IJCRR - 8(23), December, 2016
Pages: 26-28
Print Article
Download XML Download PDF
KNOWLEDGE, ATTITUDE AND PRACTICE EVALUATIONOF ADVERSE DRUG REACTIONS REPORTING AMONG HEALTH CARE PROFESSIONALS IN KIMS HOSPITAL, NARKETPALLY, TELANGANA, INDIA
Author: C. Dinesh M. Naidu, Anand Vardhan, Mangesh Bankar, Vinay Singh Raghuvanshi, Sagar Sharma
Category: Healthcare
Abstract:A huge number of cases of morbidity and mortality worldwide are attributed to Adverse Drug Reactions (ADRs), therefore this study was done with the primary Objective of assessing the Knowledge regarding pharmacovigilance and to evaluate the impact of an educational intervention on the same among healthcare professionals in a tertiary care centre.
Methods: A questionnaire containing 20 questions was given to all the participants before the educational session. Later, an interactive educational session on pharmacovigilance was conducted for them. After the educational session, the same questionnaire was given to all participants. The impact of effectiveness of educational session among health care professionals was evaluated by statistical analysis. 80 health care professionals were involved in the study. 37 Post Graduates of different departments, Assistant Professors 14, senior residents 10, Associate Professors 15, and 4 Professors were participants of this study.
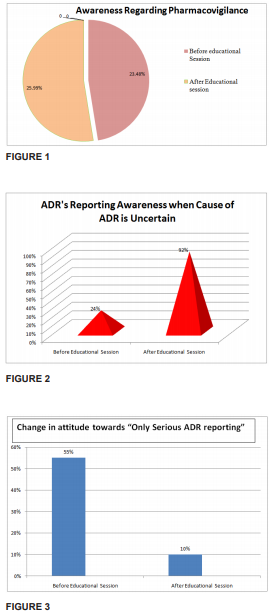
Results: The overall response rates during pre intervention was 23.48%, which significantly increased to 25.99% during post intervention (P value < 0.0001). 80% of the health care professionals admitted that awareness amongst healthcare providers would help to improve the pharmacovigilance programme.
Conclusion: Educational session in this study has shown significant improvement in the awareness about pharmacovigilance among health care professionals.
Keywords: Pharmacovigilance, Educational Intervention, Adverse Drug Reactions, Spontaneous Reporting of ADR’s
Full Text:
INTRODUCTION
Adverse drug reactions (ADRs) are the cause of morbidity and mortality worldwide. Recently it has been shown that ADRs are the fourth major cause of death in the United States.[1] According to World Health Organization (WHO) definition, an Adverse drug reaction is any unintended, and undesired effect of a drug, which occurs at doses used in humans for prophylaxis, diagnosis, or therapy.[2] Therefore to identify the drugs which cause number of ADRs, pharmacovigilance programs have been initiated in several countries in the recent past. There are variation in drug response among individuals, prescription pattern, drug regulatory system, drugs availability, it is recommended for every country to set up their own pharmacovigilance programme.[3]
Spontaneous reporting of ADR’s are a major source of information in pharmacovigilance. Spontaneous reporting of ADR’s are responsible for preventing the occurrence of tragedies caused by new medicines and also improving the safety labeling of pharmaceutical products. However, spontaneous reporting schemes are very less in number.[4]The success of any pharmacovigilance program in any country depends upon the involvement of the healthcare professionals who participate in reporting the ADRs.
Pharmacovigilance is still a young branch in India and there is very limited knowledge about this discipline. The Indian national Pharmacovigilance Programme is suffering setbacks due to lack of awareness and inadequate training about drug safety monitoring among healthcare professionals in India.[5]Different studies done in this field indicate inadequate knowledge about pharmacovigilance among healthcare professionals as well as attitudes that are associated with underreporting. Although Pharmacovigilance programs are becoming successful in improving drug use patterns, under-reporting of ADRs is still a major problem.[6]
Assessing the level of awareness of Pharmacovigilance among the healthcare professionals is extremely important, Therefore this study was conducted to assess awareness of pharmacovigilance among the healthcare professionals and to evaluate the impact of an educational session for improving awareness of pharmacovigilanceamong Physician, and post graduate residents in Kameini institute of Medical Sciences, Narketpally, Telangana.
MATERIALS AND METHODS
A Cross sectional study was conducted in Kameini institute of Medical Sciences, Narketpally, Telangana in June 2015 over a period of 1 week where All the participants were given a questionnaire as pre-test and the same question as post-test was given after one week of integrated educational session. The questionnaire consisted of information about knowledge and practice of ADR reporting and how frequently they are encountering the ADR’s, factors affecting their reporting of ADR and suggestions towards how a better ADR reporting can be achieved. The collected thus data was analysed statistically using chi-square test and p value was obtained.
RESULTS
Out of 80 health care professionals in this study, 37 participants were post Graduates, 14 were Assistant Professors,10 were senior residents,15 were Associate Professors , and 4 were Professors of different clinical departments were the enrolled. The overall response rates before the educational session(Pre Test) was 23.48%, which significantly increased to 25.99% after one week of educational session(Post Test) and the p value came out to be <0.001 which is statistically significant. (Figure: 1). 12% of the participants were of the opinion that only ADRs to new drugs should be reported, that significantly reduced to 5.5% after one week of educational session was over. 24% of the participants were positive about reporting the ADRs even when cause is uncertain, which increased significantly to 92% after one week educational session (Figure: 2). 55% of the participants were of opinion to report only serious ADRs, that reduced to 10 % after educational session (Figure: 3). Only 41% of the health care professionals agreed to have encountered ADRs in their practice frequently.
DISCUSSION
The present study was questionnaire-based, which included the prescribers of every level including Post Graduates to Professors. This study showed that although the right attitude for ADR reporting existed among most prescribers, the actual practice of ADR reporting was lacking. The overall response rates increased after one week of educational session from 23.48% to 25.99% which was also statistically significant p value <0.001. somewhat similar results were seen in a study done by Desai et al where the response rate is about 38% thus showing that educating the prescribers regarding ADR reporting is helpful in increasing awareness amongst health care providers.[7]
In this study 41% of health care providers agreed to have encountered ADR’s and were aware of Pharmacovigilance programme of India and National Pharmacovigilance Centre Location
But after one week educational session 100% of health care professionals were aware of this aspect, which is similar to a study conducted by study by Fadare et al where 44.6% of the participants were aware of the existing pharmacovigilance.[8]
Poor reporting of ADR’s in our study can be attributed to lack of knowledge about ADR reporting forms, who all can report ADR’s, type of reactions to be reported, whether serious ADR’s to be reported only, similar to the study conducted by Chatterjee et al, according to which the reason for under reporting of ADRs was the negligibility of the adverse reaction due to less time and little knowledge about the types of reactions to be preferentially reported.[9] Similarly, In a study conducted by Khalili H et al, where 21% of the participants were of the opinion not to report well known ADRs which reduced to 16% following educational intervention. [10]
The present study suggest that if such periodic awareness programs are held for the healthcare professionals, ADR monitoring programme and the ADR reporting procedure would be much better. Encouragement of the health care professionals to actively participate in the ADR reporting should also be done periodically.
CONCLUSION
This study conducted at KIMS Hospital Narketpally concludes that the health care providers were better in attitude and practice but had a limited knowledge ADRs and Pharmacovigilance. Only a few professionals had ever reported an ADR to the Pharmacovigilance center. The study suggests a huge scope for improving the ongoing Pharmacovigilance activities in the hospital. There should be continuous educational sessions for the doctors, post graduates, nurses, and the medical students as they are the pillars of Healthcare System.
Source of Funding- NIL
Conflicts of Interest- None
ABBREVIATIONS
ADR’s – Adverse Drug Reactions
KIMS – Kamineni Institute of Medical Sciences
References:
- Somayeh H, Hassan T, Hayatshahi A, Gholami K, Javadi M. Knowledge, attitudes and practice of nurse regarding adverse drug reaction reporting. Iran J Nurs Midwifery Res, 17(1), 2012, 21–25.
- World Health Organization. Safety of Medicines. A guide to detecting and reporting adverse drug reactions. Geneva, Switzerland: World Health Organization 2002. WHO / EDM / QSM /2002.2
- Safety monitoring of medicinal products. The importance of pharmacovigilance. World Health Organization, Geneva, 2002. (http://apps.who.int/medicinedocs/pdf/s4893e/s4893e.pdf.)
- Mishra H, Kumar V. Pharmacovigilance: Current Scenario in a Tertiary Care Teaching Medical College in North India. J Pharmacovigilance, 1, 2013, 108.
- Rajesh R, Vidyasagar S, Varma DM An Educational Intervention to assess Knowledge Attitude Practice of pharmacovigilance among Health care professionals in an Indian tertiary care teaching hospital. Int J PharmTech Res, 3(2), 2011, 678-92.
- Palaian S, Ibrahim MI, Mishra P. Health professionals' knowledge, attitude and practices towards pharmacovigilance in Nepal. Pharmacy Practice (Internet), 9(4), 2011, 228-35.
- Desai CK, Iyer G, Panchal J, Shah S, Dikshith RK et al. An evaluation of knowledge, attitude, and practice of adverse drug reaction reporting among prescribers at a tertiary care hospital. PerspectClin Res, 2(4), 2011, 129–36.
- Fadare JO, Enwere OO, Afolabi AO et al. Knowledge, Attitudeand Practice of Adverse Drug Reaction Reporting among Healthcare Workers in aTertiary Centre in Northern Nigeria. Trop J Pharm Res, 10 (3), 2011, 235-42.
- Chatterjee S, Lyle N, Ghosh S: A survey of the knowledge, attitude and practice of adverse drug reaction reporting by clinicians in eastern India. Drug Saf, 29, 2006, 641-2.
- Khalili H, Mohebbi N, Hendoiee N, Keshtkar AA, Khavidaki SD . Improvement of knowledge, attitude and perception of healthcare workers about ADR, a pre- and post-clinical pharmacists’ interventional study. BMJ Open, 2, 2012, e000367.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License