IJCRR - 5(3), February, 2013
Pages: 76-82
Date of Publication: 18-Feb-2013
Print Article
Download XML Download PDF
HISTOPATHOLOGICAL STUDY OF MENINGIOMA IN CIVIL HOSPITAL, AHMEDABAD
Author: Smita Shah, R. N. Gonsai, Rinku Makwana
Category: Healthcare
Abstract:Objective: This study was done to determine the clinical and histological pattern of intracranial meningioma, to determine the sociodemographic characteristic and clinical presentation and correlate this to the clinical patterns of intracranial meningioma, to find out the anatomical location of meningioma and to document the WHO histological grade of meningioma. Materials and Methods: We have studied 51 cases of meningioma. Meningioma was diagnosed primarily by contrast enhanced CT Scan and Magnetic Resonence Imaging (MRI) of brain. This was confirmed by histopathological examination. Histopatholgical results were examined according to age and sex distribution, anatomical location of tumor, histological type and WHO grading of tumor. Correlation of clinical features and radiological findings were made with histpathological results. Results and conclusion: Most of the sufferer was female 34 (67%). The commonest age group was 40-59 years. The commonest site of tumor was convexity of brain 26 (60%). The commonest histopathological type was meningotheliomatous meningioma 20 (39%). The 92% of the meningioma was WHO GRADE I tumor.
Keywords: meningioma, WHO grading of meningioma, prognosis of meningioma
Full Text:
INTRODUCTION
The tumor originating from the meninges was termed as meningioma by Cushing in 1922 . Meningiomas originate from the arachanoidal cap cell, a meningothelial cell in the arachnoidal membrane. They generally arise were arachnoidal villi are many . The arachnoid cap cells are most prevalent near collections of arachnoid villi at the dural venous sinuses and their large tributaries. Meningiomas may arise anywhere the cap cells are located Meningiomas are account for 15% of all intracranial tumors. They commonly occur in the fourth to sixth decades of life, with a mean age of 45 years at diagnosis. Females have meningiomas more often than males; ratio is 2:1 for intracranial and 4:1 for spinal meningiomas . The etiology of meningioma is unknown. Cases exist in which the tumor has arisen under a fracture, from an area of scared dura, or around a retained foreign body. Low and high dose radiation has been implicated in meningioma formation especially during childhood. Neurofibromatosis 1 and 2 genetic diseases inherited in autosomal dominant fashion may be associated with meningioma . 90% of meningiomas are located intracranialy and of these 90% are supratentorial. According to site meningiomas are located at parasaggital, convexity, sphenoid ridge, suprasellar, posterior fossa, olfactory groove, middle fossa, tentorial, peritorcular, lateral ventricle, foramen magnum , spinal, orbit or optic nerve sheath, few located at ectopic site . At spinal level meningioma clearly favors the thoracic region. Cervical being uncommon and lumber is rare. Also recognized are epidural, calvarial and intrapetrous as well as variant located entirely outside the craniospinal confines . Although most meningiomas are benign, they have a surprisingly broad spectrum of clinical characteristics, and histologically distinct subsets are associated with high risk of recurrence, even after seemingly complete resection. In rare instances, meningiomas are malignant. The WHO classification aims to better predict the divergent clinical characteristics of meningiomas with a histological grading system based on statistically significant clinicopathological correlations. Meningiomas are classified as benign, atypical, or malignant. Benign meningiomas are not encapsulated; they grow invaginating, but demarcated, from the brain. They grow with finger like projections, and penetrate surrounding mesenchymal tissue, including bone. They may produce both an osteoblastic and a lytic reaction . Meningiomas show positive immunostain with vimentin, desmoplakin, and epithelial membrane antigen. They have a grade 1 biological behaviour. Meningiomas grow in 3 primary histologic patterns: (1) meningothelial, (2) fibroblastic, or (3) transitional, a combination of meningothelial and fibrous. Meningothelial meningiomas consist of lobules of cells with oval pale nuclei, with chromatin marginated around the nucleus. The cell has an ill defined cellular membrane, and nuclear and cytoplasmic invaginations often produce pseudoinclusions . Fibroblastic meningiomas have parallel interlacing bundles of spindle shaped cells with abundant collagen and reticulin between cells. Transitional meningiomas have a mixed pattern of both meningothelial and fibroblastic features. They more often contain whorls or psammoma bodies. Benign meningiomas of WHO grade I can invade the dura, dural sinuses, skull, and even extracranial compartments, such as orbit, soft tissue, and skin. Although these types of invasion make it more d fficult to resect the tumour, they are not considered as atypical or malignant. By contrast, brain invasion is associated with recurrence and mortality rates similar to atypical meningiomas in general, even if the tumour seems completely benign otherwise . WHO grade II meningiomas include atypical, chordoid, and clear cell meningiomas . Both grade II and III WHO classifications of meningiomas require brain invasion as a criterion 10 . WHO grade II meningiomas make up 5% to 7% of all meningiomas 11 . Some pathologists feel that a meningioma should be called “malignant” only when there is frank brain invasion, although brain invasion has been documented in benign, atypical, and anaplastic meningiomas and felt to be an additional criteria for malignant classificatio . Atypical meningiomas are diagnosed based on increased mitotic index of equal to or greater than 4 mitoses per 10 high power fields or 3 or more of the following features: increased cellularity, small cells with high nuclear to cytoplasmic ratio, prominent nucleoli, uninterrupted patternless or sheet like growth, and foci of "spontaneous" or "geographic necrosis" 10 . Clear cell and chordoid variants of meningioma are associated with higher recurrence rates even without the above criteria 12,13 . Thus, these meningiomas are graded as WHO grade II by definition. Clear cell meningiomas make up only 0.2% of all meningiomas. This type usually behaves aggressively and can metastasize to the CSF. Clear cell meningiomas often occur in patients of younger age and occur more often in the spinal and cerebellar pontine region. Recurrence rate of clear cell meningiomas is 46% to 80% 14 Clear cell meningioma is composed of sheets of polygonal cells with clear, glycogen rich cytoplasm positive for periodic acid Schiff, and ense perivascular and interstitial collagenisation. Chordoid meningiomas are histologically similar to chordoma, with cords of small epithelioid tumour cells that contain eosinophilic or vacuolated (i.e., resembling physaliferous cells) cytoplasm embedded in a basophilic, mucin rich common in the spinal cord and posterior fossa, whereas chordoid meningiomas are typically supratentorial. WHO grade III meningiomas make up 1.0% to 2.8% of all meningiomas. These include anaplastic, rhabdoid, and papillary types . Malignant meningiomas have further increase in mitoses and cellularity with conspicuous necrosis 15 . Anaplastic or malignant meningiomas by definition must have equal to or greater than 20 mitoses per 10 high power field 10 . Atypical and malignant meningiomas have a much higher recurrence rate after resection than do benign meningiomas. Recurrence rates were 6.9% for benign meningiomas, 34.6% for atypical meningiomas, and 72.7% for malignant meningiomas . Papillary and rhabdoid meningiomas are rare variants and have an aggressive clinical course and higher rates of recurrence, metastases, and mortality 10 Papillary meningiomas generally occur in the pediatric population. Their cell processes terminate in papilla on blood vessels, with tapering of their processes to form pseudorosettes. Rhabdoid meningioma is a new pathologic variant of malignant meningioma with peritumoral edema, bone involvement, and significant cystic components on Magnetic Resonence Imaging (MRI) study of brain 16 The prognosis for meningiomas following gross total resection depends on the histology. In a single series of 1799 meningioma specimens from 1582 patients followed for an estimated average of 13 years, 93.1% of benign meningiomas, 65.4% of atypical meningiomas, and 27.3% of malignant meningiomas were cured by surgery . Recent research has investigated possible prognostic factors in atypical and malignant meningiomas specifically. In an analysis of 76 atypical meningiomas and 10 malignant meningiomas, high mitotic count, brain invasion, and parasagittal falcine location were significantly associated with decreased recurrence free survival 17 . Also, Ki 67 index greater than 4% was also associated with decreased time to recurrence. These appear to be important pathologic indicators of aggressiveness of these tumor types. A recent study found that anatomic location of tumor also to have prognostic significance 18 . They reviewed 378 patients with meningioma, looking for causes for high grade pathology. They found that nonskull base meningiomas, prior surgery, and male sex all increased risk of grade II or III pathology, which extrapolates to poorer prognosis and increased likelihood of recurrence.
MATERIAL AND METHOD
This was descriptive study. This study was carried out at the histopathology laboratory, Department of Pathology, B. J. medical college Ahmedabad, from January 2010 to August 2011. We have studied 51 cases. Histological subtype and WHO grading for all meningiomas were carried out. The parameters like patient’s age, gender, location of tumor, microscopic appearance of tumor were studied.
RESULTS
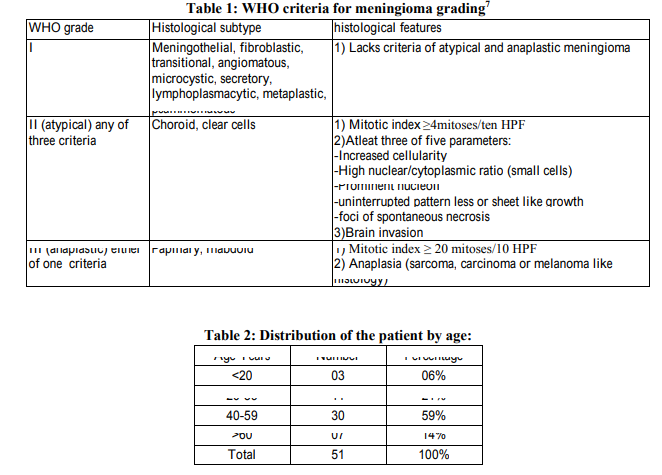
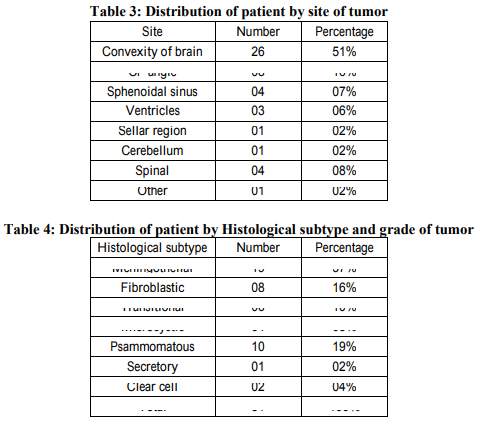
The present study was conducted at pathology department of B. J. Medical College and Civil Hospital Ahmedabad, Gujarat, India. A total of 51 cases diagnosed as meningioma by histopathological examination were studied. Detailed clinical history and radiological findings were assessed in all 51 cases. In our study meningiomas were most common in the age group of 40 59 years (59%), followed by in the age group of 20 39 years (21%). Meningiomas were least common in the age group of <20 years (06%). Out of 51 cases 34 (67%) were female and 17 (33%) were male. In our study most common clinical feature was headache, seen in 38 (75%) of cases followed by vomiting, seen in 9 (17%) of cases. In our study most common location of tumor was convexity of brain in 26 (51%) of cases. 46 (90.2%) cases were in intracranial location. In our study most common histological subtype was meningotheliomatous meningioma 19 case (37%). And most of the tumor was WHO GRADE I (47 case 92%). Whereas WHO GRADE II and WHO GRADE III tumor comprise respectively 8% and 0% of total cases.
DISCUSSION:
The present study was conducted in the department of Pathology, B. J. Medical College and Civil Hospital Ahmedabad, Gujarat, India. In present study meningiomas were most common in the age group of 40 59 years 59%. Haradhan et al 2009 19 studied 25 cases of meningiomas, with most tumor were in the age group of 40 59 years 60%. Among the 51 patients, in our study 34 (67%) were female and 17 (33%) were male, with male to female ratio being 1:2. Akyildiz EU et al 2010 20 were studied 245 cases of meningioma, 74 (30.2%) were male and 171(69.8%) were female, with male to female ratio being 1:2.3. Joseph wanjeri et al 2011 21 a study of 78 cases of meningioma, females 69.2% were more affected than males 30.2% with male to female ratio being 1:2.3. In all of these studies meningiomas were most common in female, with male to female ratio nearly 1:2. Among the 51 patients, in our study most common location of tumor was intracranial 46(90.2%) cases, with tumor located most commonly at convexity of brain 26 (51%) of cases. Akyildiz EU et al 2010 study 20 , intracranial location of tumor was found in 96% of cases. In our study most common clinical feature was headache (75%) followed by vomiting (17%). Haradhan deb nath et al 2009 19 study of 25 cases of meningioma, most common clinical feature was headache (72%) of cases followed by vomiting (60%) of cases. In our study most common histological type of tumor was meningothelial meningioma 37% , psammomatous meningioma 19% and fibroblastic meningioma 16%cases. Joseph wanjeri et al 2011 21 , The commonest cellular subtype were fibroblastic 25.4%, transitional (mixed) 25.4% and meningothelial (syncitial) 22.5%. Haradhan deb nath et al 2009 19 study, most common histological subtype was meningothelial meningioma 32% transitional meningioma 20% and fibroblastic meningioma 20% cases. In all three studies meningothelial meningioma and fibroblastic meningioma were common histological subtype. In our study, WHO GRADE I tumour were 92%, WHO GRADE II tumour were 08% and WHO GRADE III tumour were 0%. Joseph wanjeri et al 2011 21 study found that, According to WHO classification, the benign form (grade I) was the commonest at 94.7%. Grade II (atypical) and grade III (malignant) represented 4% and 1.3% respectively. Akyildiz EU et al 2010 study 20 , WHO GRADE I tumour were 82%, WHO GRADE II tumour were 6%. Our result is consistent with above studies, in all these studies most common WHO GRADE were GRADE I. In our study we found two cases of clear cell meningioma. Both are in age group <20years and one is located in spinal region and other at CP angle, which are common locations for clear cell meningioma. In our study we found 4 cases of WHO GRADE II tumour. Two of them were clear cell meningioma, as clear cell meningioma entity itself put tumour into GRADE II. And out of other two, one was meningothelial meningioma and one was fibroblastic meningioma as both of them show increased mitotic activity, ≥4 mitoses per high power field.
CONCLUSION
Meningiomas occurred more frequently in females than in males with a female to male ratio of 2:1. Adults were more affected than their elderly counterparts. The mean age was 42.6. Majority of Patients presented with complain of headache of longer duration and vomiting. Most meningiomas were at intracranial location. Most intracranial meningiomas occur in the supratentorial compartment. Majority of meningioma are histologically benign with WHO GRADE I and hence curable by surgical resection.
ACKNOWLEDGEMENT
The authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Cushing H. The meningiomas (dural endotheliomas): their source, and favoured seats of origin. Brain 1922;45:282 316.
2. Kleihues P, Burger PC, Scheithauer BW. Histological typing of tumours of the central nervous system. 2nd ed. World Health Organization. Berlin: Springer Verlag, 1993:30.
3. Das A. Tang WY. Smith Dr. meningiomas in Singapore, demographic and biological characteristics. J Neurooncol 2000.47, 153 60.
4. Al Mefty O, Kersh JE, Routh A, Smith RR. The long term side effects of radiation for benign brain tumors in adult. J Neurosurg 1990; 73; 502 512.
5. Christensen HC, Kosteljanetz M, Johanses C. Incidence of gliomas and meningiomas in Denmark. 1943 to 1997. Neurosurgery 2003, 52: 1327 34.
6. Lang FF, Macdonald OK, Fuller GN, DeMonte F. Primary extradural meningiomas. A report on nine cases and review of the literature from the era of computerized tomography scanning. J Neurosurg 2000,93:940 950.
7. Kleihues P, Cavenee WK, International Agency for research on cancer, Pathology and genetics of tumors of nervous systems, Lyons: IARC Press 2000
8. Perry A, Scheithauer BW, Staff ord SL, Lohse CM, Wollan PC. “Malignancy” in meningiomas: a clinicopathologic study of 116 patients, with grading implications. Cancer 1999; 85: 2046–56.
9. Bollag RJ, Vender JR, Sharma S. Anaplastic meningioma: Progression from atypical and chordoid morphotype with morphologic spectral variation at recurrence. Neuropathology 2010;30(3):279 87.
10. Campbell BA, Jhamb A, Maguire JA, Toyota B, Ma R. Meningiomas in 2009: controversies and future challenges. Am J Clin Oncol 2009;32(1):73 5.
11. McGovern SL, Aldape KD, Munsell MF, Mahajan A, Demonte F, Woo SY. A comparison of World Health Organization tumor grades at recurrence in patients with non skull base and skull base meningiomas. J Neurosurg 2010;112(5):925 33.
12. Zorludemir S, Scheithauer BW, Hirose T, Van Houten C, Miller G, Meyer FB. Clear cell meningioma. A clinicopathologic study of a potentially aggressive variant of meningioma. Am J Surg Pathol 1995; 19: 493–505.
13. Couce ME, Aker FV, Scheithauer BW. Chordoid meningioma: a clinicopathologic study of 42 cases. Am J Surg Pathol 2000; 24: 899–905.
14. Tong tong W, Li Juan B, Zhi L, Yang L, Bo Ning L, Quan H. Clear cell meningioma with anaplastic features: case report and review of literature. Pathol Res Pract 2010;206(5):349 54.
15. Maier H, Ofner D, Hittmair A, Kitz K, Budka H. Classic, atypical, and anaplastic meningioma: three histopathological subtypes of clinical relevance. J Neurosurg 1992;77:616 23.
16. Kim EY, Weon YC, Kim ST, et al. Rhabdoid meningioma: clinical features and MR imaging findings in 15 patients. AJNR 2007;28(8):1462 5
. 17. Vranic A, Popovic M, Cor A, Prestor B, Pizem J. Mitotic count, brain invasion, and location are independent predictors of recurrence free survival in primary atypical and malignant meningiomas: a study of 86 patients. Neurosurgery 2010;67(4):1124 32.
18. Kane AJ, Sughrue ME, Rutkowski MJ, et al. Anatomic location is a risk factor for atypical and malignant meningiomas. Cancer 2011;117(6):1272 8.
19. Haradhan Deb Nath, MD mainuddin, MD kmal Uddin, Ehsam mahmood et al, surgical outcome of supratentorial meningioma. A study of 25 cases. JCMCTA 2009;41 44.
20. Alkyildiz EU, Oz B, Comunoglu N, Aki H et al. The relationship between histomorphological characteristics and ki67 proliferation index in meningioma Bratisl Lek listy 2010;111(9) 505 509.
21. Joseph. Wanjeri et al. Histology and clinical pattern of meningiomas at the Kenyatta National Hospital Nairobi, Kenya. A thesis submitted for the award of the degree of master of medicine in neurosurgery, University of Nairobi, 2011.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License