IJCRR - 5(8), April, 2013
Pages: 11-19
Print Article
Download XML Download PDF
IN VITRO EVALUATION OF AFLATOXIN M1 CONTROL POTENTIAL OF SIX ESSENTIAL OILS IN BENIN
Author: Philippe Sessou, Fidele Tchobo, Pascal Agbangnan, Elvis Adjalian, Paulin Azokpota, Issaka Youssao, Souaibou Farougou
Category: General Sciences
Abstract:Aflatoxin M1 (AFM1) is hepatocarcinogen frequently found in cheese. Its presence in this foodstuff constitutes a serious threat for consumers. The present study had evaluated aflatoxin M1 control potential of six essential oils against synthetic aflatoxin M1. It had consisted to blend Aflatoxin M1 diluted in acetonitrile (1\?g/mL) with essential oil to achieve final concentrations of 2%, 4%, 6%, 8% and 10% of oil and to follow the disintegration of this AFM1 in the meantime of 30 minutes, 1, 2, 3, 4, 5 and 24 hours on silica gel with or without thin layer chromatography assay (TLC). The results obtained revealed that at the concentrations tested, no fluorescence was observed for the mixtures of Aflatoxin M1 with C. zeylanicum, P. racemosa and Syzygium aromaticum essential oils on silica gel without chromatography assay contrary to results obtained for the same mixtures after chromatography assay. The mixture of Cymbopogon citratus showed fluorescence with or without TLC assay. Pure extracts of Ocimum gratissimum and Zingiber officinale showed a green fluorescence. In sum, all the essential oils tested didn't possess capacity to destroy AFM1 at concentrations investigated. Although, these extracts cannot be used for controlling aflatoxin M1, their potentiality for inhibiting mycotoxins production by moulds can be considered.
Keywords: Aflatoxin M1, antiaflatoxin assay, essential oils, TLC assay
Full Text:
INTRODUCTION
Benin is underdeveloped country which produces milk with an output of 94 million liters in 2009 (FAO, 2010). Milk produced in Benin, instead of domestic consumption, is processed to many dairy products among which traditional cheese wagashi is the most consumed largely by rural than citizens populations due to its content in good source of many nutrients such as proteins and calcium (Dossou et al., 2006; Keke et al., 2008). However, Benin country has favourable warm and humid climate, socio-economic (ignorance of the toxin and poor infrastructure to manage mycotoxins prevention strategies) and compelling environmental (drought) factors that enhance the growth of aflatoxigenic fungi and subsequently aflatoxins production inside wagashi (Okeke et al., 2012; Sessou et al., 2012a, b). Aflatoxins are toxic fungal metabolites produced by Aspergillus species, mainly by Aspergillus flavus and Aspergillus parasiticus, but also by Aspergillus nomius, Aspergillus pseudotamarii, Aspergillus ochraceus (IARC, 2012; Siddappa et al., 2012). Aflatoxins consist of a group of approximately 20 related metabolites, among which aflatoxins B1, B2, G1 and G2 are often found in foods. Aflatoxin B1 (AFB1) is metabolized by the animals consuming these contaminated feeds to AFM1 mainly by the hepatic microsomal mixed-function oxidase system (Siddappa et al., 2012). Aflatoxin M1 (AFM1) was classified by the International Agency for Research on Cancer (IARC) as a group 1 human carcinogen (IARC, 2012, Okeke et al., 2012). Thus, its potential risk to human health makes its presence in milk products such as cheese wagashi undesirable. AFM1 is unaffected by pasteurization and ultra-high-temperature (UHT) treatment (Tekinsen and Eken, 2008). Alternative methods for its control are needed to be performed. The control or disintegration of AFM1 by natural agents as essential oils, safe for human (Burt, 2004) in order to reduce or eliminate this toxic metabolite in wagashi eventually contaminated is necessary for minimizing public health hazards. The objective of this work was to evaluate aflatoxin M1 control potential of six essential oils extracted in Benin.
MATERIAL AND METHODS
Material was constituted of six essential oils extracted by hydrodistillation from Cinnamomum zeylanicum, Cymbopogon citratus, Ocimum gratissimum, Pimenta racemosa, Syzygium aromaticum and Zingiber officinale previously analyzed by GC/MS and GC/FID (table 1) and Aflatoxin M1 ( 10 µg/mL in acetonitrile, Lot: LB 96767; 46319-U, quantity: 1 mL, Exp: Nov/2015; USA) purchased at SUPELCO Analytical society based at Unity State of America and diluted in acetonitrile to have final concentration of 1µg/mL which is used for the assay.
The method used in this study was based on that described by Adjou et al. (2012a, b, c and 2013). It had consisted to mix Aflatoxin M1 diluted in acetonitrile (1µg/mL) with essential oil to achieve final concentrations of 2%, 4%, 6%, 8% and 10% of oil and to follow the inhibition or disintegration of this metabolite (AFM1) in the meantime of 30 minutes, 1, 2, 3, 4, 5 and 24 hours on silica gel without or after thin layer chromatography assay (TLC). Five microliter of mixture (Aflatoxin + essential oil) or pure extract of essential was spotted and air dried on TLC plates (TLC Silica gel 60 F254, Merck, Germany) and observed under long wave (365 nm) UV and then developed in the solvents system comprising TEF (Toluene/ethylacetate/Formic acid, 5:4:1 v/v/v) and CAP (Chloroform/acetone/2-propanol, 85:15:20, v/v/v), air dried and observed under long wave (365 nm) UV.
RESULTS AND DISCUSSION
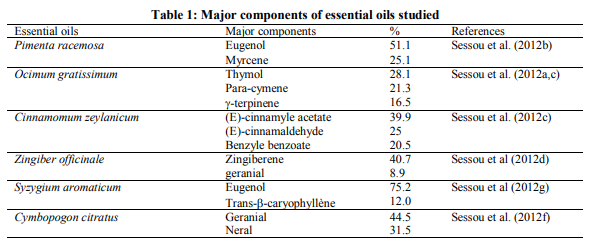
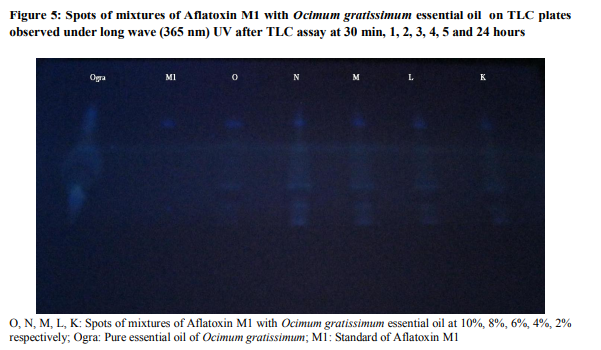
The present study had investigated the potentiality of six essential to destroy Aflatoxin M1 in perspective for their use to control mycotoxins produced eventually inside traditional cheese wagashi. The results obtained from this work are prsented in figures 1 to 5 and showed that these extracts studied didn’t possess capacity to decontaminate food contaminated by aflatoxin M1. In fact, results obtained from this study revealed that at the concentrations tested, no fluorescence was observed for the mixtures of Aflatoxin M1 with Cinnamomum zeylanicum, Pimenta racemosa and Syzygium aromaticum essential oils on silica gel without chromatography assay contrary to results obtained for the same mixtures after chromatography assay. Indeed, these extracts had masked the fluorescence of Aflatoxin M1 when mixed together and spotted on TLC plate and observed under UV light 365 nm without tthin layer chromatography assay (figure1). Based on theory of Adjou et al. (2012 a, b, c and 2013), we can conclude that our three extracts C. zeylanicum, P. racemosa and S. aromaticum possessed antiaflatoxin M1 potential. But this conclusion will be wrong when we consider the elution of these mixtures which showed the fluorescence at the same bright with the standard AFM1 spotted (figure 2 to 5). The masking of fluorescence of Aflatoxin M1 when mixed with extracts of C. zeylanicum, P. racemosa and S. aromaticum could be explained by a reaction of components of these oils with the fluorescence pole of Aflatoxin M1 which is not inevitably sinonymous of the destroying of the toxic metabolite. These extracts may possess capacity to inhibit production of mycotoxins produced by toxinogenic moulds. The mixture of Cymbopogon citratus showed fluorescence either with or without TLC assay. This last pure oil (C. citratus) with no fluorescence when spotted on TLC plate had not capacity to mask the aflatoxin M1 fluorescence when mixed together with the toxic metabolite. Pure extracts of Ocimum gratissimum and Zingiber officinale spotted alone showed a green fluorescence on silica gel without TLC assay and didn’t allow appreciating their activity at this step (figure 1). The present study had shown that the works of Adjou et al. (2012a,b,c and 2013) concerning the appreciation of aflatoxigenic inhibition potential of extracts based on their ability to inhibit fluorescence of aflotoxins presents insufficiencies. In fact, these authors have wrongly exploited the method of Nguyen (2007) and Atanda et al. (2011) whose works were made not for evaluating the aflatoxin production inhibition by extracts but for the capacity of strains of Aspergillus to produce aflatoxins revealed by fluorescence of these metabolites under UV light. Our work showed that the inhibition of fluorescence of the metabolite is not inevitably synonymous of capacity of extract to inhibit the metabolite production of toxinogenic fungus which normally grows. Also, the appearance of fluorescence after observing the mixture of essential oil plus the metabolite in culture medium is not synonymous of incapacity of extract to possess antiaflatoxin inhibition potential. Indeed, our study showed that the pure extracts of O. gratissimum and Z. officinale spotted alone showed green fluorescence. As recommendation, before conclude that the extracts possess antiaflatoxin inhibition potential based on inhibition of fluorescence, the previous authors (Adjou et al) must complete their work by proceeding to the detection and the quantification of the aflatoxins in the whole culture medium composed of extract, toxinogenic fungus which had its metabolite fluorescence inhibited or not. As alternative methods to detected capacity of mycotoxins production inhibition by essential oils, that described by Singh et al. (2010), Prakash et al. (2012a, b, c) and Shukla et al. (2012) are scientifically efficient and appropriate. In sum, this study showed that essential oils of C. zeylanicum, C. citratus, O. gratissimum, P. racemosa, S. aromaticum and Z. officinale didn’t possess potential to destroy aflatoxin M1 when presented inside food. Also, it showed that the appreciation of inhibitory aflatoxin production potential of essential oils based on their ability to inhibit fluorescence of metabolites produced by toxinogenic moulds presents more insufficiencies.
CONCLUSION
The work has assessed the potential of six essential oils to control aflatoxin M1 in perspective to their use to destroy this toxic metabolite inside cheese wagashi contaminated. Results obtained showed that these extracts had not potential to disintegrate AFM1 at concentrations investigated. However, these extract especially C. zeylanicum, P. racemosa and S. aromaticum can be considered as promising extract for mycotoxins production inhibition.
ACKNOWLEDGMENTS
The authors are grateful to University Council of Development (CUD) for its financial support. They are also thankful to Professor Dominique SOHOUNHLOUE for his scientific contributions. Authors acknowledge the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Authors are grateful to IJCRR editorial board members and IJCRR team of reviewers who have helped to bring quality to this manuscript.
References:
1. Adjou ES, Dahouenon - Ahoussi E, Degnon RG, Soumanou MM, Sohounhloue DCK, 2012a. Bioefficacy of essential oil of Lantana Camara from Benin against the growth of fungi and aflatoxin production. Journal of Resent Advances in Agriculture 1(4): 112 - 121.
2. Adjou ES, Kouton S, Dahouenon - Ahoussi E, Degnon GR, Soumanou MM, Sohounhloue DCK, 2012b. Investigations on bioactivity of essential oil of Ageratum conyzoides L., from benin against the growth of fungi and aflatoxin production. International Journal of Pharmaceutical Sciences Review and Research, 13(1): 143 - 148.
3. Adjou ES, Kouton S, Dahouenon - Ahoussi E, Sohounhloue DCK, Soumanou MM, 2012c. Antifungal activity of Ocimum canum Essential oil against Toxinogenic Fungi isolated from Peanut Seeds in post-harvest in Benin. International Research Journal of Biological Science, 1(7): 20 - 26
4. Adjou ES, Kouton S, Dahouenon - Ahoussi E, Soumanou MM, Sohounhloue DCK, 2013. Effect of essential oil from fresh leaves of Ocimum gratissimum L. on mycoflora during storage of peanuts in Benin. Mycotoxin Research, 29(1):29 - 38.
5. Atanda OO, Ogunrinu MC, Olorunfemi FM, 2011. A neutral red desiccated coconut agar for rapid detection of aflatoxigenic fungi and visual determination of aflatoxins. World Mycotoxin. Journal, 4(2), 147 - 155
6. Dossou J, Hounzangbe Adote S, Soule H, 2006. Production et transformation du lait frais en fromage peulh au Bénin; Guides de bonnes pratiques, version validée lors de l’atelier national du 14 juillet 2006. [En ligne], consulte le 29 octobre 2009. http://www.repol.info/IMG/pdf/Fiche_wagas hi_VF.pdf.
7. FAO, 2010b. Evolution de la Production Nationale de Lait. Consulté le 4 juin 2010 à l’adresse suivante : http://www.countrystat.org/ben/cont/pxwebq uery/ma/053cpd060/fr/vType/quick
8. IARC (International Agency for Research on Cancer), 2012. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, vol. 100F. IARC.
9. Kèké M, Yéhouénou B, Dahouénon E, Dossou J, Sohounhloué DCK, 2008. Contribution à l'amélioration de la technologie de fabrication et de conservation du fromage peulh waragashi par injection de Lactobacillus plantarum. Ann. Sci. Agron. Benin, 10: 73 - 86
10. N’guyen MT, 2012. Identification des espèces de moisissures potentiellement productrices de mycotoxines dans le riz commercialisé dans cinq provinces de la région centrale du Vietman: Etude des conditions pouvant induire la production de mycotoxines. Institut National Polytechnique de Toulouse (INPT). Thèse de doctorat, p. 147.
11. Okeke KS, Abdullahi IO, Makun HA, Mailafiya SC, 2012. Preliminary survey of aflatoxin M1 in dairy cattle products in Bida, Niger State, Nigeria. African Journal of Food Science and Technology, 3(10): 273- 276
12. Prakash B, Singh P, Kedia A, Dubey NK, 2012. Assessment of some essential oils as food preservatives based on antifungal, antiaflatoxin, antioxidant activities and in vivo efficacy in food system. Food Research International 49: 201–208
13. Prakash B, Singh P, Kedia A., Dwivedy KA, Singh A., Dubey N.K. Mycoflora and aflatoxin analysis of arachis hypogaea l. and assessment of anethum graveolens l. seed and leaf essential oils against isolated fungi, aflatoxin production and their antioxidant activity. Journal of Food Safety 32 (2012) 481 - 491
14. Prakash B, Singh P, Mishra KP, Dubey NK, 2012. Safety assessment of Zanthoxylum alatum Roxb. essential oil, its antifungal, antiaflatoxin, antioxidant activity and efficacy as antimicrobial in preservation of Piper nigrum L. fruits. International Journal of Food Microbiology, 153:183 - 191
15. Sessou P, Farougou S, Alitonou G, Djenontin TS, Yèhouénou B, Azokpota P, Youssao I, Sohounhloue D, 2012a. Chemical Composition and Antifungal activity of Essential oil of Fresh leaves of Ocimum gratissimum from Benin against six Mycotoxigenic Fungi isolated from traditional cheese wagashi. International Research Journal of Biological Sciences, 1(4): 22 - 27
16. Sessou P, Farougou S, Azokpota P, Youssao I, Fanou B, Kaneho S, Agniwo B., Yèhouenou B, Ahounou GS, Chabi-Egba M, Sohounhloue D, 2012b. In vitro antifungal activity of essential oil of Pimenta racemosa against fungal isolates from wagashi, a traditional cheese produced in Benin. International Journal of Natural and Applied Sciences, 8 (1): 25-34.
17. Sessou P, Farougou S, Noudogbessi J - P, Fanou B, Azokpota P, Youssao I. Sohounhloué D, 2012d. Chemical composition and in vitro antifungal activity of Zingiber officinale essential oil against foodborne pathogens isolated from a traditional cheese wagashi produced in Benin. International Journal of Biosciences, 2 (9): 20 - 28.
18. Sessou P, Farougou S, Kaneho S, Djenontin S, Alitonou GA, Azokpota P, Youssao I, Sohounhloué D, 2012e. Bioefficacy of Cymbopogon citratus essential oil against foodborne pathogens in culture medium and in traditional cheese wagashi produced in Benin. International Research Journal of Microbiology, 3(12): 406 - 415.
19. Sessou P, Farougou S, Kaneho S., Djenontin S., Alitonou G. A., Azokpota P., Youssao I., Sohounhloué D, 2012f. Bioefficacy of Cymbopogon citratus essential oil against foodborne pathogens in culture medium and in traditional cheese wagashi produced in Benin. Int. Res. J. Microbiol., 3(12): 406 0 - 415.
20. Sessou P, Farougou S, Yehouénou B, Agniwo B, Alitonou GA, Azokpota P, Youssao I, Sohounhloué D, 2013g. Biological control of spoilage and pathogens moulds in culture medium and Beninese traditional cheese wagashi by Syzygium aromaticum essential oil. African Journal of Microbiology Research (in press).
21. Shukla R, Singh P, Prakash B, Dubey NK, 2012. Antifungal, aflatoxin inhibition and antioxidant activity of Callistemon lanceolatus (Sm.) Sweet essential oil and its major component 1,8 - cineole against fungal isolates from chickpea seeds. Food Control, 25: 27 - 33
22. Siddappa V, Nanjegowda DK, Viswanath P, 2012. Occurrence of aflatoxin M1 in some samples of UHT, raw and pasteurized milk from Indian states of Karnataka and Tamilnadu. Food and Chemical Toxicology 50: 4158 - 4162
23. Singh P, Shukla R, Prakash B, Kumar A, Singh S, Mishra PK, Dubey NK, 2010. Chemical profile, antifungal, antiaflatoxigenic and antioxidant activity of Citrus maxima Burm. and Citrus sinensis (L.) Osbeck essential oils and their cyclic monoterpene, dl-limonene. Food and Chemical Toxicology, 48: 1734 - 1740.
24. Tekins KK, Eken HS, 2008. Aflatoxin M1 levels in UHT milk and kashar cheese consumed in Turkey Food and Chemical Toxicology, 46: 3287 - 3289




|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License