IJCRR - 5(3), February, 2013
Pages: 01-13
Date of Publication: 18-Feb-2013
Print Article
Download XML Download PDF
BIOREMEDIATION OF TEXTILE DYE USING WHITE ROT FUNGI: A REVIEW
Author: G. Roseline Jebapriya, J. Joel Gnanadoss
Category: General Sciences
Abstract:Industrial development worldwide has led to an increase in the amount of wastewater production leading to a considerable decrease in levels and quality of the natural water in the ecosystem. Textile dyes are an important class of pollutants in natural water ecosystem. Textile dyes are molecules designed to impart a permanent colour to textile fabrics. Effluent from textile dyeing units contain large amount of dyes and create an environmental problem, which increase toxicity and decrease the aesthetic value of rivers and lakes. A variety of physio-chemical methods are in use worldwide. However, there is an increasing concern as to their impact in effectively treating textile effluents as they introduce secondary pollutants during the 'remediation' process which are quite costly to run and maintain. Research on biological treatment has offered simple and cost effective ways of bioremediation textile effluents. This review summarizes the efficiency of white rot fungi and their enzymes for the treatment and removal of textile dye containing effluents. The advantages and disadvantages of the various methods are discussed and their efficacies are compared.
Keywords: Textile dye, decolourization, biological methods, white rot fungi.
Full Text:
INTRODUCTION
Increase in population and modernized civilization has led to flourishing of textile industries in India. Textile sector is a complicated industrial chain and high diversity in terms of raw materials, processes, productions and equipment. It is estimated that textile account for 14% of India’s industrial production and around 27% of its export earnings. India is the second largest producer of cotton yarn and silk and third largest producer of cotton and cellulose fiber. The report of the ministry of textiles says the total production of yarn during the year 2003-2004 was 3051 kt. There are about 10,000 garment manufacturers and 2100 bleaching and dyeing industries in India. Majority are concentrated at Tirupur and Karur in Tamil Nadu, Ludiyana in Punjab and Surat in Gujarat. Dyeing is a combined process of bleaching and colouring, which generates huge volumes of wastewaters which results in environmental degradation. More than 100,000 commercial dyes are available to textile industries worldwide with over 700,000 tons of commercial dyes a year being produced (45) .
Impact of textile dyes on the environment
The textile industry accounts for two-thirds of the total dyestuff market (65) and consume large volumes of water and chemicals for wet processing of textiles. The discharges of wastewater are the main cause of the harmful environmental impact of the textile industry. Robinson et al., (67) estimated about 10-15% of textile dyes are discharged into waterways as effluent and effluents discharged from such industries consist of high sodium, chloride, sulphate, hardness and carcinogenic dye ingredients. Effluents from textile industry are characterized by their high visible colour (3000- 4500 units), chemical oxygen demand (800-1600 mg/L), and alkaline pH range of 9-11. They also possess large amount of organic chemicals, low biodegradability and total solids in the range of 6000-7000 mg/L (42). The chemical used in the textile processing are varied in chemical composition, ranging from inorganic compounds to polymers and organic products and depend on the nature of the raw material and product (47) . Major pollution by textile wastewater comes from dyeing and wastewater is characterized by high suspended solids, chemical oxygen demand (COD), heat, colour, acidity and other soluble substances (82). The presence of dyes in aqueous ecosystem reduces sunlight penetration into deeper layers diminishing photosynthetic activity, declines the water quality, lowering the gas solubility which causes sensitive toxic effects on aquatic flora and fauna (51). Therefore, the release of harmful dyes in the environment can be an ecotoxic risk and can affect man through the food chain (78). Among different textile dyes used, azo dyes and nitrated polycyclic aromatic hydrocarbons are two groups of chemicals that are abundant in the environment. They cause rigorous contamination in river and ground water in the surrounding area of dyeing industries (65) . The impact of azo dyes in food industry and their degraded products on human health has caused concern over a number of years. Moreover azo dyes have been linked to human bladder cancer, splenic sarcomas, hepatocarcinomas and nuclear anomalies in experimental animals and chromosomal aberrations in mammalian cells. Some azo dyes induce liver nodules in experimental animals and there are a higher numbers of bladder cancers in dyeing industries workers. Benzidine based azo dyes are widely used in dye manufacturing, textile dyeing, colour paper printing and leather industries. Benzidine has long been recognized as a human urinary bladder carcinogen and tumorigenic in a variety of laboratory animals (23) . The chemicals used in dyeing industries are carcinogenic and mutagenic and the effluents even reduce the rate of seed germination and growth of crop plants (52). The decreased carbohydrate, protein and chlorophyll content of plants indicate the toxic nature of the dye industry effluents. Increased proline concentration was observed in plants exposed to textile dye effluent (2). Algal growth and fishes are not affected by dye concentrations below 1 mg/L. The most toxic dyes for algae and fishes are basic and acid dyes. The chemicals present in the textile industry effluents affect the normal life of animals. Toxic compounds from dye effluent get into aquatic organisms, pass through food chain and ultimately reach man and cause various physiological disorders like hypertension, sporadic fever, renal damage, cramps etc (62) .
Methods used in removal of dyes
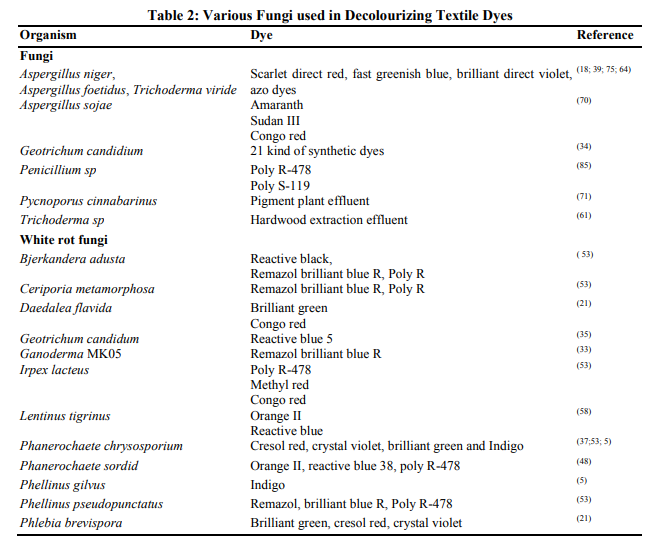
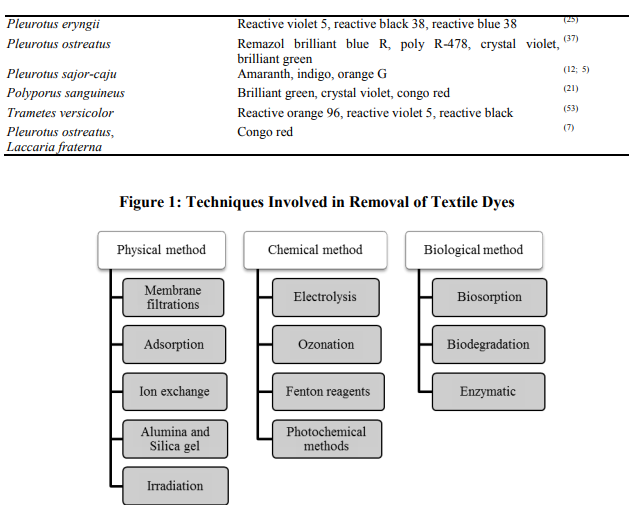
Effluent discharge from textile and dyestuff industries into water bodies and wastewater treatment systems is currently causing significant health concerns to environmental regulatory agencies. Government legislation is increasingly becoming stricter especially in the developed countries, regarding the removal of dyes from industrial effluents. Removal of dyes can occur physically, chemically and biologically (Fig.1). Generally in physical method dyes removed by adsorption, in chemical method chromophore has been modified through chemical reaction, biological method occurs through sorption and enzymatic degradation. Physical method: Adsorption refers to a process where a substance or material is concentrated at a solid surface from its liquid of gaseous surrounding. There are two types of adsorption based on the type of attraction between the solid surface and the adsorbed molecules. If this attraction forces are due to chemical bonding, the process is called chemical adsorption (Chemisorption). Adsorption techniques have gained favor recently due to their efficiency in the removal of pollutants too stable for conventional methods (9). Membrane filtration technique has been widely used for drinking and wastewater treatment. The process of filtration consists of microfiltration, ultrafiltration, nanofiltration and reverse osmosis. Ultrafiltration, nanofiltration and reverse osmosis can be applied as main or post treatment processes for separation, purification and reuse of salts and large molecules including dyes from dyebath effluents and bulk textile processing wastewater (34). Ion exchange techniques are effective in decolourizing cationic and anionic dyes and have not been used in extensively for dye wastewater treatment due to the opinion that ion exchangers cannot accommodate a wide range of dyes (73). Alumina is a synthetic porous crystalline gel available in the form of granules whereas, silica gel is a porous and non crystalline granule of different size prepared by the coagulation of colloidal silicilic acid. Alumina and silica gel have been studied by various workers for the removal of dyes (27). The irradiation process to treat dye containing effluent in a dual tube bubbling reactor requires large volumes of dissolved oxygen for organic substances to be broken down effectively by radiation (40) . Chemical method: The electrochemical technique is very efficient to remove colour from dye wastewater (22). Electrochemical removal of dyes from wastewater is a relatively new process exhibiting efficient colour removal and degradation of recalcitrant pollutants (57). This process is very simple and is based on applying an electric current to wastewater by using sacrificial iron electrodes to produce ferrous hydroxide. These ferrous hydroxides remove soluble and insoluble acid dyes from the effluent. Ozonation is a technology initially used in 70’s and it was carried out by ozone generated from oxygen. Oxidation by ozone is capable of degrading chlorinated hydrocarbons, phenols, pesticides and aromatic hydrocarbons (83). Ozone rapidly decolourize water soluble dyes but with non soluble dyes react much slower. Moreover, textile processing wastewater usually contains other refractory constituents that will react with ozone (50). A solution of hydrogen peroxide and an iron catalyst, known as Fenton’s reagent, is a suitable chemical means of treating wastewaters that are either resistant to biological treatment or poisonous to live biomass. Chemical separation used the action of sorption or bonding to remove dissolved dyes from wastewater and has been shown to be effective in decolourization of both soluble and insoluble dyes (55). Fenton oxidation process can decolorize a wide range of dyes and in comparison to ozonation; the process is relatively cheap and results generally in a larger COD reduction (28). Photochemical or photocatalytic method degrades dye molecules to CO2 and H2O by UV treatment in the presence of H2O2. Degradation is caused by the production of high concentrations of hydroxyl radicals. UV light may be used to activate chemicals such as H2O2, and the rate of dye removal is influenced by the intensity of the UV radiation, pH, dye structure and the dye bath composition. UV light has been tested in combination with H2O2, TiO2, Fenton reagents, O3 and other solid catalysts for the decolourization of dye solution (24). Merits and demerits of physical and chemical methods are listed in Table 1. Biological method: Biological method is generally considered to be the most effective and less energy intensive to removing the bulk of pollutants from wastewater. Different microorganisms have been used for the treatment of various dye effluents. The most important advantage of this method is the low running costs. Biosorption, biodegradation and ligninolytic enzymes have been explored as methods of biological treatments for removal of dye containing effluents. Removal of dye by low cost adsorbents has been extensively reviewed (15; 17). Fungal and bacterial biomass which is a byproduct of fermentations can be used as a cheap source of biosorbent (19) . Knapp et al. (38) reports both bacterial and fungal cells are capability of partial or complete removal of industrial dyes by using adsorption process. However, with some fungi, adsorption is the only decolourization mechanism, but with white rot fungi both adsorption and degradation can occur simultaneously or sequentially. There are many reports on decolourization of dye wastewater by live or dead fungal biomass. However, only limited information is available on interactions between biomass and molecular structure of dyes (18). The major mechanism of removal of dye by dead cells is biosorption, which involves physicochemical interactions (adsorption, deposition, and ion exchange). Hu (26) demonstrated the ability of bacterial cells to adsorb reactive dyes. Zhou and Zimmerman (86) used actinomyces as an adsorbent for decolourization of effluents containing anthroquinone, phalocyanine and azo dyes. Yesilada et al., (84) investigated decolourization of textile dyes by using pellets of the white rot fungus Funalia trogii. The decolourization activity was significantly affected by dye concentration, amount of pellet, temperature, and agitation of the media. Removal of dye effluents by biosorption is still in the research stage. It was not practically approached for treating large volumes of dye effluents due to the disposal of the large volumes of biomass after biosorption Biodegradation of the dye are broadly demonstrated by pure and mixed cultures of bacteria and fungi under aerobic and anaerobic conditions. Aerobic biodegradation process influence by several environmental and nutritional factors such as pH, temperature, amount of oxygen and co-metabolic carbon sources. Bacteria and fungi are the two major groups of microorganisms that have been extensively studied in the treatment of dye wastewater. The enzymes secreted by aerobic bacteria can breakdown the organic compounds. Thus, the isolation of aerobic bacterial strains capable of degrading different dyes has been carried out for more than two decades (63) . Biodegradation of the dye by certain groups of fungi during dye removal has been extensively demonstrated. The degradation and mineralization of dyes is successful by certain white rot fungi (16). Ligninolytic enzymes secreted by white rot fungi bind non-specifically to the substrate and therefore can degrade a wide variety of recalcitrant compounds and even complex mixtures of pollutants including dyes (20). Various enzymes involved in dye decolourization are Laccase, Manganese Peroxidase (MnP), Manganese Independent Peroxidase (MIP), Lignin Peroxidase (LiP), Tyrosinase etc. Production of these enzymes and their activity in biodegradation of dyes is often judged from the appearance of the mycelial mat, which ultimately appears colourless (37). The potential advantages of using enzymes instead of fungal cultures are mainly associated to the following factors: shorter treatment period, operation of high and low concentrations of substrates, absence of delays associated with the lag phase of biomass, reduction in sludge volume and no difficulty of process control (1). However there are several practical limitations in the use of free enzymes such as the high cost associated with production, isolation and purification of enzymes and the short life times of enzymes. To overcome this limitations enzyme immobilization has shown to improve enzyme stability (46). In addition, enzyme immobilization allows enzyme reultilization and continuous operation in bioreactor which is very important for an industrial application of the enzyme. Some of the work done so far using fungi is listed in Table 2.
Dye decolourization by white rot fungi
White rot fungi have been studied for nearly three decades and new species are being shown to decolourize various textile dyes with their lignindegrading enzymes (13). Tien and Kirk (76) reported the first dye decolourization by white rot fungi Phanerochaete chrysosporium. Banat et al., (8) also reported that other white rot such as Hirschioporus larincinus, Inonotus hispidus, Phlebia tremellosa and Coriolus versicolor can be used to decolourise dye effluent. Several other white rot fungus were used for the decolourization of distinct synthetic textile dyes and its effluents (29). Wesenberg et al. (81) surveyed 29 white rot fungi capable of dye decolourization. Since then, several investigators have evaluated the decolourization of commercial dyes by new species (3; 41; 67). Dye decolourization capabilities vary with the fungal species or enzymes (54). Lignin-modifying enzymes play significant roles in dye metabolism by white rot fungus (45) . The ligninolytic enzymes (lignolytic or lignin modifying or lignin degrading enzymes) are extracellularly excreted by the white rot fungi initiating the oxidation of lignin in the extracellular environment of the fungal cell. The ligninolytic enzymes produced by white rot fungi have been categorized into two groups: peroxidases (MnP and LiP) and laccases. Depending on the species and the environmental conditions, white rot fungi produce one or more types of these ligninolytic enzymes. Ligninolytic enzymes are directly involved not only in the degradation of lignin in their natural lignocellulosic substrates but also in the degradation of various xenobiotic compounds (60) including dyes (74). The physiology and production of lignin modifying enzymes (LME) by white-rot fungi has shown great potential in degrading azo dyes and related effluents. Decolourization of dyes by using lignin modifying enzymes were studied extensively example using laccase from Trametes versicolor (81; 32) , Trametes hirsute, Trametes modesta, Sclerotium roysii (54) Laccaria fraterna, Pleurotus ostreatus (6) , Lentinus polychrous (33). LiP from Phanerochaete chrysosporium and MnP from Phanerochaete chrysosporium, Bjerkandera adusta, Pleurotus eryngii (25) .
Mechanism of dye decolourization by white rot fungi
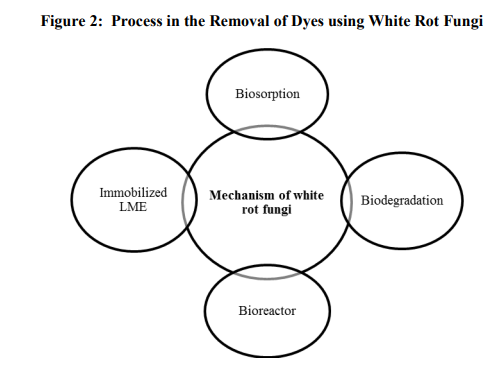
The mechanism of dye decolouization by white rot fungi are mainly categorized into four types namely biosorption, biodegradation, bioreactor and immobilized lignin modified enzymes (Fig.2). Among these mechanisms biodegradation plays a major role in dye decolourization by white rot fungi because they can produce ligninolytic enzymes to mineralize the dyes. However, the relative contributions of laccase, MnP and LiP to the decolourization of dyes may be different for each fungus. Phanerochaete chrysosporium LiP played a major role in dye decolourization. Anthraquinone dye act as a laccase substrates but in the case of azo and indigoid dyes were not the substrates of laccase (80). Pathway of indigo dye degradation by laccase has been demonstrated by Campos et al. (11) and laccases used in their experiment was produced and purified from Trametes hirsuta and Sclerotium rolfsii. Kitwechkun and Khanongnuch (36) studied the decolourization of azo dye (Orange II) by immobilized white rot fungus Coriolus versicolor. Martin et al. (43) screened several fungi for degradation of syringol derivatives of azo dyes possessing either carboxylic or sulphonic group. Trametes versicolor showed the best biodegradation performance and its potential was confirmed by the degradation of differently substituted fungal bioaccessible dyes. Biodegradation assays using mixtures of these bioaccessible dyes were performed to evaluate the possibility of a fungal wastewater treatment for textile industries. Biosorption mechanisms play a major role in decolourization of dyes by fungi. Knapp et al., (37) reported that the extent of colour removal by adsorption was always limited and generally less than 50%. In the case of Coriolus versicolor adsorption accounted for only 5-10% of colour removal (18). However, the adsorption level depends on type of microorganisms and their environmental condition. In some fungi, biosorption is the only decolourization mechanism. With white rot fungi, adsorption does not appear to be the principle mechanism of decolourization. It is likely that adsorption can play apart in the overall process, since prior adsorption to fungal mycelium may serve to bring chromophores onto closer contact with the degradative enzymes, which are often largely associated with cell surface. The biosorbents were reused in three repeated adsorption/desorption cycles without a significant loss in the biosorption capacity. Numerous studies have appeared on various reactor designs for LME production including stirred tanks, packed beds, airlifts, bubble columns, rotating disks, etc., there is a dearth of analogous reports on the use of reactor systems employing white rot fungus for waste treatment. Towards the design of bioreactor systems for decolourization, Zhang et al. (85) used alginateimmobilized basidiomycetes, producer of LiP, MnP and Lac, in several reactor configurations. In addition, they used the three different reactor configuration (continuous packed-bed bioreactor, fed batch fluidized-bed bioreactor and continuous fluidized-bed bioreactor) to design and test for decolorization of an azo dye, Orange II using white rot fungus. It was found that the fed batch fluidized-bed bioreactor was particularly suitable for Orange II decolourization since it showed very high decolorization efficiency. Detailed studies on bioreactor performances are starting to emerge, seeking to extend the capacity of white rot fungus to decolourize dyes in continuous (56) or sequencing batch mode (10) over long periods of time without the need for supplementation of new mycelium and, though a challenge, under nonsterile conditions. Immobilised fungal cells have several advantages over dispersed cells such as simple reuse of the biomass, easier liquid–solid separation and minimal clogging in continuous-flow systems (85) . In addition, immobilised cultures tend to have a higher level of activity and are more resilient to environmental perturbations such as pH, or exposure to toxic chemical concentrations than suspension cultures (72). Schliephake and Lonergan (71) studied the decolourization of the synthetic dye Remazol Brilliant Blue R (RBBR) by the white rot fungus Pycnoporus cinnabarinus grown on nylon web cubes in a 200 L packed bed bioreactor. They found that the dye was rapidly decolourised due to the action of the laccases produced by the fungus. Kapdan et al. (30) studied the decolouration of the textile dye Everzol Turquoise Blue G by the white rot fungus Coriolus versicolor immobilised on discs in a rotating biological contactor (RBC). The white rot fungus Phanerochaete chrysosporium immobilised on cubes of polyurethane foam (PUF) decolourised the polymeric dye Poly R478 (69). Rodriguez Couto et al. (68) also studied the decolourization of the polymeric dye Poly R478 by cultures of Phanerochaete chrysosporium grown on grape seeds, wheat straw and wood shavings under solid-state conditions. The most important criteria for good immobilized enzyme activity are the mechanical properties (rigidity and durability), physical form (granules, sheets, inner tube walls, etc), and resistance to chemical and microbial attacks, material hydrophilicity, price, and availability (49). The stability of a decolourization process depends on the properties of the enzyme support. The investigation by Wang et al (79) on dye decolourization by laccase encapsulated by alginate-gelatin-PEG (polyethylene glycol) confirms that a support material must be chosen to maximize enzyme activity and its mechanical properties and will dictate the configuration of the reactor.
resulting in a huge threat of pollution. It is worthwhile noting that the removal of dyes can be done by various techniques. The removal of dyes described in this review has advantages and disadvantage. Conventional technology was not efficient against the removal of all types of dyes because their molecular structure and chemical properties is complex. Few current technologies such as coagulation, ozonation and activated carbon can efficiently remove only restricted group of dyes. Combination of physical, chemical and biological processes more efficient for textile dye removal but can be expansive. Last two decades vast number of research work has under gone on efficiency of white rot fungus on dye decolourization. Even though various methods to assess decolourization of dyes researchers has been paid more attention on using lignin degrading enzymatic system of white rot fungi for solving the serious environmental pollution problem. Although lignin modifying enzymes gives effective decolourization results this approach is not optimal. So, future studies should be conducted using advanced analytical techniques, to elucidate the catabolic processes involved in the degradation of distinct dye groups by the lignin modifying enzymes of white rot fungus. Advances in molecular techniques can help to create microbes with improved metabolic capabilities by cloning the gene coding for the decolourizing enzymes into suitable expression systems using strong promoters.
References:
1. Akhtar S, Husain Q. Potential applications of immobilized bitter gourd (Momordica charantia) peroxidase in the removal of phenols from polluted water. Chemosphere 2006; 65: 1228–1235.
2. Ameta C, Punjabi PB, Kothari S, Sancheti A. Effect of untreated and photo catalytically treated dyeing industry effluent on growth and biochemical parameters of Allium cepa. Pollut Res 2003; 22: 389.
3. Asgher M, Batool S, Bhatti H, Noreen R, Rahman S, Asad MJ. Laccase mediated decolourization of vat dyes by Coriolus versicolor ibl-04. Int Biodeterior Biodegrad 2008a; 62(4): 465–470.
4. Avlonitis SA, Poulios I, Sotiriou D, Pappas M and Moutesidis K. Simulated cotton dye effluents treatment and reuse by nanofiltration. Desalination 2008; 221: 259– 267.
5. Balan DSL, Monteiro RTR. Decolourization of textile indigo dye by ligninolytic fungi. J Biotechnol 2001; 89: 141–5.
6. Balaraju K, Gnanadoss JJ, Arokiyaraj S, Agastian P, Kaviyarasan V. Production of cellulase and laccase by Laccaria fraterna and Pleurotus ostreatus under submerged and solid state fermentation. The ICFAI Journal of Biotechnology 2007; 1: 23-34.
7. Balaraju K, Gnanadoss JJ, Muthu S, Ignacimuthu S. Decolourization of azodye (congored) by Pleurotus ostreatus and Laccaria fraterna. The ICFAI Journal of Life Sciences 2008; 2: 45-50.
8. Banat IM, Nigam P, Singh D, Marchant R. Microbial decolourization of textile-dyecontaining effluents: a review. Bioresour. Technol 1996; 58: 217-227.
9. Bansal RC, Goyal M. Activated carbon adsorption. London, Taylor and Francis Group, 2005; 351-353.
10. Borchert M, Libra JA. Decolourization of reactive dyes by the white rot fungus Trametes versicolor in sequencing batch reactors. Biotechnology and Bioengineering 2001, 3: 312-321.
11. Campos R, Kandelbauer A, Robra KH, Cavaco-Paulo A, Gubitz GM. Indigo degradation with purified laccases from Trametes hirsute and Sclerotium rolfsii. J. Biotechnology 2001; 89:131-139.
12. Chagas E, Durrant L. Decolourization of azo dyes by Phanerochaete chrysosporium and Pleurotus sajorcaju. Enzyme Microb Technol 2001; 29: 473–477.
13. Champagne PP, Ramsay JA. Contribution of manganese peroxidase and laccase to dye decoloration by Trametes versicolor. Appl Microbiol Biotechnol 2005; 69(3): 276–285.
14. Cheng MM, Ma WH, Li J, Huang YP, Zhao JC. Visible-light-assisted degradation of dye pollutants over Fe(III)- loaded resin in the presence of H2O2 at neutral pH values. Environmental Science Technology 2004; 38: 1569–1575.
15. Chuah TG, Jumasiah A, Azni I, Katayon S, Choong SYT. Rice husk as a potentially lowcost biosorbent for heavy metal and dye removal: an overview. Desalination 2005; 175: 305–16.
16. Coulibaly L, Gourene G, Agathos NS. Utilization of fungi for biotreatment of raw wastewaters. Afr J Biotechnol 2003; 2: 620– 30.
17. Crini C. Non-conventional low-cost adsorbents for dye removal: a review. Bioresour Technol 2006; 97:1061–85.
18. Fu Y. Viraraghavan T. Fungal decolourization of dye wastewaters: A review. Biores. Technol 2001; 79: 251–262.
19. Gazso LG. The key microbial processes in the removal of toxic metals and radionuclides from the environment. CJOEM 2001; 7:178.
20. Ge Y, Yan L, Qinge K. Effect of environment factors on dye decolourization by P. sordid ATCC90872 in a aerated reactor. Process Biochem 2004; 39:1401.
21. Gill PK, Arora DS, Chander M. Biodecolourization of azo and triphenylmethane dyes by Dichomitus squalens and Phlebia sp. J Ind Microbiol Biotech 2002; 28: 201–203.
22. GuptaVK, Jain R, Varshney S. Electrochemical removal of the hazardous dye Reactofix Red 3 BFN from industrial effluents. Journal of Colloidal Interface Science 2007; 312: 292–296.
23. Haley TJ. Benzidine revisited; A review of the literature and its congeners. Clin Toxicol 1975; 8: 13.
24. Hao OJ, Hyunook K, Chiang P. Decolourization of wastewater. Critical reviews in environmental science and technology 2000; 30: 449-505.
25. Heinfling A, Martinez MJ, Martinez AT, Bergbauer M, Szewzyk U. Transformation of industrial dyes by manganese peroxidases from Bjerkandera adusta and Pleurotus eryngii in a manganese-independent reaction. Appl Environ Microbiol 1998; 64(8): 2788– 2793.
26. Hu TL. Kinetics of azoreductase and assessment of toxicity of anaerobic products from azo dyes by Pseudomonas luteola. Water Sci. Technol 2001; 43: 261–269.
27. Huang YH, Hsueh CL, Huang CP, Su LC, Chen CY. Adsorption thermodynamic and kinetic studies of Pb(II) removal from water onto a versatile Al2O3-supported iron oxide. Separation Purification Technology 2007; 55: 23–29.
28. Ince NH, Tezcanli G. Treatability of textile dye-bath effluents by advanced oxidation: Preparation for reuse. Water Sci. Technol 1999; 40:183-190.
29. Jarosz-Wilkolazka A, Kochmanska J, Malarczyk E, Wardas W, Leonowicz A. Fungi and their ability to decolourize azo and anthraquinonic dyes. Enz Microb Technol 2002; 30: 566-572.
30. Kapdan I, Kargia F, McMullan G, Marchant R. Effect of environmental conditions on biological decolorization of textile dyestuff by Coriolus versicolor in a rotating biological contactor. Enzyme Microb Technol 2000; 26(5-6): 381–387.
31. Khammuang S, Sarnthima R. Laccase from spent mushroom compost of Lentinus polychrous Lev. and its potential for remazol brilliant blue R decolourisation. Biotechnology 2007; 6: 408-413.
32. Khammuang S, Sarnthima R. Mediatorassisted rhodamine B decolourization by Tramates versicolor laccase. Pak J Biol Sci 2009;12: 616-623.
33. Khammuang S, Rakrudee S. Laccase activity from fresh fruiting bodies of Ganoderma sp. MK05: Purification and remazol brilliant blue r decolourization. J Biol Sci 2009; 9: 83- 87.
34. Kim SJ, Shoda P. Purification and characterization of a novel peroxidase from Geotrichum candidum dec 1 involved in decolourization of dyes. Appl Environ Microbiol 1999; 65: 1029–1035.
35. Kim TH, Park C, Kim S. Water recycling from desalination and purification process of reactive dye manufacturing industry by combined membrane filtration. J Clean Produc 2005; 13: 779 786.
36. Kitwechkun W, Khanongnuch C. Decolourization of azo dye (OrangeII) by immobilized white-rot fungus Coriolus versicolor RC3. Proceeding of The 15th Annual Meeting of the Thai Society for Biotechnology 2004.
37. Knapp J, Newby P. The microbiological decolorization of an industrial effluent containing a diazo-linked chromophore. Water Res 1995; 29(7): 1807–1809.
38. Knapp JS, Vantoch-Wood EJ, Zhang F. Use of wood-rotting fungi for the decolourization of dyes and industrial effluent. In ‘‘Fungi in Bioremediation’’ (G. M. Gadd, ed.), Cambridge University Press 2001; 242–304.
39. Kousar N, Seshikala D, Singara Charya MA. Decolourization of textile dyes by fungi. Ind J Microbiol 2000; 40: 191–197.
40. Kumar MNVR, Sridhar TR, Bhavani KD, Dutta PK. Trends in color removal from textile mill effluents. Colorage 1998; 40: 25– 34.
41. Levin L, Papinutti L, Forchiassin F. Evaluation of Argentinean white rot fungi for their ability to produce lignin-modifying enzymes and decolourize industrial dyes. Bioresour Technol 2004; 94(2): 169–176.
42. Manu B, Chaudhari S. Anaeobic decolourization of simulated textile wastewater containing azo dyes. Biores Technol 2002; 82, 225–231.
43. Martin MAM, Lima N, Silvestre AJD, Queiroz MJ. Comparative studies of fungal degradation of single or mixed bioaccessible reactive azo dyes. Chemosphere 2003; 52: 967-973.
44. McKay G, Porter JF, Prasad GR. The removal of dye colours from aqueous solutions by adsorption on low-cost materials. Water Air Soil Pollution 1999; 114: 423–438.
45. McMullan G, Meehan C, Conneely A, Kirby N, Robinson T, Nigam P, Banat IM, Marchant R, Smyth WF. Microbial decolourization and degradation of textile dyes. Appl Microbiol Biotechnol 2001; 56: 81–87.
46. Michniewicz A, Ledakowicz S, Ullrich R, Hofrichter M. Kinetics of the enzymatic decolourization of textile dyes by laccase from Cerrena unicolor. Dyes and Pigments 2008; 77(2): 295–302.
47. Mishra G, Tripathy M. A critical review of the treatments for decolourization of textile effluents. Colourage 1993; 40: 35–38.
48. Moreira MT, Mielgo I, Feijo G, Lema JM. Evaluation of different fungal strains in the decolourization of synthetic dyes. Biotechnol Lett 2000; 22: 1499–503.
49. Mosbach R, Koch-Schmidt AC, Mosbach K. Immobilization of enzymes to various acrylic copolymers. Immobilized enzymes, Methods in Enzymology 1976; 44: 53–65.
50. Muthukumar M, Sargunamani D, Senthilkumar M, Selvakumar N. Studies on decolouration, toxicity and the possibility for recycling of acid dye effluents using ozone treatment. Dyes and Pigments 2005; 64: 39- 44.
51. Nilsson R, Nordlinder R, Wass U. Asthma, rhinitis and dermatitis in workers exposed to reactive dyes. British Journal of Industrial Medicine1993; 50: 65-70.
52. Nirmalarani J, Janardhanan K. Effect of south India Viscose factory effluent on seed germination seedling growth and chloroplast pigments content in five varieties of Maize (Zea mays I). Madras Agric J 1988;75: 41.
53. Novotny C, Rawat B, Bhatt M, Patel M, Sasek V, Molitors HP. Capacity of Irpex lacteus and Pleurotus ostreatus for decolourization of chemically different dyes. J Biotechnol 2001; 89: 113–122.
54. Nyanhongo GS, Gomes J, Gubitz GM, Zvauya R, Read J, Steiner W. Decolourization of textile dyes by laccases from a newly isolated strain of Trametes modesta. Water Res 2002; 36(6): 1449–1456.
55. Pak D, Changm W. Decolourizing dye wastewater with low temperature catalytic oxidation. Water Sci Technol 1999; 40: 115– 121.
56. Palma C, Moreira M, Mielgo I, Feijoo G, Lema J. Use of a fungal bioreactor as a pretreatment or post-treatment step for continuous decolourisation of dyes. Water Sci Technol 1999; 40(8): 131–136.
57. Pelegrini R, Peralto-Zamora P, De Andrade AR, Ryers J, Duran N. Electrochemically assisted photocatalytic degradation of reactive dyes. Appl Catal B Environ 1999; 22: 83–90.
58. Pera-Titus M, García-Molina V, Banos MA, Gimenez J, Esplugas S. Degradation of chlorophenols by means of advanced oxidation processes: a general review. App Cat B Environ 2004; 47: 219-256.
59. Pilanee V, Waraporn A, Oncheera P, Jirawate C. Production of ligninolytic enzymes by white rot fungus Datronia sp. KAP10039 and their application for reactive dye removal. International Journal of chemical engineering 2010;10: 162504.
60. Pointing SB. Feasibility of bioremediation by white-rot fungi. Appl Microbiol Biotechnol 2001; 57(1-2): 20–33.
61. Prasad DY, Joyce TW. Colour removal from kraft bleach plant effluents by Trichoderma sp. Tappi J 1991; 74: 165-169.
62. Puvaneswari N, Muthukrishnan J, Gunasekaran P. Toxicity assessment and microbial degradation of azo dyes. Indian Journal of Experimental Biology 2006; 44: 618-626
63. Rai HS, Bhattacharyya MS, Singh J, Bansal TK, Vats P, Banerjee UC. Removal of dyes from the effluent of textile and dyestuff manufacturing industry: a review of emerging techniques with reference to biological treatment. Critical Reviews in Environmental Science and Technology 2005; 35: 219–238.
64. Rajendran R, Sundaram SK, Maheswari KU. Aerobic biodecolourization of mixture of azo dye containing textile effluent using adapted microbial strains. J Environ Sci Technol 2011; 4: 568-578.
65. Riu J, Schonsee I, Barcelo D. Determination of sulfonated azo dyes in ground water and industrial effluent by automated solid-phase extraction followed by capillary electrophoresis/mass spectrometry. J Mass Spect 1998; 33: 653.
66. Robinson T, Nigam PS. Remediation of textile dye waste water using a white-rot fungus Bjerkandera adusta through solidstate fermentation. Appl Biochem Biotechnol 2008; 151(2-3): 618–628.
67. Robinson T, McMullan G, Marchant R, Nigam P. Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Biores Technol 2001; 77: 247- 255
68. Rodriguez Couto S, Lopez E, Sanroman MA. Utilisation of grape seeds for laccase production in solid-state fermentors. Journal of Food Engineering 2006; (74)2: 263–267.
69. Rodriguez Couto S, Rivela I, Munoz M, Sanroman A. Ligninolytic enzyme production and the ability of decolourisation of Poly R-478 in packed-bed bioreactors by Phanerochaete chrysosporium. Bioprocess Biosyst Eng 2000; 23(3): 287–293.
70. Ryu BH, Weon YD. Decolourization of azo dyes by Aspergillus sojae B-10. J Micobiol Biotechnol 1992; 2: 215-219.
71. Schliephake K, Lonergan G, Jones C, Mainwaring D. Decolourisation of a pigment plant effluent by Pycnoporus cinnabarinus in a packed-bed bioreactor. Biotechnology Lett 1993; 15(11): 1185-1188.
72. Shin M, Nguyen T, Ramsay J. Evaluation of support materials for the surface immobilization and decolouration of Amaranth by Trametes versicolor. Appl Microbiol Biotechnol 2002; 60: 218–223.
73. Slokar Y, Majcen Le Marechal A. Methods of decolouration of textile wastewaters. Dyes and Pigments 1998; 37(4): 335–356.
74. Spadaro JT, Gold MH, Renganathan V. Degradation of azo dyes by the lignindegrading fungus Phanerochaete chrysosporium. Appl Environ Microbiol 1992; 58: 2397–2401.
75. Sumathi S, Manju BS. Uptake of reactive textile dyes by Aspergillus foetidus. Enz Microb Technol 2000; 27: 347–355.
76. Tien M, Kirk TK. Lignin-degrading enzyme from the hymenomycete Phanerochaete chrysosporium burds. Science 1983; 221(4611): 661–663.
77. Tieng YP, Sun G. Use of polyvinyl alcohol as a cell entrapment matrix for copper biosorption by yeast cells. Journal of Chemical Technology and Biotechnology 2000; 75: 541–546.
78. Van der Zee FP, Villaverde S. Combined anaerobic aerobic treatment of azo dyes-A short review of bioreactor studies. Water Res 2005; 39: 1425.
79. Wang S. A Comparative study of Fenton and Fenton-like reaction kinetics in decolourisation of wastewater. Dyes and Pigments 2008; 76: 714–720.
80. Wang Y, Yu J. Adsorption and degradation of synthetic dyes on the mycelium of Trametes versicolor. Water Sci Technol 1998; 38: 233–238.
81. Wesenberg D, Kyriakides I, Agathos SN. White-rot fungi and their enzymes for the treatment of industrial dye effluents. Biotechnol Adv 2003; 22(1-2): 161–187.
82. World Bank. Environmental, Health, and Safety Guidelines for Textile Manufacturing. International Finance Corporation, World Bank Group 2007.
83. Xu Y, Leburn RE. Treatment of textile dye plant effluent by nanofiltration membrane. Separ Sci Technol 1999; 34: 2501–2519.
84. Yesilada O, Cing S, Asma D. Decolourization of the textile dye Astrazon Red FBL by Funalia trogii pellets. Biores Technol 2002; 81: 155–157.
85. Zhang F, Knapp J, Tapley K. Development of bioreactor systems for decolourization of Orange II using white rot fungus. Enzyme Microb Technol 1999; 24(1-2): 48–53.
86. Zhou W, Zimmerman W. Decolourization of industrial effluents containing reactive dyes by Actinomycetes. FEMS Microbiol Lett 1993; 107: 157–162.

|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License