IJCRR - 5(8), April, 2013
Pages: 63-68
Date of Publication: 25-Apr-2013
Print Article
Download XML Download PDF
FLOW CYTOMETRY DIAGNOSIS OF (CD4+/CD8+) T CELL LYMPHOBLASTIC LEUKEMIA ASSOCIATED WITH NORMAL KARYOTYPE - CASE REPORT
Author: Trajkova Sanja, Cevreska Lidija, Ivanovski Martin, Dukovski Dusko, Simjanovska-Popova Marija, Panovska-Stavridis Irina
Category: Healthcare
Abstract:Adult T-cell leukemia/lymphoma (ATLL) is a peripheral T-cell neoplasm caused by human T-cell lymphotropic virus type I (HTLV-I). The neoplastic cells are highly pleomorphic and are usually CD4+ and CD8- phenotypically, but some rare cases have unusual immmunophenotype presented by co-expression of CD4/CD8 double positive cells. We present a rare case of a patient with T cell lymphoblastic leukemia associated with double-positive (CD4+/CD8+) blast cells. The diagnose was accessed with multiparameter immunophenotyping by flow cytometry analyses of bone marrow, and for the first time in our country, we confirmed isolated relapse of double-positive (CD4+/CD8+) T cell lymphoblastic leukemia in CNS.
Keywords: T cell lymphoblastic leukemia, flow cytometry, cerebrospinal fluid
Full Text:
INTRODUCTION
Adult T-cell leukemia/lymphoma (ATLL) is T-cell neoplasm that is linked to infection by the human T-cell lymphotropic virus 1 (HTLV-1). ATLL is an aggressive type of leukemia/lymphoma which is diagnose with multiparameter immunophenotype analyses by flow cytometry. The neoplastic cells are highly pleomorphic and are usually CD4+ and CD8- phenotypically, but some rare cases have unusual immmunophenotype presented by co-expression of CD4/CD8 double positive cells.
Here we report a rear case of CD4/CD8 double positive ATLL with normal karyotype. For the first time in our county we confirmed isolated relapse of CD4/CD8 double positive ATLL in CNS by flow cytometry analyses of cerebrospinal fluid.
CASE REPORT
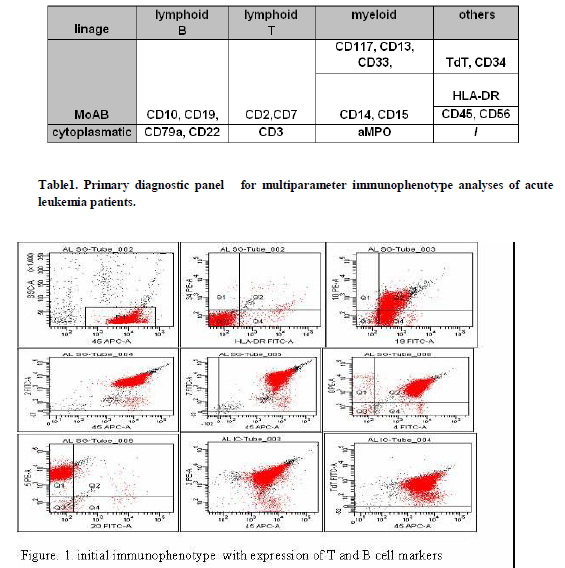
A 20-year old male presented in August 2010, with one week history of weakness, myalgia, lethargy, malaise, high temperature, hemorrhagic syndrome associated with purpura, melena, bleeding from nose, severe headache, vomiting, and pain under left rib. Physical examination disclosed palpable lymphadenopaty in all peripheral regions with dimension of 1sm, palpable spleen of 3sm. Routine laboratory test revealed a white blood cell count 174x10 9/L, hemoglobulin 118gr/L, platelet 10x109/L. Other laboratory tests including liver functional tests, urine analyses, serum protein electrophoresis, blood urea, and creatinine were within normal limits. Examination of peripheral blood smear revealed presence of 90% blast cells. Immmunophenotype study was performed by 4-color-2-laser flow cytometry FACS CantoII (BD-Biosciensies, San Jose, CA, USA) using the standard protocol and panel of monoclonal antibodies (MoAb) according to European Group for the Immunological Classification of Leukemia (EGIL), and clinical groups of the European Leukemia Net. (table1.)
Reading the data was using the FACS DIVA TM software. Using the CD45 gating strategy we found blast cell population of 93,3%: T –cell population CD2, CD7 ,CD5,CD3, CD4, CD8(93%), CD10(59,4%), CD19(84,6%), cytoplasmic Terminal deoxynucleotidyl transferase (TdT)(89%).We confirmed the diagnose of acute lymphoblastic leukemia with expression of T and B cell markers( figure 1).
The patient underwent Hyper CVAD regiment (Cyclophosphamide,Doxorubicin, Vincristine, Dexamethasone , Methotrexate and Cytarabine) course A and B, 3 cycles in total until April 2011 when complete response was achieved. Hematological response was maintained by Methotrexate and Purinethol until September 2011. In George Papanicolau Hospital in Thessalonica, Greece minimal residual disease analysis was done from a sample of bone marrow. The minimal residual disease analysis was positive and revealed CD34+CD10+CD200+CD38+BCL2+TdT+ of 0, 2%. Cytogenetic study from bone marrow showed normal male karyotype 46 x, y.
Few days after returning from Greece patient presented with meningeal syndrome symptoms: visual disturbances, severe headache, and nausea. Routine laboratory test revealed a white blood cell count 3,9x10 9/L, hemoglobulin 148gr/L, platelet 133x109/L, with normal peripheral blood smear. Lumbar puncture (LP) was performed with 293 elements counted in Fuchs- Rosenthal chamber at 400x magnification, with 94% lymphocyte counted. In blood serum and cerebrospinal fluid (CSF) IgM antibodies of West Nile virus was detected by ELISA. The treatment was symptomatic but under suspecion of CNS relapse of ATLL, intrathecal therapy started with Methotrexate, Dexamethasone, Cytarabine. In December 2011 control test of blood serum and CSF were negative of West Nile virus.
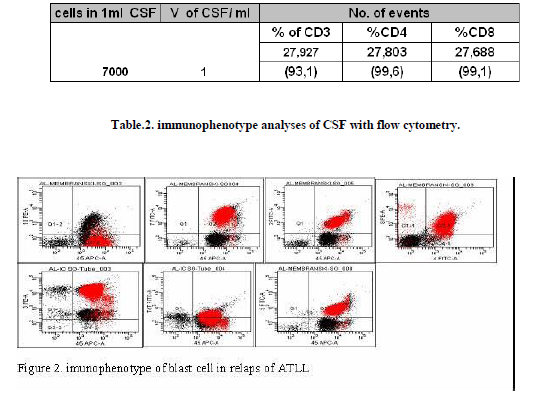
In February 2012 the patient was admitted to the clinic with severe pain in lumbar spine, enlargement of peripheral lymph nodes, high temperature. Routine laboratory test revealed a white blood cell count 53,9x10 9/L, hemoglobin 157gr/L, platelet 123x109/L. Examination of peripheral blood smear revealed presence of 90% blast cells. Immunophenotype analyses of the blast cells in bone marrow simples showed a CD45/CD2/CD7/CD5/CD3 positive with double positive of CD4/CD8, cytoplasmatic TdT and CD3 were positive and ATLL was confirmed (figure 2).
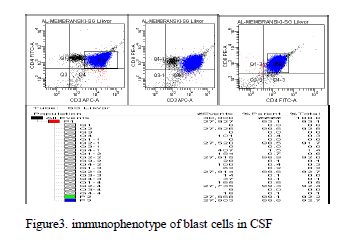
The patient was given a chemotherapy regimen FLAG-Ida (Fludarabine, Cytarabine, Idarubicyn) along with 20Gy radiotherapy of CNS. In Mart 2012 patient underwent allogeneus stem cell transplantation with Busulfan-Cyclophasphamide-Melphalan regimen and peripheral stem cells of HLA-DNA identical sister donor. After two month of allo - transplant patient was admitted to the clinic under suspection of leukemia involvement of CNS because of presence meningeal syndrome symptoms. Lumbar puncture was performed with 2775 elements in CSF detected in Fuchs- Rosenthal chamber at 400 x magnifications, with 98% lymphocyte and lymphoblast counted. A cytological finding of CSF was V classification group with conformation of lymphoblast cells in CSF. Several lumbar punctures were repeated with elevation of elements to 7000 in 1ml of CSF. For the first time in our country, in our immunohematology unit immunophenotype analyses of CSF was performed.
Up to 1 ml extra CSF was obtained during routine LP. Immediately after withdrawal, CSF was put in sterile tube with 1ml plasma. CSF cells were concentrated by centrifugation (8 minutes 450g) and re-suspended in 0,5ml phosphate – buffered saline (PBS). Next the samples ware incubated for 10 minutes at room temperature in the dark with 5microliters of each of the following (Moab): CD3 conjugated with allophycocyanin (APC), CD4fluorescein isothiocyanate (FITC) and CD8 phyoerytrin (PE).We applied only this three moAb knowing the immunophenotype of the patient, because we had insufficient material in volume and cells in 1ml of CSF. After incubation, cells were washed and subsequently re-suspended in PBS. Immediately after staining list mode data were acquired on a by 4-color-2-laser flow cytometry FACS CantoII (BD-Biosciensies, San Jose, CA, USA).Using BD DIVA TM software analysis were performed. The immunophenotype definition of leukocytes and major T subsets are shown on table 2, figure 3.
Salvage regimen which included high doses of Cyterabine and Methotrexate was started with intrathecal administration of Cyterabine, Methotrexate, and Dexamethasone. The patient underwent second allo-transplant from his HLA-DNA identical sister with administration of absolute number of CD34 of 1,428x10 6/L (determined with flow cytometry analysis according to ISHAGE recommendation).
Second pulse of CNS radiotherapy was applied followed by several intrathecal applications of Cyterabine, Methotrexate, and Dexamethasone. However the disease recurred again with infiltration of medulla spinalis, being complicated with quadriparesis, vital centre dysfunction. Overall the patient survived 24 month from the time of the diagnosis and 5 month after first allo-transplant.
DISCUSSION
We have reported a rare case with ATLL with expansion of a CD4+CD8+ double positive cell population in a young patient with normal karyotype. For several years in our country flow cytometry is tool which is part of standard diagnostic algorithm of acute leukemia. For the first time we confirmed the usefulness of flow cytometry in imunophenotype analysis of CSF in young patient with CNS involvement of the disease.
Babusikova O (1) in her study explores the same problem like us. Immunophenotypic profiles of specimen from different sites (CSF, BM, and PB) of the same patient have been performed and no phenotypic changes were found. So we use well know immunophenotype profile from BM analysis, and we applied positive Moab like CD3, CD4, and CD8 which were positive in BM analysis. The results from our study are the same like those from Babusikova O (1), that positive CSF immunology is a useful indicator of malignancy and reflects leptomeningeal involvement; also the technique is reliable and quick.
In the past flow cytometry has been considered to have limitation for the study of CSF samples, mainly related to the low cell count observed in this type of sample. Few authors have reported about this limitation, but Subirá D ET all. (2) proposed a flow cytometric approach that has been applied in our study. Our results have shown that flow cytometry could be reliable for the identification of total T cells, major CD4subsets. Moreover CD8+ T lymphocytes were identified as well. Graof TM et all. (3) helped us with their protocol of addition of serum-containing medium immediately after CSF sampling, and to prevent cell loss.
On the clinical side of the study double positive CD4/CD8 is associated with very aggressive course. Kim et all (4) reported an extremely aggressive course with poor survival in a patient with double positive CD4/CD8ATLL with skin manifestation.
Kamihira S et all (5) reported a poorer prognosis with a medium survival of 7, 8 months as compared to patient with typical CD4-CD8-phenotype. Ciminale V et all(6) et Raza et all(7)have reported a rare cases of double positive CD4/CD8 ATLL with hypercalciemia and short overall survival. There are several clinical subtypes of ATLL, associated with mature CD4+CD8- T- cell phenotype. However same rare cases of immunophenotype characterized by co-expression of CD4+CD8+ T- cells have been reported (8).
Our patient had initial flow cytometry analyses on a sample of BM with expression of T and B cell markers, but when relapse occurred the analyses from BM reveal expression only of T cell markers. Initial event was on early hematopoietic progenitor, but when relapse occurred only T cell markers express on blastic cells with double positive CD4/CD8, which presented leukemic clone with characteristics of cortical ATLL.
Another interest feature is cytogenic study which showed normal karyotype associated with aggressive disease. In literature abnormal karyotype is found in 35-36% of patients with ATLL. Itoyama T et all(9) reveled cytogenetic findings in 50 cases of ATLL where multiple breaks( at least 6), abnormalities of chromosome 1p,1p22, 1q,1q10-21, 2q, 3q, 3q10-12, 3q21, 14p, 14q32 and partial loss of chromosome 2q,9p,14p, 14q, 17q correlated with shorter survival. Several authors have suggested that14q32 translocation is the most common abnormalities of T cell malignancy (10).
FUTURE ASPECTS AND CONCLUSION
We presented a case of a young male with normal karyotype with double positive CD4/CD8 ATLL. Based on this study we recommend immunophenotype analyses of CSF to be routine diagnostic tool. The method will improve the diagnostic accuracy of classical cytological studies in acute leukemia (T, B ALL) and acute myeloid leukemia especially M5, extra nodal Non Hodgkin Lymphoma etc. This diagnostic tool will perform risk stratification of patient with adequate therapeutically approach.
ACKNOWLEDGEMENT
Authors acknowledge the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Authors are grateful to IJCRR editorial board members and IJCRR team of reviewers who have helped to bring quality to this manuscript.
References:
- BabusikovaO, ZeleznikovaT. The value of multiparameter flow cytometry of cerebrospinal fluid involved by leukemia/lymphoma cells. Neoplasia.2004;51(5):345-51.
- Subirá D, CastananS, AceitunoE, et al. Flow Cytometric Analysis of Cerebrospinal Fluid Samples and Its Usefulness in Routine Clinical Practice. AmJ Clin Pathol.2002;117:952-958.
- Graaf MT, Broek PDM, Kraen J et all. Addition of serum – containing medium to cerebrospinal fluid prevents cellular loss over time.J Neurol.2011;258:1507-1512
- Kim YJ, Hwang ES, Kim IH, et al. CD4/CD8 double-positive, acute type of adult T-cell leukemia/lymphoma with extensive cutaneous involvement. Int J Dermatol. 2006; 45:1193–1195.
- Kamihira S, Sohda H, Atogami S, et al. Phenotypic diversity and prognosis of adult T-cell leukemia. Leuk Res. 1992; 16:435–441.
- Ciminale V, Hatziyanni M, Felber BK, et al. Unusual CD4+CD8+ phenotype in a Greek patient diagnosed with adult T-cell leukemia positive for human T-cell leukemia virus type I (HTLV-I) Leuk Res. 2000;24:353–358.
- RazaS, NaikS, Venkat P. Dual-Positive (CD4+/CD8+) Acute Adult T-Cell Leukemia/Lymphoma Associated with Complex Karyotype and Refractory Hypercalcemia: Case Report and Literature Review. Case Rep Oncol. 2010 Sep-Dec; 3(3): 489–494.
- Yamada Y, Nagata Y, Kamihira S, et al. IL-2-dependent ATL cell lines with phenotypes differing from the original leukemia cells. Leuk Res. 1991; 15: 619–625.
- Itoyama T, Chaganti RS, Yamada Y, et al. Cytogenetic analysis and clinical significance in adult T-cell leukemia/lymphoma: a study of 50 cases from the human T-cell leukemia virus type-1 endemic area, Nagasaki. Blood. 2001; 97: 3612–3620.
- Tsukasaki K, Krebs J, Nagai K, et al. Comparative genomic hybridization analysis in adult T-cell leukemia/lymphoma: correlation with clinical course. Blood. 2001; 97: 3875–3881.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License