IJCRR - 5(6), March, 2013
Pages: 46-50
Date of Publication: 30-Mar-2013
Print Article
Download XML Download PDF
EFFECT OF ANTI-PROGESTROGEN ON THE EPITHELIAL CELL HEIGHT OF RAT ENDOMETRIUM
Author: Khadija Qamar, Samina Anjum, Zarmina Saga
Category: Healthcare
Abstract:Objective: To investigates the effect of mifepristone on height of the epithelial cells of the endometrium. Study Design: Laboratory based randomized controlled trials Place & Duration of the study: Department of Anatomy, Army Medical College Rawalpindi from Jan. 2007-march 2007. Method: Sixty adult female rats were divided randomly into two groups, comprising of 30 animals in each group. In group A one ml of normal saline was given orally daily for three months while in group B mifepristone was given orally in a dose of 1 mg/kg body weight daily for three months. Results: In the experimental group reduction was observed in epithelial cell height. Significantly lower level of progesterone while higher level of estrogen level in experimental group was noted as compared to the control group. Conclusion: Long term mifepristone administration suppresses the endometrial proliferation and epithelium height was decreased. It also lowered the plasma concentration of progesterone. While the plasma concentration of the estrogen was raised
Keywords: Mifepristone, estrogen, progesterone, epithelium
Full Text:
INTRODUCTION
The progesterone and estrogen hormones induce proliferation and differentiation in the endometrium1 . The inner mucosa, or endometrium, consists of a surface columnar epithelium overlying a thick lamina propria containing numerous blood vessels and endometrial glands2 . Mifepristone acts on the receptors just like the progesterone but may produce different conformational changes. 3With the rising trend of the use of antiprogestin and insufficient knowledge regarding its effects on histomorphology of uterus, this study has been designed to see the long term effects of mifepristone treatment on rat endometrium.
MATERIAL AND METHODS
These laboratory based randomized controlled trials were conducted at the department of Anatomy, Army Medical College Rawalpindi from Jan. 2007-march 2007. Sixty healthy adult female Sprague Dawley rats weighing 200-300 g were procured from the National Institute of Health Sciences Islamabad. The animals were randomly divided into two groups of 30 each.
Group A (Control)
Thirty female rats were given one ml of normal saline orally daily for three months.
Group B (Experimental)
Thirty female rats were given the drug (Mifepristone) orally in a dose of 1 mg/kg body weight daily for three months. All the animals were sacrificed next day after the last oral dose. Two ml blood was taken directly from the heart for measurement of estrogen and progesterone levels. Uterine horns along with a portion of vagina was removed, trimmed and placed into 10% Formalin for 24 hours. About ½ cm piece of tissue was taken from the middle of the right uterine horn. Approx five microns thick sections were cut stained with hematoxylin and eosin for light microscopic study.
MICROSCOPIC OBSERVATIONS
Height of Epithelium
The height of the epithelium was taken from the basement membrane up to the upper limit of the cell facing the lumen under high power field (x40 objective) from three regions and their mean was taken as the reading for that animal.
Statistical analysis
Data had been analyzed using SPSS version 15. Descriptive statistics were used to describe the data. P-value < 0.05 was considered as significant.
RESULT
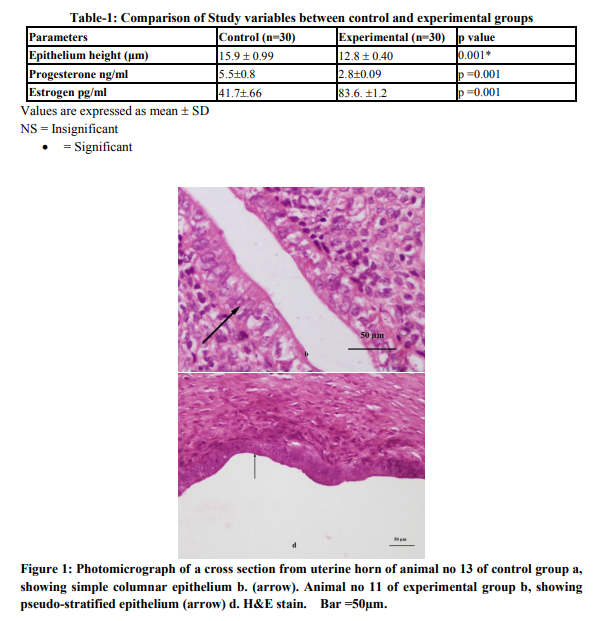
Total sixty animals were included in the study, 30 in each group. The tubular sections showed three distinct layers (inner, middle and outer). In control group the luminal side of inner layer (Endometrium) was lined by single regular row of cylindrical cells. These closely packed cells having rectangular outlines had round ovoid or elongated nuclei. Their nuclei tended to be aligned at the center of the cells and the cytoplasm was basophilic reflecting the microscopic picture of simple columnar epithelium (Fig. 1b). In experimental group the inner layer was folded giving it an overall ruffled appearance as a result lumen was much reduced as compared to normal group .The epithelium appeared pseudostratified with decrease in height as compared with control group (Fig. 1d). The mean epithelium height in experimental group was 12.8±0.40 μm which was lower than that of the control group. The difference was regarded highly significant statistically (p =0.001) when compared with the control group. Average progesterone level was significantly lower in experimental group as compared to control group (p = 0.001) while average estrogen level was significantly higher in experimental group as compared to control group (p = 0.001). (Table-1)
DISCUSSION
In this study of 3 months we have studied the long term effects of mifepristone treatment on rat endometrium. Uterine epithelium height was decreased in the experimental group after administration of mifepristone compared with control animals. This may be due to effect of drug on the uterus. It has also been reported by others that mifepristone blocks estrogen-induced increase in uterine growth inhibits true uterine growth. 25 hrs, following an injection of progesterone, a significant increase in the height of cells of the luminal epithelium is observed4 . Our data indicated decrease in the height of the cells of the luminal epithelium after mifepristone treatment. Changes caused in the luminal epithelium in response to antiprogesterone are usually attributed to the direct affect of the hormone. 5 Most of the glandular epithelial cells had sub nuclear vacuoles in various studies. Ultra structurally there is noted the presence of large vacuoles, membrane inclusions and myelin like bodies in the endometrial taken from treated animals, features usually suggestive of the disintegration of cytoplasm proteins.6 In treated animals, vacuolization is one of the structural indicators of energy deficit and permeability disorder of membranes in the endometria7 . This hypothesis, that protein synthesis is proceeding at a faster rate in these cells in response to progesterone, is thus consistent with the morphometric data 8 . Endometrial cells require a balance between estrogen and progesterone production. The absence of progesterone removes the progesterone brake leading to persistent estrogenicity and constant endometrial proliferation. The endometrium can become disordered, although the ratio of stroma to glands remains normal, and vascular abnormalities such as dilated capillaries become apparent. It is unlikely to be solely due to the effect of high levels of unopposed estrogen because it occurred in some women, e.g. in Shanghai where there were low estrogen levels with complete suppression of ovarian follicular development9 In a study on ovariectomized rhesus macaques were treated for 5 months with either estradiol (E2) alone, E2 + progesterone (two doses) or E2 + ZK (0.01, 0.05 or 0.25 mg/kg) all doses of ZK blocked endometrial proliferation and induced endometrial atrophy. 10The absence of progesterone removes the ‘progesterone brake’, leading to persistent estrogenicity and constant endometrial proliferation., Though usually there is normal ratio of stroma to glands, vascular abnormalities such as dilated capillaries become apparent and the endometrium can become disordered11 . Rats receiving long-term PR antagonist treatment show endometrial stimulation under the effect of estrogen12. High doses of mifepristone (25 and 50 mg/d) lead to variable effects in women, such as atypical cystic changes, as have been described in eutopic endometrium13. Serum progesterone levels declined in experimental groups after mifepristone administration. Level of estrogen hormone was elevated in the experimental group as compared with the control group. Less congruency has been found in the earlier reports about the effect of mifepristone on progesterone secretion. 14 No statistically significant change in the progesterone levels was observed in the 2-day follow-up using 200 mg of mifepristone 15. With administration of 600 mg mifepristone, there was observed an increase in progesterone levels on day 1 followed by a significant decrease in another study16
CONCLUSION
We concluded that in long term mifepristone affected the endometrial proliferation and induced histomorphological changes in the uteri of the experimental rats. Significant reduction in epithelial height was observed in epithelium height. It also lowered the plasma concentration of progesterone .While the plasma concentration of the estrogen was raised.
ACKNOWLEDGEMENTS
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Connelly OM. Female Steroid Hormone Action. Endocrinology 2001; 142(6): 2194- 99.
2. Benakanakere C, Besch-Williford M, Ellersieck R, Hyder SM. Regression of progestin-accelerated7, 12-dimethylbenz[a] anthracene-induced mammary tumors in Sprague-Dawley rats by p53 reactivation and induction of massive apoptosis: a pilot study. Endocr Relat Cancer March 1, 2009; 16(1): 85-98.
3. Antoniou G, Kalogirou D, Karakitsos P.Transdermal estrogen with a levonorgestrel- releasing intrauterine device for climacteric complaints versus estradiolreleasing vaginal ring with a vaginal progesterone suppository. Clinical and endometrial responses. Maturitas. 1997; 26: 103-11.
4. De Vivo I, Hankinson SE, Colditz GA, Hunter DJ. A functional polymorphism in the progesterone receptor gene is associated with an increase in breast cancer risk. Cancer Res 2003; 63: 5236–8.
5. Gopalkrishnan K, Katkam RR, Sachdeva G, KholKute SD, Padwal V, Puri CP. Effects of an Antiprogestin Onapristone on the Endometrium of Bonnet Monkeys: Morphometric and Ultra Structural Studies. Biol Reprod 1959; 68(6): 779-87.
6. Brenner RM, Slayden OD. Estrogen action in the endometrium and oviduct of rhesus monkeys during RU 486 treatment. In: Beier HM, Spitz IM, editors. Progesterone antagonists in reproductive medicine and oncology. Cary, NC: Oxford University Press; 1994; p.82-97.
7. Jeffrey R. Goldberg, MD; Marcus G. Plescia, MD, MPH; Geraldine D. Anastasio. Mifepristone (RU 486) Current Knowledge and Future Prospects. Arch Fam Med. 1998; 7:219-222.
8. Van Look PFA, Ven herizen H. Post ovulatory methods of fertility regulation: the emergence of antiprogestens. In: Van Look PFA, Perez – Polacies G, editors. Contraceptive research and development. New Delhi: Oxford University Press; 1994; P. 151-201.
9. Baird DT, Glasier AF. Science, medicine and the future: contraception. Br Med J 1999; 319: 969-72.
10. Hargrove JT, Maxson WS, Wentz AC, Burnett LS. Menopausal hormone replacement therapy with continuous daily oral micronized estradiol and progesterone. Obstet Gynecol. 1989; 73: 606-12.
11. Maxson WS. The use of progesterone in the treatment of PMS. Clin Obstet Gynecol. 1987; 30: 465-77.
12. Schaison G, George M, Lestrat N, Reinberg A, Baulieu EE. Effects of the antiprogesterone steroids RU 486 during midluteal phase in normal woman. J Clin Endocrinol Metab 1985; 61: 480-9.
13. Murphy AA, Castellano PZ. RU 486: pharmacology and potential use in the treatment of endometriosis and leiomyomata uteri. Curr Opin obstet Gynecol 1994; 6(3): 269-78.
15. O.D. Slayden, M.B. Zelinski, K. Chwalisz, H. Hess-Stumpp, R.M. Brenner Chronic progesterone antagonist–estradiol therapy suppresses breakthrough bleeding and endometrial proliferation in a menopausal macaque model .Hum. Reprod. (2006) 21(12): 3081-3090
16. Chwalisz K, Hsiu JG, Williams RT, Hodgen GD. Evaluation of the antiproliferative actions of the progesterone antagonist’s Mifepristone and onapristone on primate endometruim. Proceeding of the 39th Annual Meeting of the society for Gynecologic Investigation; Texas, USA. Mar 1992; p12-8.
17. S. Schäfer-Somi, O.A. Aksoy, H.B. Beceriklisoy, A. Einspanier, H.O. Hoppen S. Aslan Repeated induction of abortion in bitches and the effect on plasma concentrations of relaxin, progesterone and estradiol-17β. 2007;68: 889-895
18. Ishwad PC, Katkam RR, Hinduja IN, Chwalisz K, Elger W, Puri CP. Treatment with a progesterone antagonist ZR>98.299 delays endometrial development without blocking ovulation in bonnet monkeys. Contraception 1993; 48: 57-70.
19. Sitruk-Ware R. Approval of Mifepristone (RU486) in Europe. Zentraibl Gynakol 2000; 122(5): 241-7.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License