IJCRR - 5(16), August, 2013
Pages: 63-70
Date of Publication: 28-Aug-2013
Print Article
Download XML Download PDF
PREDICTIVE ANALYSIS OF SULPHUR REMOVAL BASED ON ITS AS-BENEFICIATED CONTENT AND INPUT CONCENTRATION OF ACETIC ACID DURING LEACHING
Author: T. O. Chime
Category: Technology
Abstract:An attempt has been made to investigate the predictive analysis of desulphurization of as received iron ore using acetic acid. The investigation is based on as beneficiated iron ore content and input concentration of acetic acid. A solution of acetic acid was used for leaching the iron oxide ore and the reaction vessel containing the solution and ore was maintained at a temperature of 300C for 10 minutes. A model was derived and used as a tool for the analysis. The validity of the two-factorial model: γ = 0.6235 \?3 \? 1.1661 \?2 + 0.5327\? + θ2 \? 0.081θ + 0.0005 Was rooted on the expression γ + 0.081θ - 0.0005 = 0.6235 \?3 \? 1.1661 \?2 + 0.5327\? + θ2 where both sides of the expression are correspondingly approximately equal. Statistical analysis of the results of removed sulphur from derived model and experiment for each value of the input concentration of acetic acid shows standard errors of 0.034 and 0.04% respectively. Removed sulphur per unit input concentration of acetic acid as obtained from derived model-predicted and experimental results are 0.0079 and 0.0085 %/M. The maximum deviation of the model-predicted concentration of removed sulphur iron is less than 24%, implying an operational confidence level above 76%. It is inferred that desulphurization of the iron ore is feasible under the prevailing conditions of the experiment and model analysis.
Keywords: Model, Sulphur Removal, Acetic Acid Solution, Iron Ore Leaching.
Full Text:
INTRODUCTION
Failure of steel structures in hot service conditions has awakened a lot of concern and the need to intensify research and development aimed at producing high quality defect free steel materials with admissible sulphur content. The failure of steel serving in very hot enclosure or environment resulting from the presence of a membrane of high concentration of sulphur as iron sulphide in the steel crystal has been studied (Chapman, 1972). Under this condition, the material becomes embrittled and abruptly fails due to hot shortness. Desulphurization mainly takes place at the metal-slag interface but the behavior of sulphur in the bulk of metal is also of greater importance (Nwoye,2007). In the presence of oxygen, sulphur can form oxysulphides, which have low melting point. The intergranular sulphurrich layers of the metal has been found to be soften on heating the ingots before rolling or forging, hence the bonds between the grains are destroyed and cracks can form during plastic working resulting in red shortness (Ednernal,1979). If manganese is present in steel, oxysulphide solidifies with the formation of manganese sulphide and complex sulphides preventing red shortness of the metal in hot working but impairs mechanical properties, since brittle manganous sulphides are located at the grain boundaries (Nwoye,2007). Wide applicability has been given to desulphurization of pig iron outside of or during the blast furnace operation by way of filling a ladle with the pig iron and adding to it as a single charge or in a series of smaller incremental charges, soda in granulated or powder form (Kosmider and Danckert, 1973). Application of lime in various desulphurization methods has been as a mix (lime/magnesium mix) or a lone addition. The researchers observed that usage of "fluidized" lime and magnesium not only decreases explosion risks associated with usage of calcium carbide but reduces the cost of the treatment compared to the process of using calcium carbide. This technique also has less environmental impact. The researchers also explored the potentiality of using another technique for desulphurizing pig iron which involves blow by pneumatic means lime or calcium carbide powder into the inactive pig iron in the ladle by means of an immersion lance. This complements a discontinuous method; where the pig iron to be desulphurized must first be collected in the ladle in order to desulphurize the total ladle content. Such large quantities of pig iron can be desulphurized in most metallurgical plants, however, this method is obviously relatively expensive. Kosmider and Danckert (1973) observed that the degree of desulphurization is dependent: (1) upon the quantity of the desulphurizer (kg per ton of pig iron) (2) upon the initial sulphuric content (3) upon the desired final content of sulphur in the pig iron (4) upon the grain size of the desulphurizer, (5) upon the holding time of the pig iron in the reactor and (4) upon the immersion depth of the blowing lance. Nwoye (2009) carried out gaseous desulphurization process on Agbaja iron ore using powdered potassium trioxochlorate (v) KCLO3 as oxidant. He observed that the oxygen used for the desulphurization process was gotten from the decomposition of KCLO3 . The process suggests that the mechanism of the desulphurization process involves gaseous state interaction between oxygen and sulphur through molecular combination. The results of his work reveal that simultaneous increase in both the percentage of the oxidant added and treatment temperature used are the ideal conditions for the best degree of desulphurization. The present work is to derive a model for predictive analysis of sulphur removal based on as-beneficiated sulphur content and input concentration of acetic acid during leaching of Agbaja (Nigeria) iron ore.
MATERIALS AND METHODS
The iron ore was crushed for the purpose of liberation size. Tyler standard was employed to produce particle size of 250µm. The raw Agbaja iron ore was then sent for chemical analysis using X-ray fluorescence diffraction spectrometer and atomic absorption spectrophotometer.
Scrubbing processing
Scrubbing was carried to remove argillaceous materials from the raw iron ore. The iron ore was poured into a head pan and water was poured to a reasonable level. The ore was washed and the water decanted. This was repeated for five times until clear water was observed. At this point 5g of sodium silicate and 25drops of oleic acid were sprinkled and distributed uniformly throughout the ore.20litres of distilled water was also introduced into the pan and the content mixed thoroughly. After mixing, the argillaceous materials were removed leaving behind the iron ore. The residue was washed thoroughly and was sun dried for 24hours.Some quantities were sent for chemical analysis.
Chemical leaching process
The dried scrubbed iron ore was further pulverized and sieved to obtain a particle size of 10µm. Analar grade of acetic acid solutions of different moles of 0.25M, 0.50M, 0.75M, 1.00M and1.25M were prepared. 50g of constant particle size of 10microns of scrubbed iron ore was poured into the crucible (reactor). 25ml of 0.25M of acetic acid was poured into the crucible containing the iron ore. The mixture was thoroughly mixed to ensure homogeneity. The content was allowed to leach for 10, 20, 30, 40, 50, and 60minutes at 30ºC. At the end of each period the solution was cooled and filtered. The residue was collected, washed to neutrality with distilled water, air dried and oven dried at 150ºC for 24hours. The experiment was repeated for different concentrations and temperatures of 40, 50 and 60ºC. The samples were analyzed using atomic absorption spectrophotometer and X-ray fluorescence diffraction spectrometer.
Model Formulation
Computational analysis of experimental data in Table 1 resulted to Table 3 which indicates that;
Boundary and Initial Condition
Consider iron ore (in a reactor) placed with in acetic acid solution (oxidant).The reactor atmosphere is not contaminated i.e (free of unwanted gases and dusts). Initially, atmospheric levels of oxygen are assumed (due to air in the reactor). Mass of iron oxide ore: (50g), leaching time considered: 10 mins., range of concentration of acetic acid: 0.25-1.25M, constant treatment temperature: 30oC, constant ore grain size; 10µm, were also used. The boundary conditions are: reactor oxygen atmosphere since the reactor was air-tight closed at the bottom and top of the ore particles interacting with the gas phase. At the bottom of the particles, a zero gradient for the gas scalar are assumed and also for the gas phase at the top of the particles. The reduced iron is stationary. The sides of the particles are taken to be symmetries.
RESULTS AND DISCUSSIONS
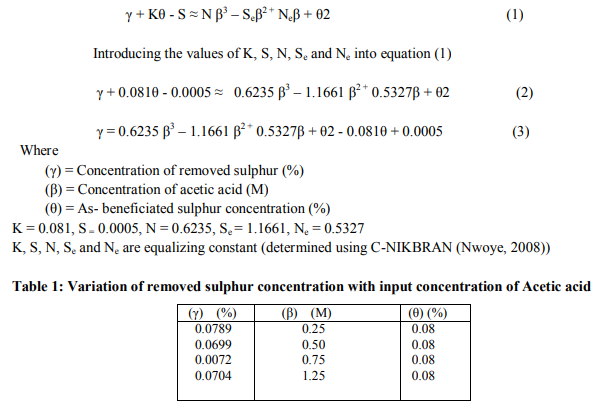
The result of the chemical analysis carried out on the beneficiated iron ore concentrate is presented in Table 1. The table shows that the percentage of total iron (FeT) in the as-beneficiated ore is 52.67%.
Furthermore, the derived model was validated by comparing removed sulphur concentrations predicted by the model and that obtained from the experiment. This was done using various evaluative techniques such as computational, statistical, graphical and deviational analysis.
Computational Analysis
Computational analysis of the experimental and model-predicted removed sulphur concentration was carried out to ascertain the degree of validity of the derived model. This was done by comparing removed sulphur concentration per unit input concentration of acetic acid evaluated from model-predicted results with those from actual experimental results Removed sulphur concentration per unit input concentration of acetic acid γR (%/ M) was calculated from the equation;

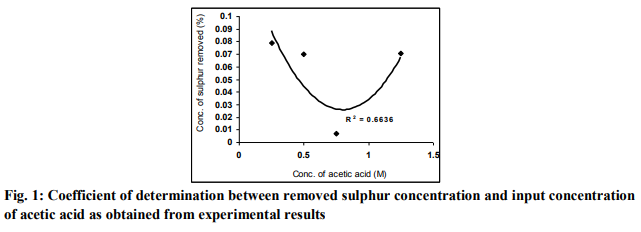
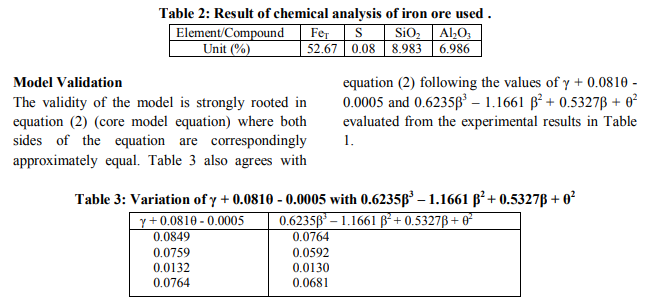
A plot of the removed sulphur concentration against input concentration of acetic acid (as in Fig. 2) using derived model-predicted results gives a slope: - 0.0079 %/ M on substituting the points (0.25, 0.0704) and (1.25, 0.0625) for (β1, γ1) and (β2, γ2) respectively into equation (6). This is the modelpredicted removed sulphur concentration per unit input concentration of acetic acid. It is important to state that the evaluated removed sulphur concentrations per unit input concentration of acetic acid obtained as slopes (from derived model-predicted and experimental results) are just the magnitude of the slopes i.e 0.0079 and 0.0085 respectively. The negative sign preceding the slopes indicates that sulphur removal decreased with increase in the input concentration of acetic acid from 0.25 to 0.75M and then increased at 1.25M. However, the removed sulphur concentration at 1.25M is lower than that obtained at 0.25M. Based on the foregoing, removed sulphur concentrations per unit input concentration of acetic acid as obtained from derived model and experiments are 0.0079 and 0.0085 %/ M respectively.
A comparison of this set of values for removed sulphur concentration (per unit input concentration of acetic acid) also shows proximate agreement and a high degree of validity of the derived model. Statistical Analysis Standard errors (STEYX)
The standard errors (STEYX) in predicting the removed sulphur concentration (using results from derived model and experiment) for each value of the input concentration of acetic acid are 0.034 and 0.04% respectively. The standard error was evaluated using Microsoft Excel version 2003.
Correlation R = √R2 ----------------------- (7)
The correlations between removed sulphur concentration and input concentration of acetic acid as obtained from derived model and experiment was calculated using equation (7) considering the coefficient of determination R2 from Figs. 1 and 2. The evaluated correlations are 0.8904 and 0.8146 respectively. The proximity in these correlations indicates significant reliability and hence validity of the model.
Graphical Analysis
Comparative graphical analysis of Fig. 3 shows very close alignment of the curves from modelpredicted removed sulphur concentration (MoD) and that of the experiment (ExD). The degree of alignment of these curves is indicative of the proximate agreement between both experimental and model-predicted removed sulphur concentration.
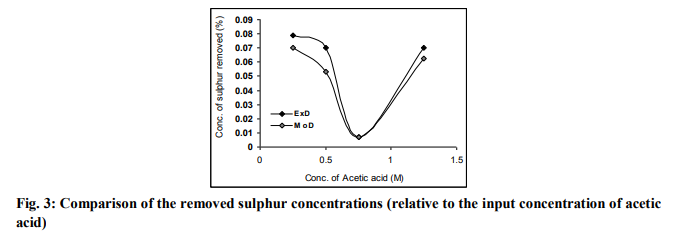
Deviational Analysis
Critical analysis of removed sulphur concentration from the experiment and derived model revealed deviations on the part of the model-predicted values relative to values obtained from the experiment. This is attributed to the fact that the surface properties of the iron ore and the physiochemical interactions between the ore and the acetic acid solution which were found to have played vital roles during the process were not considered during the model formulation. This necessitated the introduction of correction factor, to bring the model-predicted removed sulphur concentration to those of the corresponding experimental values.

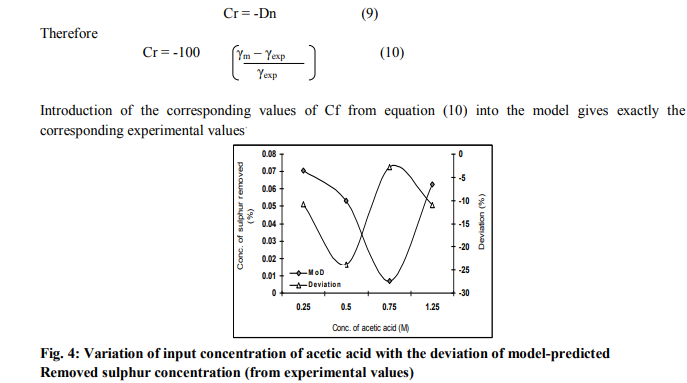
Fig. 4 shows that the maximum deviation of the model-predicted removed sulphur concentration from the corresponding experimental value is less than 24%. The figure show that the least and highest magnitudes of deviation of the modelpredicted removed sulphur concentration (from the corresponding experimental values) are -2.78 and – 23.89 % which corresponds to removed sulphur concentrations: 0.007 and 0.0532 % and input concentrations of acetic acid: 0.75 and 0.5M respectively. Comparative analysis of Figs. 4 and 5 indicates that the orientation of the curve in Figs. 5 is opposite that of the deviation of model-predicted removed sulphur concentration (Fig.4). This is because correction factor is the negative of the deviation as shown in equations (9) and (10).
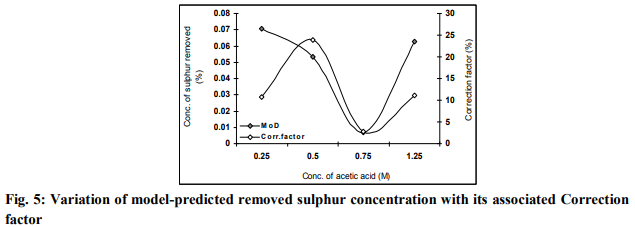
factor
It is believed that the correction factor takes care of the effects of surface properties of the iron ore and the physiochemical interactions between the ore and the acetic acid solution which have played vital roles during the process, but were not considered during the model formulation. Fig. 5
indicates that the least and highest magnitudes of correction factor to the model-predicted removed sulphur concentration are +2.78 and + 23.89%. These correction factors correspond to removed sulphur concentrations: 0.007 and 0.0532% and input concentrations of acetic acid: 0.75 and 0.5M respectively. It is important to state that the deviation of model predicted results from that of the experiment is just the magnitude of the value. The associated sign preceding the value signifies that the deviation is a deficit (negative sign) or surplus (positive sign). CONCLUSION Predictive analysis of sulphur removal during iron ore leaching was carried out based on asbeneficiated sulphur content and input concentration of acetic acid. A model was derived and used as a tool for the analysis. The validity of the two-factorial model was rooted on the expression γ + 0.081θ - 0.0005 = 0.6235 β 3 – 1.1661 β 2 + 0.5327β + θ2 where both sides of the expression are correspondingly approximately equal. Statistical analysis of the results of removed sulphur from derived model and experiment for each value of the input concentration of acetic acid shows standard errors of 0.034 and 0.04% respectively. Removed sulphur per unit input concentration of acetic acid as obtained from derived model-predicted and experimental results are 0.0079 and 0.0085 %/M. The maximum deviation of the model-predicted concentration of removed sulphur is less than 24%, implying an operational confidence level above 76%.
References:
1. Chapman, W. A. P. (1972). Workshop Technology, part 1, 5th edition, Edward Arnold, London, pp. 31-51.
2. Edneral, F. P. (1979). Electrometallurgy of steel and Ferroalloys. MIR publishers, Moscow, 1, pp. 99.
3. Kosmider, H., Danckert, D. (1973). Methods for Desulphurizing Pig Iron. U. S. Patent 3715202, Application No. 05/115296.
4. Nwoye, C. I. (2008). C-NIKBRAN; Data Analytical Memory.
5. Nwoye, C. I. (2009). Process Analysis and Mechanism of Desulphurization of Agbaja Iron Oxide Ore. Journal of Metallurgical and Materials Engineering, 8:27-32.
6. Nwoye, C. I. (2007). Gaseous state Desulphurization of Agbaja Iron Ore Concentrate. Journal of Engineering and Applied Sciences,3:72-75.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License