IJCRR - 5(2), January, 2013
Pages: 135-139
Date of Publication: 26-Jan-2013
Print Article
Download XML Download PDF
EVALUATION OF ANTIOXIDANT LEVEL AND TRACE METALS IN ANEMIA
Author: Naila Masood, Imran Ali Shaikh, Ali Raza Memon, Saira Baloch
Category: Healthcare
Abstract:Background: Anemia is a disorder characterized by reduction in hemoglobin concentration of blood below the normal level.
It is a global problem, mainly affecting poor people in developing countries.
Methods: to determine trace metals fifty intravenous blood samples anemic patients and normal subjects were collected and immediately centrifuged to obtain the supernatant liquid, serum of both the
groups for analysis.
Results: The mean serum levels of magnesium, zinc, copper, and iron and antioxidant activity in anemic patients were 16.05\?1.14ppm, 4.33\?0.28 ppm, 2.5\?0.2ppm,
1.50\?0.28ppm and 1.27\?0.05ppm respectively. Serum magnesium, zinc, iron and antioxidant levels
were significantly lower whereas, the higher level of Copper was observed in anemic patients
compared to the controls.
Conclusion: Supplementation with deficient trace elements may reduce the
severity of symptoms and complications associated with anemia.
Keywords: Anemia, Iron, Zinc, Copper, Magnesium, Antioxidant Activity.
Full Text:
INTRODUCTION
Anemia is one of the world’s most prevalent health problems, especially among adult and pregnant females approximately 35% to 75% (56% on average) are anemic in developing countries (1). “The World Health Organization has identified that iron and folic acid deficiency is a severe problem in Pakistan and it has devastating impacts on health and survival. It has been estimated that iron deficiency leads to 22,000 maternal deaths for the last 10 years (2). There are various other nutritional and communicable causes of anemia, iron (Fe) deficiency is often a causative aspect in many of these cases (3). Fe deficiency anemia leads to weakness (4), poor physical growth (5), and delayed psychomotor development (6). A recent study on the prevalence and etiology of nutritional anemia in urban areas of Hyderabad, Pakistan indicated a high prevalence of anemia and Fe deficiency in 60% of adults (7). Zinc (Zn) deficiency in humans has been found in infants, schoolchildren and adults (8). Zinc deficiency associates with Fe deficiency anemia in areas where Fe deficiency is a problem (9). It was investigated that anemia is one, of the major manifestations of copper (Cu) deficiency both in animals and human beings (10). It is essential to maintain Fe homeostasis in the human body; its deficiency leads to anemia and neutropenia (11). Cuassisted enzymes are necessary for the utilization of Fe to make hemoglobin, a main component of red blood cells (11). Recent attention has been directed to the element chromium (Cr); it can improve insulin sensitivity and therefore may be involved in carbohydrate and lipid metabolism (12). The determination of trace quantities of metals in biological samples requires the use of sensitive and selective techniques such as atomic absorption spectrometry. In this study, we evaluate whether Fe, Cu, Mg and Zn deficiency is related to increased level of toxic metals in blood samples of anemic and controls of both gender with age range 16– 50 years. The samples were prepared method. Fe, Cu, Mg and Zn concentrations in samples under study were evaluate by atomic absorption spectrometer, while antioxidant activity was analyzed by spectrophotometer. In this study, we aimed to evaluate the levels of Cu, Mg, Zn, and Fe and antioxidant activity in serum of anemic patients.
MATERIAL AND METHODS
This investigation has been conducted among three medical units of Liaquat University of Medical and Health Sciences (LUMHS) Jamshoro. The group of patients were been selected within the age range of 16-50years of both gender. The metals copper, iron, magnesium, and zinc in the blood serum were determined by Atomic Absorption Spectrometry (AAS) (Model, A-20 Varian). Whereas anti oxidative activity was measured as a ratio of Fe (II)/Fe (III) present in the blood serum. Iron (II) was determined spectrophotometrically as Fe (II)- TPTZ (2, 4, 6-tris (2-pyridyl)-s-triazine) colored complex formed at pH 4-5 and measured at 595nm wavelength. Whereas, for the determination of total iron, iron (III) was first reduced to Fe++ using vitamin C as reducing agent and then complexed with TPTZ .The complex so produces is directly proportional to the concentration of Fe (II) produced and Fe (II) already present i.e. total iron in the blood sample in presence of antioxidant. The difference of the total iron and the iron (II) present in the serum is the measure of iron (III). Trace metals were determined using air-acetylene flame. The standards from 1 to 5 ppm for each of the metal separately were run on the spectrometer and the calibration curves were obtained prior to run the samples for the determination of metals in the blood serum of normal subjects and the malarial patients. Blood samples were collected from 50 healthy controls in fasting conditions and a similar condition was maintained while taking blood samples of anemic patients. Each blood sample was centrifuged at 5000 rpm for 20 minutes. The supernatant blood serum was used for the analysis of metals copper, iron, magnesium, and zinc using Atomic Absorption Spectrometer inserting appropriate hollow cathode lamp in it. All standards used were of analytical grade.
Chemicals and reagents
Sulphosalicylic acid was obtained from Merck, Damstadt, Germany and other chemicals to prepare standards were purchased from Sigma Chemical Co. All chemicals were of analytical grade.
Stock Solutions and working
Metal standards Stock solution of 1000 ppm Cu, Fe, Mg, and Zn for each were prepared for corresponding sulphate salts of analytical grade (Sigma Chem.). Working standards were prepared from the stock solutions by diluting with appropriate volume of deionized water and addition of few drops of corresponding concentrated acid.
Statistical Analysis
All statistical analyses were performed using computer program Excel X State (Microsoft Corp., Redmond, WA) Student’s t test was used to assess the significance. Results are expressed as mean ± SD.
RESULTS AND DISCUSSION
Table 1 shows the blood serum levels of trace metal and antioxidant levels in anemic patients with compare to control subjects. The results show significant increase in serum copper levels, where as serum iron, magnesium, zinc and antioxidant levels were decreased in anemic patients compared to the controls.
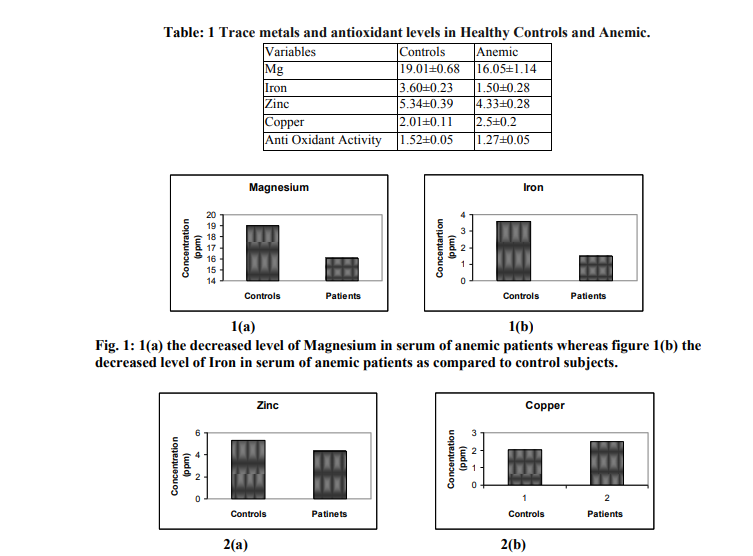

The Table 1 show the levels of trace metals (Fe,Cu, Mg and Zn) and antioxidant activity evaluated in blood samples of anemic patients as compared to controls. Fe deficiency is probably the most common nutritional disorder in the world. Presently estimate based on the WHO criteria specify that around 600–700 million people worldwide have a marked Fe deficiency anemia and about half the adults in developing countries are affected (13). The data of the trace metals under study in blood samples of control and anemic patients are shown in Table 1 and Figure 1(a) to 3(a). The results are given as mean values with standard deviation (±SD) for each metal and antioxidant activity. The mean values of iron in the anemic patients were significantly lower compared to healthy controls (Fig: 1(b). The results indicate the prevalence of Fe deficiency anemia in both genders. In the population of developing countries, the amount of Fe absorbed from the diet is not sufficient to meet many individuals’ requirements. The consumption of a predominantly cereal-based diet, rich in phytate, oxalate, phosphate, fiber, and other inhibitors of Fe absorption, was the main cause of Fe deficiency diseases. Phytates strongly inhibit Fe absorption in a dosedependent fashion, and even small amounts of phytates have a marked effect (14). If the amount of absorbable Fe in the diet cannot be immediately improved, Fe supplement must be included in the diet to control Fe deficiency anemia (15). The concentrations of Zn in anemic patients were found to be significantly lower compared to healthy controls (Fig: 2(a). Zn deficiency may be a contributing factor in anemia (16). Zn deficiency in developing countries is due to low consumption of meat and fish along with food rich in phytate. Food rich in phytate significantly reduce the absorption of Zn, increasing the chance of Zn deficiency. The higher level of Cu was observed in anemic patients as compared to the healthy controls (Fig: 2(b) Copper is required for normal infant development, red and white blood cell maturation, Fe transport, bone strength, cholesterol metabolism, myocardial contractility, glucose metabolism, brain development, and immune function (17). A deficiency of either Fe or Cu will result in anemia, namely, Fe deficiency anemia or Cu deficiency anemia. Copper is essential for the functioning of many Cu-dependent enzymes (18) such as ceruloplasmin (responsible for antioxidant protection, Fe metabolism, and Cu transport), and it was established that the anemia appears to be related to defects in Fe mobilization due to the combined defect of both red ceruloplasmin ferroxidase activity and intracellular utilization (19). Cu is a major component of catalytic centers of different redox enzymes, and thus, its presence is essential for normal physiologic function such as cellular respiration, free radical defense, synthesis of melanin pigment, connective tissue biosynthesis, and cellular Fe metabolism (20). About 95% of the Cu in the blood is bound to ceruloplasmin. These enzymes play a role in the regulation of Fe metabolism. The mean values of magnesium in the anemic patients were significantly lower compared to healthy controls (Fig: 1(a). Low serum magnesium causes potassium, calcium and neuromuscular disturbances, central nervous system and cardiovascular alterations, like arrhythmias (21). Also it can alter glucose homeostasis, increase atherosclerosis, hypertension, myocardial infarction, osteoporosis, migraine, asthma, chronic fatigue syndrome, among others (21, 22). More research on the effects of magnesium deficiency on the health of people is needed to warrant interventions to prevent it. The mean values of Anti-oxidant activity in the anemic patients were significantly lower compared to healthy controls (Fig: 3(a). Antioxidant activity which shows decreasing tendency in patients and this parameter could be used as a biomarker for obtains the status of these patients under medical therapy and its affectivity.
CONCLUSION
It suggests that use of magnesium as a supplementary diet and use antioxidant activity as the status of drug response to greater number of patients. The decreased levels of iron and zinc can be maintained by given supplement of these metals as therapy.
References:
1. Hall A, Miguel E (2001) Anemia in schoolchildren in eight countries in Africa and Asia. Public Health Nutr 4(3):749–756.
2. ADB to pilot project to combat nutritional problems Pakistan. [Online]. 2006 Mar 26[2007 April 5]. Available fr om : htt p://www.adb.org/ Media /Articl/2006/9538. Pakistan-flourfortification/default.asp.
3. Lanzkowsky P (1995) Pediatric hematology and oncology, 2nd edn. Churchill Livingstone, New York.
4. Haas JD, Brownlie TT (2001) Iron deficiency and reduced work capacity: a critical review of the research to determine a causal relationship. J Nutr 131:676S–688S.
5. Grantham-McGregor S, Ani C (2001) A review of studies on the effect of iron deficiency on cognitive development in children. J Nutr 131:649S–666S.
6. Sherriff A, Emond A, Bell JC, Golding J (2001) Should infants be screened for anaemia? A prospective study investigating the relation between haemoglobin at 8, 12, and 18 months and development at 18 months. Arch Dis Child 84:480–485.
7. Afridi HI, Kazi TG, Kazi GH (2005) Evaluation of status of trace and toxic metals in scalp hair samples of normal and anemic human subjects. American Biot Lab 23(8):27.
8. Castillo-Duran C, Garcia H, Venegas P (1994) Zinc supplementation increases growth velocity of male children and adolescents with short stature. Acta Paediatr 83:833.
9. Singh S, Ravishanker R, Singhi P, Nath R (2003) Low plasma zinc and iron in pica. Indian J Pediatr 70(2):139–143.
10. Percival SS (1995) Neutropenia caused by copper deficiency. Nutr Rev 53:59–66.
11. Tammura H, Hirose S, Watanave O (1994) Anemia and neutropenia due to copper deficiency in enternal nutrition. J Parenter Enteral Nutr 18:185–189.
12. Lukaski HC (1999) Chromium as a supplement. Annu Rev Nutr 19:279–302.
13. Oski FA (1993) Iron deficiency in infancy and childhood. N Engl J Med 329(3):190–193.
14. Hallberg L, Brune M, Rossander L (1989) Iron absorption in man: ascorbic acid and dosedependent inhibition by phytate. Am J Clin Nutr 49:140–144.
15. Lozoff B, Jimenez E, Wolf W (1991) Longterm developmental outcome of infants with iron deficiency.N Engl J Med 325:687–693.
16. Sondstrom B (1990) Effect of inositol hexaphosphate on retention of zinc and calcium from the human colon. Eur J Clin Nutr 44:705– 708.
17. L’Abbe MR, Friel JK (1992) Copper status of very low birth weight infants during the first 12 months of infancy. Pediatr Res 32:183–188.
18. Larsson S, Kallebring B, Wittung P, Malmstrom BG (1995) The Cu A center of cytochrome-c oxidase: electronic structure and spectra of models compared to the properties of Cu A domains. Proc Natl Acad Sci 92:7167– 7171.
19. Tapiero H, Townsend DM, Tew KD (2003) Trace elements in human physiology and pathology. Copper. Biomed Pharmacother 57(9):386–398.
20. Gacheru N, Trackman PC, Shah MA (1990) Structural and catalytic properties of copper in lysyl oxidase. J Biol Chem 265(31):19022– 19027.
21. Swaminathan R. Magnesium metabolism and its disorders. Clin Biochem Rev 2003;24(2):47- 66.
22. Barbagallo M, Dominguez LJ. Magnesium metabolism in type 2 diabetes mellitus, metabolic syndrome and insulin resistance. Arch Biochem Biophys 2007;458(1):40-4
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License