IJCRR - 5(18), September, 2013
Pages: 35-40
Date of Publication: 25-Sep-2013
Print Article
Download XML Download PDF
COMPARATIVE STUDY OF OXIDANT AND DIETARY ANTIOXIDANTS IN BENIGN AND MALIGNANT BREAST TUMORS- A PILOT STUDY
Author: Shinde Atul, Abhang Subodhini A, Shelgikar Prachi J.
Category: Healthcare
Abstract:Background: The incidence of breast cancer has been increasing in India in the past few years. Oxidative stress is considered to be implicated in the pathophysiology of breast cancers. In normal conditions, a balance between the oxidants and their destruction by the cellular antioxidant system in the human body is maintained. However, any imbalance between the levels of oxidants and antioxidants might cause DNA damage and may lead to tumor development. Though a large number of women are affected with breast tumors, very few studies have been undertaken in India to compare oxidant/ antioxidant status in benign and malignant breast tumors. Therefore the present study was undertaken with following aim. Aim: To compare the oxidative stress and dietary antioxidants in benign and malignant breast tumors. Material and Methods: This study included clinically diagnosed and histopathologically proven 30 each benign and malignant breast tumor patients of stage III and IV as per inclusion criteria. Fresh tumor and fibro adenoma tissues were obtained from patients immediately after surgery. Adjacent normal tissue served as control. The levels of malondialdehyde [MDA]) as a marker of oxidative damage and dietary antioxidants (Vitamin A, E and C) were estimated from the benign, malignant and normal tissues. Results: Significantly increased levels of MDA and decreased levels of dietary antioxidants (Vit E, C, A) were observed in benign and malignant breast tissue as compared to control tissue. A strong negative correlation in MDA Vs all vitamins were found. Conclusion: The results of our study indicate that the imbalance between oxidants and antioxidants leads to oxidative stress which might be playing an important role in tumorogenesis.
Keywords: Oxidative stress, dietary antioxidants, breast tumors
Full Text:
INTRODUCTION
In India, breast cancer is the most common cancer with an estimated 115,251 new diagnoses and 53,592 breast cancer deaths in 2008 according to breast cancer factsheet.[1] Carcinoma of breast continues to be one of the most frequent cancers in women all over the world. Cancer begins in cells, the building blocks that make up all tissues and organs of the body, including the breast. The buildup of extra cells often forms a mass of tissue called a lump or tumor. Breast tumors can be benign (not cancer) or malignant (cancer).
The etiology of breast tumors is multifactorial. Risk factors are early menarche, late menopause, first pregnancy at late age, obesity, oral contraception, hormone replacement therapy, dietary factors, genetic history, nulliparity etc. [2] Apart from these factors, there are some environmental factors that are behind the process of tumorogenesis. One of the most accepted factors is oxidative stress. Cells in tissues and organs are continuously being subjected to oxidative stress and free radicals on a daily basis. Oxidative stress caused by increased free radical generation and/or decreased antioxidant level in the target cells and tissues have been suggested to play an important role in carcinogenesis. Prime targets of free radicals are polyunsaturated fatty acids in cell membrane and their interaction results in lipid peroxidation. As Breast tissue is loaded with lipids they are more susceptible to oxidative insults. Normally, the free radical is counter acted by the cells’ antioxidant system without damage to the cellular DNA. As a woman ages, cellular protection may become less effective and DNA damage is wrought faster than the free radicals can be controlled and damage repaired. [3]
The levels of free radical molecules are controlled by various cellular defense mechanisms, consisting of enzymatic (catalase, glutathione peroxidase, superoxide dismutase) and non-enzymatic (glutathione and dietary vitamins E and C) antioxidants. Antioxidant vitamins can protect against a number of degenerative diseases including cancer. The main antioxidant vitamins are Vitamin C, E and β-carotene. Dietary antioxidant vitamins and retinol have been proposed to be protective against breast cancer on the basis of their ability to inhibit mutagenesis and cell transformation by reducing oxidative DNA damage. [4, 5, 6]
Thus, it is necessary to determine the role of free radicals and dietary antioxidants in tumorogenesis. We therefore undertook this study to examine the extent of lipid peroxidation in terms of MDA and the status of dietary antioxidant vitamins A, E and C in the tissue of benign and malignant breast tumor patients.
MATERIAL AND METHODS
Thirty each clinically diagnosed and histopathologically confirmed benign and malignant breast tumor cases of stage III and IV were selected. The mean age of patients in the study group was 46 years (range 30-55 years). The patients were not using hormones, oral contraceptives and were nonsmokers. Exclusion criteria includes conditions such as diabetes mellitus, liver disease, rheumatoid arthritis or any other prolonged illness, which could have caused conflict with the results of this study. This study was approved by our Internal review Board.
Fresh tumor and fibro adenoma tissues obtained from patients immediately after surgery were blotted on filter paper, weighed (1gm) and placed in cold 0.9% NaCl solution. Adjacent normal tissue (histopathologically proven) from benign and malignant tumors serves as control. The tissues were homogenized under standardized conditions in saline and 20% tissue homogenate was prepared. The supernatant was kept in an ice cold condition until assayed. Lipid peroxidation was estimated by thiobarbituric acid (TBA) reactivity.[7] Malondialdehyde (MDA) an end product of fatty acid peroxidation reacts with TBA to form a colored complex having maximum absorbance at 532 nm. Vitamin E was measured using the spectrophotometric method of Baker and Frank. [8] Vitamin E from tissue was extracted into n-heptane, which reacts with ferric chloride and reduces ferric to ferrous ions. Ferrous ions then forms a red colored complex with 2, 2 dipyridyl which was read at 520 nm. A correction for ß carotene was made after reading vit E at 520 nm. ß carotene was determined in mg% by multiplying absorbance at 460nm by 856 factor. This is the modified method of Quaife et al. [9] Vitamin C was determined by the method of Ayekyaw. [10] Vitamin C from serum reacts with phosphotungstate to give a blue color that has maximum absorbance at 600 nm.
Statistical analysis
After estimation of sample and control for different assay, the data for the biochemical analysis is expressed as mean ± SD. Statistical comparisons were performed by analysis of variance (ANOVA) followed by Duncan’s multiple range tests.
RESULTS
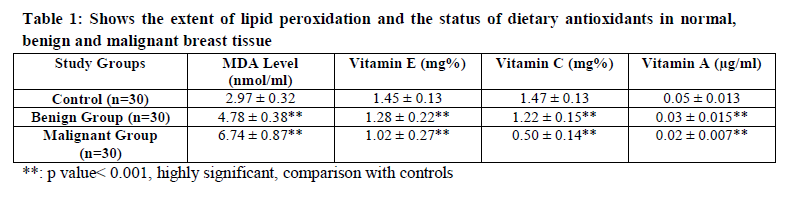
Malondialdehyde (MDA) is a marker of oxidative stress. The levels of MDA in benign tumor were found to be significantly increased (p < 0.001) than control tissue. When MDA levels of control tissue were compared with malignant tissue the rise was still higher (p< 0.001) than the rise observed in benign tissue [Table1].
Levels of Vitamin E in benign tumor tissue were significantly lower (p < 0.001) than control tissue whereas vitamin E status of control tissue when compared with malignant tissue was further decreased as compared to benign tissue [Table1].
There was significant decline (p < 0.001) in the levels of vitamin C in both benign and malignant tissues as compared to normal tissue [Table1].
Highly significant (p < 0.001) decrease in malignant and benign tissues as compared to control tissue were seen in the levels of vitamin A [Table1].
DISCUSSION
Breast cancer is a leading cause of morbidity and mortality in women’s lives and its incidence is on the rise in metropolitan population. [11] Most of the population is ignorant about breast examination. Screening programme is also not very effective in India. This leads to late detection and poor prognosis. This led us to select the cases of stage III and IV. Though a large number of women are affected with breast cancer, very few studies have been undertaken in India on relation between oxidants and dietary antioxidant status among women with the risk of breast cancer.
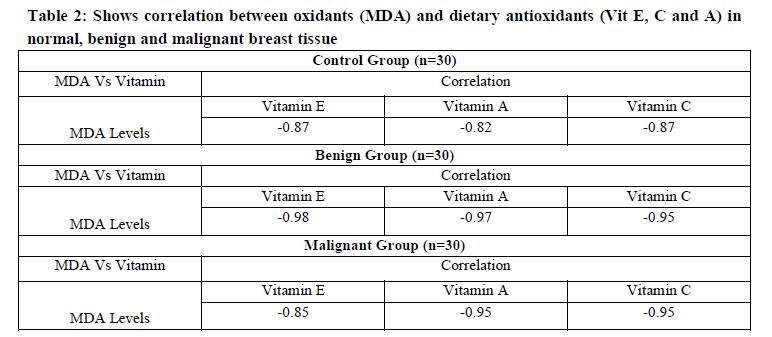
We studied the correlation between oxidative stress and dietary antioxidants in both type of tumors by studying MDA levels and Vit E, C, A levels. A strong negative correlation has been observed between oxidant (MDA) and antioxidants (dietary vitamins) in control, benign and malignant tumor tissue [Table 2].
Development of breast tumor is through damage to the breast epithelium by reactive oxygen species which lead to fibroblast proliferation, epithelial hyperplasia, cellular atypia and breast tumor. [12] The reactive oxygen species (ROS) play an important role in tumor initiation and may influence breast cancer by altering gene expression or by promoting oxidative DNA damage. [13, 14] Oxidative stress caused by increased free radical generation and/or decreased antioxidant level in the target cells and tissues have been suggested to play an important role in carcinogenesis.
The evidence for increased action of oxidants in breast cancer is related with the increase in the level of oxidative damage. In our study, it was observed that there was a higher production of oxygen free radicals in benign and malignant tumors as evidenced by high MDA levels. In malignant tissue this rise is more than benign tissue [Table 1].
Similar results were obtained by Tas et al (2005), Sinha RJ et al (2009) in tissues of benign and malignant breast cancer patients. [15, 11] Even Rajneesh CP et al (2008), Seema Khanna (2012) reported same results in serum sample of breast cancer patients.[12, 16] On the contrary Gonenc et al (2006) reported that serum sample of malignant breast disease had lower MDA levels than benign breast disease but both test groups showed prominent rise than controls. [2]
In healthy persons balance between oxidants and antioxidants is always maintained. But this balance is disturbed in diseased conditions. The probable reason for the elevated level of tissue lipid peroxides observed in breast carcinoma may be due to less availability of antioxidants than required, which leads to the accumulation of lipid peroxides in cancer tissue. Recent reports suggest that there is disturbance of the pro-oxidant/antioxidant balance, resulting from increased free radical production, antioxidant enzyme inactivation or excessive antioxidant consumption, is a causative factor in oxidative damage. [2, 11, 16]
Dietary vitamins are involved in direct elimination of reactive oxygen metabolites in association with antioxidant enzymes. They also act as anti-carcinogens and inhibitors at initiation and promotion/transformation stage in carcinogenesis. [5] In this study we estimated the levels of dietary vitamins in breast tumors as less work was carried out about them by researchers.
In our study we found decreased levels of vitamin E, C and A [Table 1] as compared to controls. This decrease may be due to the overutilization of these antioxidant vitamins in order to quench the free radicals. Vitamin E is used up in eliminating lipid peroxide radicals. This utilized vitamin E is regenerated again by vitamin C. This could also be the cause of reduced vitamin levels observed. [17]
Our results are in good correlation with the results obtained by Singh P (2005) about vitamin E and C in serum of breast cancer patients. [18] Ramaswamy (1996) noted fall in serum vitamin A and C levels in benign breast disease than in controls while in cancer patients, there was a significant trend of lower serum vitamin levels with increasing stage of the disease. [19] While Hou MF (1998) found that, the serum vitamin A levels were not decreased in early breast cancer patients but the serum vitamin A levels were significantly decreased in the metastatic breast cancer group. [20]
Thus, it has been hypothesized that the increased production of ROS along with decreased dietary antioxidant vitamin level might be characteristic of tumour cells [Table2].
CONCLUSION
Oxidative stress is in the foreground of both benign and malignant tumor, but its level is more in malignant as compared to benign tumour. The additional oxidative stress produced in case of malignant tumor may contribute to malignancy. Thus, in conclusion oxidative stress plays an important role in tumorogenesis and its intensity decides the nature of tumor i.e. malignant or benign. The results of our study provide strong evidence regarding the definitive role of dietary vitamins in breast tumours. To prove the role of antioxidant vitamins further study is needed with their supplementation in both study groups.
ACKNOWLEDGEMENT
The authors are grateful to all the subjects for actively participating in this study. Our thanks are also to all laboratory managers and staff for their support during our investigations.
Authors acknowledge the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Authors are grateful to IJCRR editorial board members and IJCRR team of reviewers who have helped to bring quality to this manuscript.
References:
- Breast Cancer Factsheet By Dhillon PK. South Asia Network for Chronic Disease, Public Health Foundation of India.
- Gonenc A, Erten D, Aslan S, Akinci M, Simsek B, Torun M. Lipid peroxidation and antioxidant status in blood and tissue of malignant breast tumor and benign breast disease. Cell Biol Int 2006; 30:376-80.
- Heinz G. Breast cancer: the role of free radicals. The Lancet Oncol 2001; 2:196.
- Kasapovi? J, Peji? S, Todorovi? A, Stojiljkovi? V, Pajovi? SB. Antioxidant status and lipid peroxidation in the blood of breast cancer patients of different ages. Cell Biochemistry and Function 2008; 26: 723-730.
- Borek C. Dietary Antioxidants and Human Cancer. Integrative Cancer Therapies 2004; 3:333-341.
- Sivakumar S, Devaraj N. Enzymatic and nonenzymatic antioxidant status of breast cancer patients in Tamilnadu. IJPBS 2011;2(4):B46- 53.
- Buege and Aust. Microsomal lipid peroxidation. Methods in Enzymol 1978; 105: 302 - 310.
- Baker H, Frank O. Determination of serum ?-tocopherol. In: Gowenlock, McMurry. Varley’s Practical Clinical Biochem; London. 6th ed. 1968: 902-903
- Quaife et al. Determination of serum ß carotene. In: Gowenlock, McMurry. Varley’s Practical Clinical Biochem; London. 6th ed. 1968: 903-903.
- Ayeqaw. A simple colorimetric method for ascorbic acid determination. Clinica Chem Acta 1978; 86: 153-157.
- Sinha RJ, Singh R, Mehrotra S, Singh RK. Implications of free radicals and antioxidant levels in carcinoma of the breast: A never-ending battle for survival. Indian J cancer 2009; 46: 146-50.
- Rajneesh C P, Manimaran A, Sasikala K R, Adaikappan P. Lipid peroxidation and antioxidant status in patients with breast cancer. Singapore Med J 2008; 49: 640-643.
- Halliwell B, Gutteridge JMC Free Radicals in Biology and Medicine. 2007; 4th edition. Oxford: Oxford University Press,UK.
- Feng JF, Lu L, Zeng P, Yang YH, Luo J. Serum total oxidant/antioxidant status and trace element levels in breast cancer patients. Int J Clin Oncol 2011 Oct 5.
- Tas F, Hansel H, Belce A, Ilvan S, Argon A. Oxidative Stress in Breast Cancer. Medical Oncology 2005; 22: 11–15.
- Khanna S, Pande D, Negi R, Karki K. Oxidative stress induced damage in benign and malignant breast diseases: histopathological and biochemical aspects. Journal of Stress Physiology and Biochemistry 2012; 8: 209-214.
- Kline K, Yu W, Sanders B. Vitamin E and Breast Cancer. J Nutr 2004; 134: 3458S–3462S.
- Singh P, Kapil U, Shukla NK, Deo S, Dwivedi SN. Association between breast cancer and vitamin C, vitamin E and selenium levels: results of a case-control study in India. Asian Pac J Cancer Prev 2005; 6: 177-80.
- Ramaswamy G, Krishnamoorthy L. Serum carotene, vitamin A, and vitamin C levels in breast cancer and cancer of the uterine cervix. Nutr Cancer 1996; 25: 173-7.
- Hou MF, Tsai LY, Huang CJ, Huang YS. Serum vitamin A level in breast cancer patients. Kaohsiung J Med Sci 1998; 14:673-8.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License