IJCRR - 9(6), March, 2017
Pages: 08-14
Date of Publication: 31-Mar-2017
Print Article
Download XML Download PDF
BIODEGRADATION OF TANNERY EFFLUENT, ITS REUSE IN AGRICULTURE AND BIOCHEMICAL CHARACTERIZATION BY ELECTROPHORESIS
Author: C. M. Noorjahan, Sheeba Ali Siddiqui
Category: General Sciences
Abstract:Aim: Tannery industry is one of the important industry that pose environmental pollution through the disposal of its effluent. Hence this study was carried out to analyze physico chemical parameters of the effluent already treated by the industry (industry treated effluent) to degrade the pollutants of the effluent using fungi and finally to reuse the biotreated sample for the agriculture purpose. (ie) growth of vegetable plant Lycopersicum esculentum and growth parameters were studied. Biochemical characterization (ie) proteins and DNA present in the roots of Lycopersicum esculentum were also studied using electrophoresis.
Methodology: 5 litres of industry treated tannery effluent was collected in polythene containers from the tannery treatment plant situated in Chennai, Tamil Nadu India. The samples were collected for a period of 3 months from July 2015 to September 2015. The physico-chemical parameters were determined. Biotreatment of industry treated tannery effluent was carried out using native fungi Aspergillus niger and Rhizopus sp. Biotreated tannery effluent was used for growth of vegetable-tomato plant Lycopersicum esculentum for a period of 60 days and biochemical characterization such as protein and DNA of plant were studied by using electrophoresis.
Result: The results of physico-chemical characteristics of the industry treated tannery effluent has revealed that the effluent was brownish in colour with offensive odour, pH was alkaline in nature and has higher concentration of EC, BOD, COD, TSS and TDS though the effluent was treated by the industry which surpassed the permissible limits prescribed by CPCB (1995). The results of microbial analysis of the effluent revealed the occurence of 4 species of fungi in the industry treated tannery effluent namely, Aspergillus niger, Mucor sp, Pencilluim sp and Rhizopus sp. The results of analysis of degradation of the effluent showed that colour and odour of 100% treated sample has changed to colourless nature and odorless condition and also other pollutants were reduced using native fungi, Aspergillus niger and Rhizopus sp. The results of the plant growth showed that Lycopersicum esculentum was increased in bio-treated and control compared to industry treated sample.
Conclusion: The study revealed that native fungi, Aspergillus niger and Rhizopus sp plays a key role in the degradation of industry treated tannery effluent and the treated water can be utilized for agricultural purposes.
Keywords: Tannery effluent, Degradation, Aspergillus niger, Rhizopus sp, Lycopersicum esculentum, Molecular characterization
Full Text:
INTRODUCTION
Environmental pollution is the biggest menace to the human race on this planet today. The water of river and seas are being constantly polluted all over the world by various dangerous chemical and biological wastes. Mills and factories discharge very harmful waste water into rivers and seas. (Goyal, 2003).Tannery is one of the important industries causing water pollution. There are about 2,165 tannery industries in India. In Tamil Nadu alone, there are about 1,120 tanneries located in Vellore, Ranipet, Trichy, Dindigul, Erode and Pallavaram in Chennai. (Raniperumal and Singaram, 1996).Tannery effluent is the collection of water which was formed during various stages of processing of leather is collectively called as composite effluent. The tannery effluent is ranked as high pollutant among all other industrial wastes. (Eye and Lawrence, 1971).Tannery effluent induces health hazards to human and other aquatic organisms. It also makes the soil infertile, the ground and surface water turns to be unfit for irrigation and drinking. (Aruna U. Kakade, 2012). Disposal of such tannery waste with high pollution load into water courses or onto land, with or without prior treatment creates a great problem in the environment in the vicinity. So, it has become essential to treat the waste prior to its disposal. Thus the preliminary study was undertaken to analyse physico-chemical parameters of industry treated tannery effluent and to isolate and identify microbes of the effluent, native fungi Aspergillus niger and Rhizopus sp were used for the biotreatment of tannery effluent. Biotreated effluent was reused for agriculture (i.e) growth of vegetable plant Lycopersicum esculentum, growth parameters and biochemical characterization (i.e) proteins and DNA present in the roots of Lycopersicum esculentum were also studied using electrophoresis.
MATERIALS AND METHODS
Industry treated tannery effluent was used as the material in this study. The sample was collected in polythene containers (5 litres capacity) from the tannery treatment plant situated in Chennai, Tamil Nadu, India. Samples were brought to the laboratory and maintained at 25°C for further analysis. The samples were collected for a period of 3 months from July 2015 to September 2015. The physico-chemical parameters such as Colour, Odour, pH, Electrical Conductivity (EC), Total Suspended Solids (TSS), Total Dissolved Solids (TDS), Biochemical Oxygen Demand (BOD) and Chemical Oxygen Demand (COD), of industry treated tannery effluent were determined by following the Standard Methods outlined by APHA (1995).
Tannery effluent of about 1 litre was collected in sterile bottles and brought to the laboratory for the analysis of microbes (fungi) on the same day. Industry treated tannery effluent was diluted to 10-1 using sterile distilled water. 1 ml of diluted sample was cultured on Malt Extract Agar Medium (MEA) following pour plate method for fungal identification. Fungal species developed on the medium was observed. The fungal colonies grown on Malt Extract Agar Medium were sub-cultured on Potato Dextrose Agar (PDA) slants. Lactophenol cotton blue stain was used to identify the fungi by following the procedure of Onions et.al. (1981).
The seeds of vegetable - Tomato plant Lycopersicum esculentum were procured from a local nursery located in Chennai for the germination and growth in industry treated and biotreated tannery effluent.
Mycelial mats of native fungi, Aspergillus niger and Rhizopus sp. grown separately in liquid culture were recovered, washed with sterile distilled water and approximately 10 gms (fresh weight) mycelia of fungi were transferred to 100% industry treated tannery effluent in a conical flask separately. Conical flask with effluent and fungi both native Aspergillus niger and Rhizopus sp were incubated separately at 30 0.5oC for 96 hours on rotary shaker at 2000 rpm. After incubation the samples were centrifuged at 5000 rpm for 20 minutes. Control (conical flask with industry treated tannery effluent without fungus) was also run simultaneously. The procedure for degradation process was carried out by following the procedure of Krishna priya (2010). The supernatant were analysed for physico chemical parameters like pH, EC, Total Suspended Solids, Total Dissolved Solids, Biological Oxygen Demand, Chemical Oxygen Demand and heavy metals - chromium. Physico chemical parameters of the effluent were analysed before biotreatment (control) and after biotreatment by following the Standard procedure of APHA (1995).
0.5oC for 96 hours on rotary shaker at 2000 rpm. After incubation the samples were centrifuged at 5000 rpm for 20 minutes. Control (conical flask with industry treated tannery effluent without fungus) was also run simultaneously. The procedure for degradation process was carried out by following the procedure of Krishna priya (2010). The supernatant were analysed for physico chemical parameters like pH, EC, Total Suspended Solids, Total Dissolved Solids, Biological Oxygen Demand, Chemical Oxygen Demand and heavy metals - chromium. Physico chemical parameters of the effluent were analysed before biotreatment (control) and after biotreatment by following the Standard procedure of APHA (1995).
After degrading the industry treated tannery effluent using native fungi Aspergillus niger and Rhizopus sp for 96 hrs, the degraded water were reutilized for growth of vegetable-tomato plant Lycopersicum esculentum for a period of 60 days by following the procedure of Jerin (2011).
The seeds of Lycopersicum esculentum were washed with mercuric chloride solution for 2 minutes and then thoroughly washed in distilled water. Each earthen pot filled with farm yard manure was sown with 10 seeds allowed to germinate by irrigating with equal volume of industry treated tannery effluent and biotreated water. One set was irrigated with water as control. Five replicates were maintained for each sample (100%). Three trials (20th, 40th& 60thday) were made with an interval of 20thdays for each. For each trial, the vegetative features (i.e) root length, shoot length, no. of subroots, and number of leaves of the above plant were recorded.
After the growth of Lycopersicum esculentum (tomato) for 60 days in industry treated and biotreated samples, biochemical characterization such as protein and DNA were studied by using electrophoresis by following the procedure of Choi et.al.(2004).
RESULTS
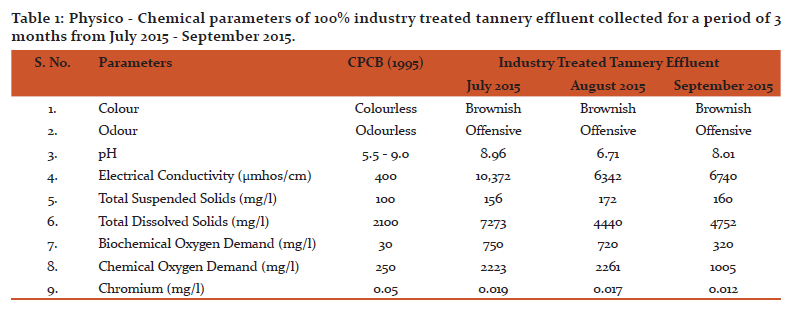
Result of the analysis of physico-chemical parameters of industry treated tannery effluent collected for a period of 3 months (July 2015 to September 2015) are depicted in Table 1.
Colour of the industry treated tannery effluent is brownish in colour. Odour of industry treated tannery effluent is offensive. The pH of industry treated tannery effluent has a minimum range of 6.71 (August 2015) and a maximum range of 8.96 (July 2015). The conductivity of the industry treated tannery effluent range between 6,342 µmhos/cm (August 2015) and 10,372 µmhos/cm (July 2015). The values of EC are higher than the permissible limits (400 µmhos/cm) of CPCB (1995). TSS level of Industry treated tannery effluent range from 156 mg/l (July 2015) to 172 mg/l August (2015) indicating that the TSS levels was higher than the permissible limits (100 mg/l) perscribed by CPCB (1995). TDS of Industry treated tannery effluent range between 4,440 mg/l (August 2015) and 7,273 mg/l (July 2015) the values of TDS found to be higher than the permissible limits (2,100 mg/l) of CPCB (1995). BOD levels of Industry treated tannery effluent has a maximum value of 750 mg/l (July 2015) and the minimum range of 320 mg/l (September 2015) which are higher than the permissible limits (30 mg/l) of CPCB (1995).COD levels of Industry treated tannery effluent ranges between 1005 mg/l (September 2015) and 2,261 mg/l (August 2015), the values are higher than the permissible limits (250 mg/l) of CPCB (1995). Chromium levels of effluent was within the permissible limits of CPCB (1995).
The result of isolation and identification of fungi present in the Industry treated tannery effluent are presented in Table 2. The results of the study revealed that 4 fungal species were isolated and identified from industry treated tannery effluent which include Aspergillus niger, Mucor sp, Pencillium sp and Rhizopus sp.
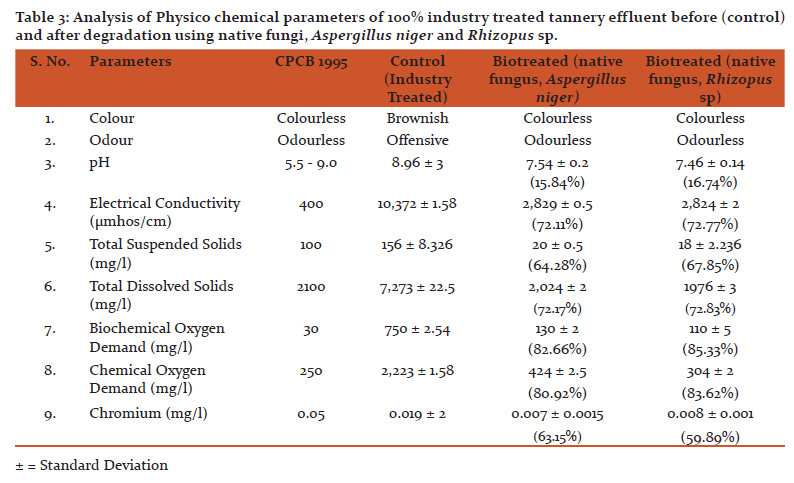
Result of analysis of degradation of Industry treated tannery effluent using native fungi (Aspergillus niger and Rhizopus sp.) are presented in Table 3.The results of the biodegradation study showed that Industry treated sample is brownish in colour before degradation but after degradation for 96 hrs using native fungi, Aspergillus niger and Rhizopus sp there is a change in colour from brownish to almost colourless nature of the sample. pH of industry treated tannery effluent was changed to almost neutral pH after biodegradation using fungi. TSS of Industry treated tannery effluent was reduced to a maximum percentage (67.85%) using Rhizopus sp and (64.28%) using Aspergillus niger. Maximum reduction of TDS (72.83%) was recorded using Rhizopus sp and Aspergillus niger (72.17%) after biodegradation. BOD level was reduced using Rhizopus sp (85.33%) and Aspergillus niger (82.66%) and COD level was reduced using native fungus, Rhizopus sp (83.62%) compared to other native fungus Aspergillus niger (80.92%). The level of heavy metal chromium was reduced after biodegradation for 96 hrs using native fungus, Aspergillus niger (63.15%) compared to that of native fungus, Rhizopus sp (59.89%).
From the present study, Aspergillus niger and Rhizopus sp, showed efficient degrading capabilities by degrading the contaminants of the effluent as they use it for their growth and reproduction. Hence after degradation of 100% industry treated tannery effluent, the treated water were reused for germination and growth of Tomato Plant Lycopersicum esculentum for a period of 60 days using 100% Industry treated and biotreated (native fungi, Aspergillus niger and Rhizopus sp) tannery samples.
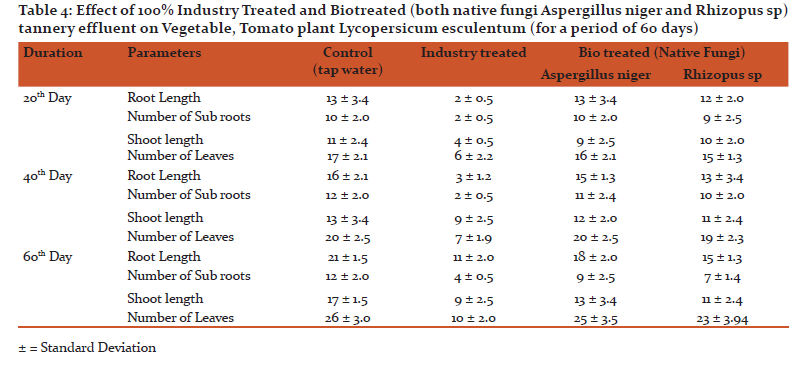
The results of the study (Table 4) showed that maximum germination and growth of plants were recorded when exposed to 100% biotreaed sample and control. The germination and growth of vegetable, Tomato plant Lycopersicum esculentum on 20th, 40th and 60th day were recorded their vegetative features such as shoot length, root length, number of sub-roots and number of leaves were measured and recorded.
Bio Chemical Characterization
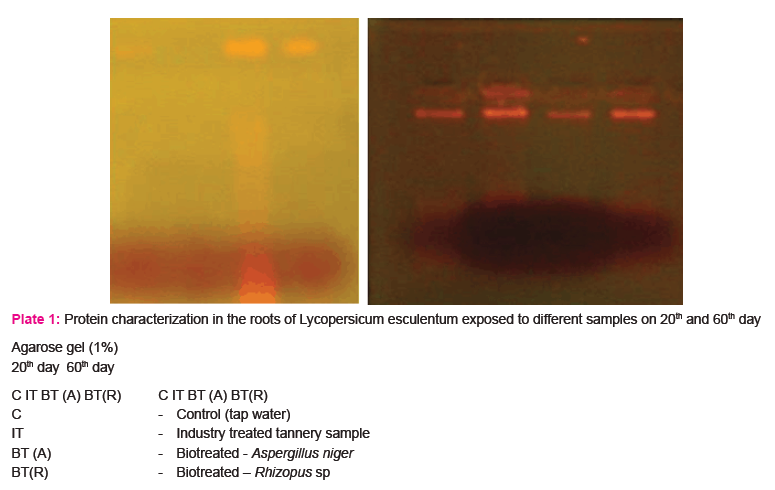
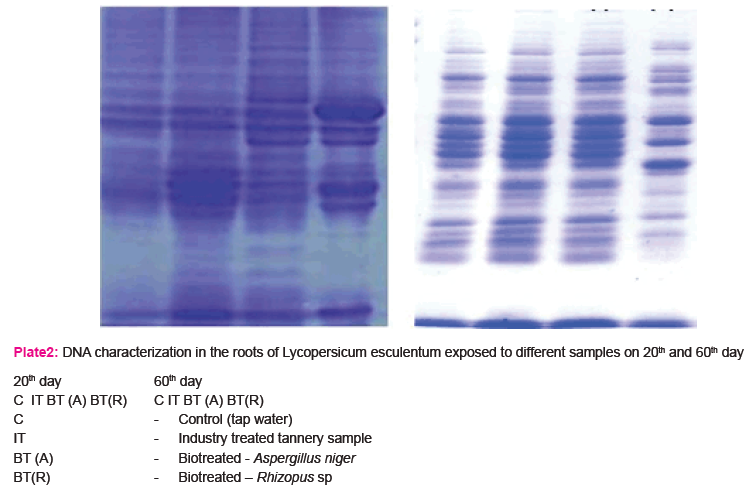
The study was further extended to carry out the biochemical characterization of vegetable tomato plant Lycopersicum esculentum. The results of characterization of the biochemical components (i.e) Proteins and DNA present in the roots of Tomato plant. Lycopersicum esculentum are presented in plate 1a & 1b.
Protein Characterization
The result of the study (Plate 1a) showed the formation of low, medium and high mobility bands indicating the presence of proteins of different molecular weight in the roots of Lycopersicum esculentum on 20th and 60th day when exposed to control (tap water), industry treated and biotreated tannery samples.
DNA Characterization
The results of the study (Plate - 1b) showed relative band intensities indicating the presence of damaged DNA in the roots of Lycopersicum esculentum exposed to industry treated sample, while band intensity in the roots of Lycopersicum esculentum exposed to biotreated sample resembles the bands of roots of control (tap water).
Seeds of Lycopersicum esculentum when treated with 100% industry treated and biotreated water showed interesting results. Germination and growth of seeds - no of leaves, root length, shoot length and no of subroots of Lycopersium esculentum in 100% industry treated sample on 20th, 40th and 60th days were decreased. Whereas maximum germination and growth of plants were recorded that exposed to 100% biotreated sample like control.
The biochemical characterization of protein and DNA were studied in the roots of Lycopersicum esculentum after 20th and 60th days of exposure to tannery samples. The results showed that the low, medium and high mobility bands were observed in all the samples indicating the presence of proteins of different molecular weight which is due to the effect of heavy metals and hazardous materials present in the industry treated effluent. The presence and absence of protein separation varied in different samples. (Choi et.al., 2004).
DNA isolated from roots of Lycopersicum esculentum on 20th and 60th day of growth showed relative band intensities results in the presence of damaged DNA on exposure to heavy metals and other toxic substances in the case of industry treated tannery effluent, while in the biotreated sample, band intensity resembles the control (tap water) which may be due to absence or less amount of toxic substances present in the treated samples. (Choi et.al., 2004).
DISCUSSION
This colour and odour could be due to decomposition of organic or inorganic matter (Singh et.al.,1998). Coloured effluent when discarded to land surface negatively affects the soil fertile capacity (Chhonkar et.al., 2000). pH of the effluent was alkaline in nature which affects the physico chemical properties of receiving water which in turn adversely affects the aquatic life and human beings. This also changes soil permeability which results in polluting underground resources of water. (Krishna Priya 2010). Industry treated effluent has high level of Electrical conductivity which gives the measure of water conductivity as well as the indication of the level of inorganic constituents in water. (Ramamurthy et.al., 2011). High amount of TSS found in the effluent may have adverse effects on aquatic flora and fauna and reduce the diversity of life in aquatic system and promote depletion of oxygen and sliting in ponds during rainy season (Goel, 1997). High levels of Total Dissolved Solids (TDS) of the industry treated effluent are one of the major sources of sediments which reduce the light penetration into water and ultimately decrease the photosynthesis process of aquatic flora. (Ramamurthy et.al., 2011). The present study has revealed high levels of BOD in the industry treated tannery effluent indicating high organic load. Further high COD may be due to high amount of inorganic compounds which are affected by the bacterial decomposition (Kulkarni 1992).
Tannery effluent is rich in organic and inorganic nutrients which would have supported the growth of fungal population, Sekar (2011) also reported the presence of various fungi in tannery effluent, which contains high organic load. Sukumaran et.al., (2008) identified 10 species of fungi in tannery effluent. Kaushik (2011) reported the presence of 2 fungal species in Direct Dye effluent. Ramamurthy et.al. (2011) reported the presence of 6 species of fungi in textile effluent. After biodegradation of sample using fungi following changes occurred color changed from brownish to almost colourless nature this may be due to the action of microbes - Aspergillus niger and Rhizopus sp, which decomposed the toxic pollutants present in the effluent and made the change in colour and odour of the effluent (Krishna priya, 2010). Shafiquzzaman Siddiquee et.al. (2015) removed heavy metal contaminants from wastewater using the potential filamentous fungi biomass. Ihsan flayyih Hassan et.al. (2015) studied efficiency of some filamentous fungi for treatment of refinery wastewaters and suggested that fungi helps in reduction of refinery contaminants from the effluent. pH levels decreased in biotreated samples which may be due to accumulation of organic acids and also indicating the efficiency of the microbes to biodegrade the effluent (Ramamurthy et.al.,2011). The reduction in TSS and TDS value may be due to the action of microbes – Rhizopus sp and Aspergillus niger, which decomposed the toxic pollutants present in the effluent and made the change in colour, odour, TSS and TDS of the effluent (Krishna priya 2010). BOD and COD levels were reduced after biotreatment thereby indicating the degrading efficiency of native fungi, Aspergillus niger and Rhizopus sp (Kulkarni 1992). Chromium levels were reduced in effluent Removal of chromium by Penicillium chrysogenum in tannery effluent was recorded to be high when compared to Aspergillus niger (Jayanthi et.al., 2014).
The presence of toxic substances present in waste water has decreased the growth of Lycopericum esculentum exposed to 100% industry treated samples. Whereas increased rate of germination and growth of Lycopersicum esculentum in 100% biotreated sample is due to the maximum removal of toxic substances by native Fungi, Aspergillus niger and Rhizopus sp. The untreated tannery effluent showed decrease in the seed germination percentage of Lycopericum esculentum with increasing the concentration in all treated samples as well as higher levels of effluent accumulation cause inhibition of seed germination and seedling growth at lower concentration. (Mandakini et.al., 2016).
CONCLUSION
Thus it may be concluded from the above study that industry tannery effluent with high pollutants can be reduced by using native fungi, Aspergillus niger and Rhizopus sp both the native fungi has the same degrading efficiency of the effluent and as evidenced in the present work this biotreated tannery effluent can be utilized for agricultural purpose.
ACKNOWLEDGEMENT
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.

References:
- APHA. Standard methods for the examination of water and waste water. American public Health Association, Washington, D.C.1995; 17thed.
- Aruna U Kakade. Biodegradation of tannery effluent by using tannery effluent isolate. International Multiple disciplinary Reasearch Journal 2012; 2(3): 2231 – 6302.
- Chhonkar PK, Datta SP, Joshi HC, Pathak H. Impact of industrial effluents on soil health and agriculture - Indian experience: Part II - Tannery and textile industrial effluents. J. Sci. Ind. Res 2000;59: 446 - 454.
- Choi NS, Yoo KH, Yoon KS, Maeng PJ, Kim SH. Nano-scale proteomics approach using two-dimensional fibrin zymography combined with fluorescent SYPRO ruby dye. J BiochemMolBiol 2004; 37: 298-303.
- CPCB. Pollution control, acts, rules and modifications issued their under Central Pollution Control Board, New Delhi 1995.
- Eye JD, and Lawrence L. Treatment of waste water from a sole leather industry. Journal of water Pollution control, Fed 1971;43 : 2291 - 2302.
- Goel PK. Water pollution, causes, effects and control. New Age International (P) Ltd., publishers, New Delhi 1997;269.
- Goyal RR, Mohan MK, Kansal A. Treatment of dyeing and printing effluent using photo assisted fenton reaction. Nature Environmental pollution technology 2003; 2(2): 179 - 182.
- Ihsan Flayyih Hasan AI-Jawhari, Noor Jabar Mhail, Saher Abed AI-Rutha Ali. Efficiency of some filamentous fungi to treatment of effluent petroleum wastewaters from refinery.Int.J.Curr.Microbiol.App.Sci 2015;4(4): 625-641.
- Jayanthi M, Kanchana D, Saranraj P, Sujitha D. Bioadsorption of Chromium by Penicillium chrysogenum and Aspergillus niger isolated from tannery effluent. Intl. J. Microbiol. Res 2014; 5 (1): 40-47.
- Jerin S. Isolation of microbes treatment of flavour effluent using native fungus, Aspergillus sp and reuse of biotreated water for germination and growth of ornamental plant, chrysanthemum sp. B.Sc. Dissertation, University of Madras, Chennai 2011.
- Kaushik CP, Sharma JK. Decolourisation studies in direct dyes effluent using fungi, Phanerochacte chrysosporium and Aspergillus niger, Indian Journal of environmental protection 2011;31: 129 - 132.
- Krishna Priya E. Biodegradation of Tannery Effluent using native fungus, Penicillium sp. B.Sc. Dissertation, University of Madras, Chennai 2010.
- Kulkarni TT. Source and characteristics of dairy wastes from a medium size effluent on microorganisms, plant growth and their microbial change. Life science of Advance 1992;3 : 26 - 28.
- Mandakini Magre R, Khillare YK. Effect of tannery industry effluent on germination and growth of tomato plant. World Journal of Pharmacy and Pharmaceutical Sciences 2016;5(2):1064-1074.
- Onions AHS, Allsopp D, Egginns MOW. Smith's introduction to industrial mycology, Edward Arnold publishers 1981;(7ed).
- Ramamurthy N, Balasarawathy S, Sivasathivelan P. Bio degradation and physico chemical changes of textile effluent by various fungal species, Journal of Biophysics 2011;112-123.
- Rani Perumal P, Singaram. Chemical of Soil and irrigation water by tannery effluent, Tamilnadu experience. Indian Journal of Research, Agri Chemi 1996; 1: 1-8.
- Sekar P. Efficacy of microbes in Bioremediation of tannery effluent. International journal of current research 2011;3(4):324 - 326.
- Shafiquzzaman Siddiquee, Kobun Rovina, Sujjat Al Azad, Laila Naher, Saallah Suryani, Pasicha Chaikaew. Heavy metal contaminants removal from wastewater using the potential filamentous fungi biomass: a review. J Microb Biochem Technol 2015;7:384-393.
- Singh SM, Varshneya I, Nagarkoti M. Assessment of physico chemical parameters of effluents of three factories of bareilly district and their possible effects on grazing animals and cereals. J. Environ. Biol 1998;19(3): 271-274.
- Sukumaran M, Ramamurthy, Raveendran S, Solidharan. Biodiversity of microbes in the tannery effluent, Journal of Ecotoxicology, Environmental monit 2008; 18(4) : 313-318.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License