium; Murashige and Skoog 1962) using sterile forceps under aseptic conditions. The explant placed verticallally on the culture medium.2 (MS Medium; Murashige and Skoog 1962)3 using sterile forceps under aseptic conditions. The shoots induced from the in-vitro cultures were used as explants for further experiment (Razdhan).
Selection of explants -Apical soot tip and meristems node of 25 day old in vitro raised seedlings were selected as explants for direct shoot initiation. The apical shoot tip and meristems node segment of 5-8 mm in length were excised aseptically.
Chemicals - Apical shoot bud and meristems induced were cultured on MS basal medium supplemented with 3 % (w/v) sucrose (Sd-fine Chemicals, India) for shoot induction. The pH of the medium (supplemented with respective growth regulators) was adjusted to 5.7 with 1N NaOH or 1N HCl before gelling with 0.8 % (w/v) agar. In all the experiments, the chemicals used were of analytical grade (Merck and SD-fine Chemicals, India). The medium was dispensed into culture vessels (Borosil, Mumbai, India) and autoclaved at 105 kPa at 125°C for 15 minute. The surface sterilized explants were placed vertically on the culture medium. All the cultures were incubated at 25±2°C under 16h light/8h dark photoperiod with irradiance of 45 - 50 μ mol/ m²/s photo synthetically active radiation (PAR) provided by cool white fluorescent tubes (Philip, India) and with 60 - 65 % relative humidity. All subsequent subcultures were done at four weeks intervals. Culture media consisted of MS (Murashige and Skoog 1962) supplemented with 3% (w/v) sucrose and 0.8% (w/v) agar (Himedia, India) was evaluated for their effects on in-vitro growth and development of L. Inermis. For induction of shoots, explants were cultured on MS medium supplemented with different concentration of cytokines, including BAP (0.1- 0.5mg/l), Kn (0.1-0.5 mg/l) and CW (0.1-0.5 mg/l) either individually or in combination. Application of tissue culture to plant conservation in India has been largely restricted to economically important species However, the approach could usefully be extended to conserve all threatened plants so that vital biodiversity and the ecological network is sustains can be preserved.
Shoot induction - Apical soot tip and meristems node were excised and inoculated by vertical orientation on the culture medium containing different concentration of BAP (0.1-0.5mg/l), Kn (0.1-0.5 mg/l) and CW (0.1-0.5 mg/l). Explants were assigned randomly to each treatment and culture were kept under 16 h light/day photoperiod at 25±2°C.shoot induction was assessed after process same different media composition for further experiment.
RESULTS
The explant show better response when collected and inoculated from august to march. The explants sterile for 40 sec to 2min with 0.1% HgCl2 . This solutions show 75 to 85 % sterile culture.
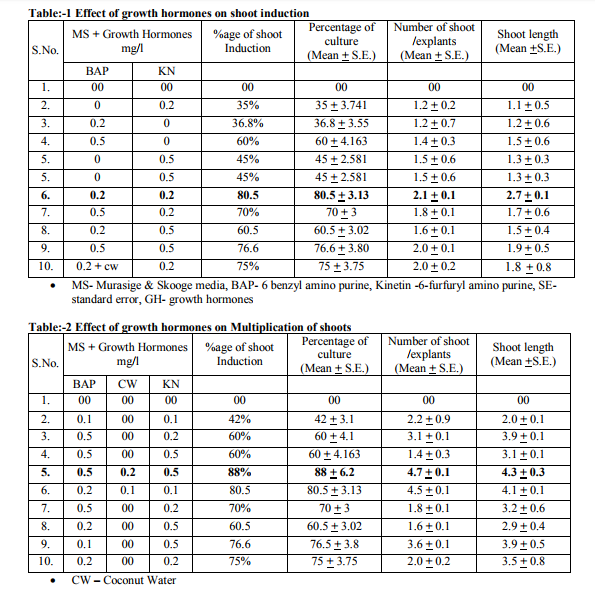
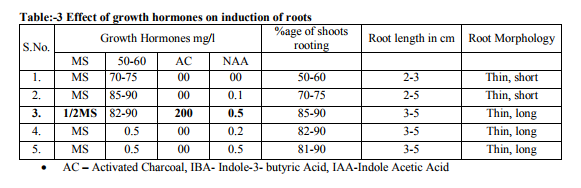
The meristem (apical and axillary) was placed on semi-solidbasal MS(MurashigeandSkoog (1962) medium supplemented with different concentrations and combinations of 6-benzylaminopurine (BA:0.0, 0.1, 0.2 and 0.5 mg/l), kinetin (Kn: 0.0, 0.1, 0.2 and 0.5 mg/l), and for shootinduction. The pH of t h e m e d i a w a s a d j u s t e d t o 5 . 8 u s i n g 0.1 N NaOH or 0.1 N HCl before autoclaving. Routinely, 25 ml of the molten medium was dispensed into culture tubes, plugged with non-absorbent cotton wrapped in one layer of cheese cloth and sterilized at 121ºC and 1.06kg/cm2 of pressure for 15 minute cultures were maintained at 25±2ºC either under a 14 hr photoperiod or continuous light from cool, white, fluorescent lamps. Theculturesweremaintainedbyregular subcultures at 4 week intervals on fresh mediumwiththesamecompositions.The induced shoot was placed on semi-solidbasalMS (Murashigeand Skoog (1962) medium supplemented with different concentrations and combinations of 6-benzy- laminopurine(BA:0.0, 0.1, 0.2,and0.5mg/l), kinetin (Kn: 0.0,0.1, 0.2, and 0.5mg/l), and (CW: 0.0, 0.1 and 0.2 mg/l) for shootproliferationandmultiplication. The explant undergoes rapid tissue or shoots multiplication (see pic 3-5). This process can be repeated several times, depending upon how many plants are ultimately desired. Further, Invitro induced shoot of Lawsonia inermis L. excised and cultured separately in fresh medium Containing NAA(0.1, 0.2 and 0.5) along or combination with IBA(0.1, 0.2 and 0.5) and activated charcoal(200mg/l) to encourage formation of long shootsbroad leaves and basal roots(pic 6-7).
DISCUSSION
The important part of present study was the preparation of contamination for explant.This was achieved by using Apical as wel as nodal meristem as explant as explants. Sterilization of Meristem required 0.1% (w/v) HgCl2 1.5- 2 min depending on the thickness of meristem. Treatment for maximum germination and minimum contamination and effect of growth hormones on shoot regeneration. Apical and nodal part can be successfully surface sterilized using 0.1 HgCl2 with an exposure time of minutes followed by dipping in 70% ethanol for 1 minutes. They can be germinated successfully invitro in Basel medium solidified with agar. Shoots excised from in-vitro grown multiplied shoots can be MS medium with 0.2 BAP mg/l, 0.2mg/l Kin and 0.2 CW to highest percentage. Future experiments should be focused on shoot in few sub cultures, rooting of shoots and hardening before commercialization of the technology. And this study describes a procedure for in-vitro micro propagation through shoot induction and successful growth of induced shoots of Lawsonia inermis in in-vitro conditions. The results of this study shown that tissue culture techniques can play an important role in clonal propagation of elite genotypes of. Lawsonia inermis which has diverse medicinal applications and eventually due to over exploitation this plant is facing local extinction. There is pressing need to conserve this medicinal herb of high commercial value Lawsonia inermis usually multiply by root secures and seeds but due to low germination capability it restricts for the regeneration. It seems likely that this protocol for shoot induction, possibly with modification, can be used for in-vitro shoot induction of other species using apical shoot bud and meristem segments.
References:
1. A Akter, FA Neela, M. S. I. Khan, MS Islam and MF Alam, Screening of ethanol, petroleum ether and chloroform extracts of medicinal plants, Lawsonia inermis L. and Mimosa pudica L. for antibacterial activity, Indian J Pharm Sci,72(2010).
2. Anon, Theuseful plants of India. Publications and Information Directorate, CSIR, New Delhi, India, (1986).
3. B.Upadhyay, A. K. Dhaker, K.P. Singh andA. Kumar , Phytochemical analysis and influence of Edaphic factors on L:awsonia inermis, Journal of Phytology, 2(2010).
4. G.R.Rout,G.Das, S.SamantarayandP.Das, InvitromicropropagationofLawsoniainermis(L ythraceae), International Journal of Tropical Biology and Conservation, 49(2001).
5. GagandeepChaudhary, Sandeep Goyal and Priyanka Poonia, Lawsonia inermis L.:A Phytopharmacological Review, International Journal of Pharmaceutical Sciences and Drug Research, 2(2010).
6. Gallo FR, Multari G, Lemmens RHMJ and Wulijarni-Spetjiptoed, Dyeandtannin producing plants: Plant Resources of SouthEast Asia. No. 3. Pudoc Wageningen. Netherlands, (1991)
7. Giambenedetti M andFederici E, Chemical fingerprinting of Lawsonia inermis L. using HPLC, HPTLC and densitometry, Phytochemical analysis, 19(2008).
8. Haddad Khodaparast, Mohammad Hosein and Dezashibi Zinab, Phenolic Compounds and Antioxidant Activity of Henna Leaves Extracts (Lawsonia Inermis),WorldJournal ofDairyandFoodSciences, 2(2007).
9. HaddadKhodaparast,MohammadHoseinandDez ashibi Zinab,Phenolic Compounds and Antioxidant Activity of Henna Leaves Extracts, World Journal of Dairy and Food Sciences, 2(2007)
10. Hikmat Ullah Jan, Zabta Khan Shinwari and Ashfaq Ali Khan, Staining Effect of Dye Extracted from Dry Leaves of lawsonia inermis on Angiospermic Stem Tissue, Pak. J. Bot, 43(2011).
11. Kathem K., Al-Rubiay, Nawres N, Jaber, AlMhaawe BH, Laith K.and Alrubaiy, Antimicrobial Efficacy of Henna Extracts,Oman Medical Journal, 23(2008).
12. Mehmet Emin, Zumrutdal,Mehmet Ozaslan,Mehmet Tuzcu, Mehmet Emin Kalender, Kenan Dagl?oglu,et.al, Effect of Lawsonia inermis treatment on micewith sarcoma,AfricanJournal ofBiotechnology, 7(2008).
13. Phirke, S. S., Saha, M. and Naresh Chandra, In vitro callus induction from leaf explants of Lawsonia inermis L. used as herbal dye, ASIAN J.EXP.BIOL.SCI.SPL(2010)
14. Razdan M.K. An introduction to plant tissue culture . Oxford and IBH Publishing Co. Pvt. Ltd. New Delhi(1983) 15. Williams R.O and OBE, Theusefuland ornamental plantsin Zanzibarand Pemba Zanzibar Protectorate, (1949).


|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License