IJCRR - 5(20), October, 2013
Pages: 125-130
Date of Publication: 02-Nov-2013
Print Article
Download XML Download PDF
STUDY OF EFFECTS OF SOME CATALYSTS IN BIOMASS PYROLYSIS
Author: M. Bardalai, D. K. Mahanta
Category: Technology
Abstract:To convert biomass into gas and liquid for efficient utilization of bioenergy is getting very interest now a day. In this regard several researches have been carried out on the biomass pyrolysis to produce bio-oil. In this work main aim is to see the effect of some catalysts like Al2O3, CaCO3 and ZnO on the biomasses like saw dust and rice husk pyrolysis in terms of thermal degradation as the rate of thermal degradation effects the product of the pyrolysis of the biomass sample. Proximate analysis of the samples have been done to see the ash content, moisture content, volatile matter etc. that contained in the biomass sample. The ultimate analysis is carried out to check the elemental components like C, H, N, O, S etc. using CHN (Carbon, Hydrogen and Nitrogen) analyzer. The Bomb calorimeter is used to determine the calorific value of the biomass sample. The calorific value of both the samples are found to be around 16.32 MJ/kg which is lower than the high grade fossil fuel like anthracite coal having calorific value of 27MJ/kg. In TGA (Thermo gravimetric analysis) the behavior of the thermal degradation of the biomasses with and without the mixing of catalysts where significant differences were found by the help of curves obtained. It has been observed that the total time required for complete vaporization of the biomasses is more as compared to the bare biomass samples on heating although by the mix of some catalysts with some biomasses, there is negligible changes occur in the thermal degradation.
Keywords: biomass, catalyst, proximate analysis, ultimate analysis, thermo gravimetric analysis, pyrolysis.
Full Text:
INTRODUCTION
As the crisis of fossil fuel all over the world is increasing, the utilization of renewable energy and its up gradation is also a big source of research and development. Biomass is one of the sources of renewable energy. In the country like India comprises lots of biomass which are wasted in most of the cases. In place of wasting the biomass it can be used as a good resource of renewable energy directly or by making some conversion. If it is directly used then its efficiency is quite less and some difficulties occur like storing and transportation. So, now days the research has been progressed to convert biomass from solid into gas or liquid in various ways to make it as an effective source of renewable energy. Pyrolysis is the thermo chemical process where biomass is converted into liquid. It is the thermal degradation of solid in the absence of oxygen in the temperature range of 5000C to 8000 C and then condensed the vapor to make it liquid. The up gradation of bio oil produced by pyrolysis is very essential as this oil is not suitable for direct use in engines. In most of the cases it contains more water, more acidic, more oxygen etc. Upgradation can be done in many ways which is already shown by many researchers. Catalytic pyrolysis is one of the processes which can be used to upgrade the bio oil. Every catalyst has some different effects on the biomass conversion process and ultimately on the bio oil. In this work a comparative study of effects of 3 types of catalysts on two different biomasses has been analyzed.
FEEDSTOCKS AND CATALYSTS
In this work two types of feedstock are taken- teak tree (Tectona Grandis) saw dust which is easily found in the powder form in the size of 0.5 to 1mm because the teak tree is mostly cut for the furniture purpose. The collected dust in this work is found in the size of 0.5 to 1 mm. The another feed stock taken here is the rice (Oryza Sativa/ Asian Rice) husk which is also easily available in small or large size rice mill. These are found in the size of 1 to 2mm. Since many works has already been done on saw dust and rice husk pyrolysis, here it is tried to observe the effect of catalyst on these feed stocks. The catalysts taken here for the mixing with the biomasses are – CaCO3, ZnO and Al2O3. The catalysts can be taken at different proportions with the biomass sample.
EXPERIMENTAL METHODLOGY
The proximate and ultimate analysis of the biomass samples before mixing with the catalysts are carried out to determine its characteristics. For proximate analysis a Muffle furnace (vertical type) is used. A hot air oven and a moisture cane are used for heating up to 1100 C to determine the moisture contents. A silica crucible is used to determine the ash content (heating up to 7500C) and volatile matter (heating up to 9000C). The tests are carried out as per the ASTM (American Society for Testing and Materials) standards.
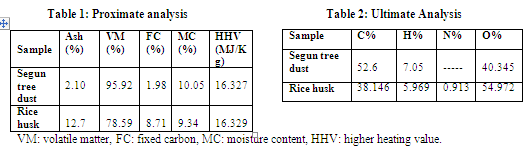
The results of the proximate analysis are shown in the table 1. The heating value, i.e. the HHV (Higher Heating Value or Gross Calorific Value) is calculated by using bomb calorimeter (company: RICO, model: RBC) in ASTM method.
RESULTS AND DISCUSSION
The calorific values of the Segun tree saw dust and rice husk are almost same with the fossil fuel like lignite (coal), the range of calorific value of which is around 16.32 MJ/Kg. These values are small as compared to the high grade coal like anthracite (HHV=27MJ/Kg). From this study it can be observed that both saw dust and rice husk have the potential for pyrolysis oil production. The ash content and fixed carbon in the rice husk is much greater than those of the teak tree saw dust and this leads to high char content in pyrolysis rather than saw dust. The ultimate analyses of the biomass sample are carried out using a CHN (Carbon, Hydrogen and Nitrogen) analyzer and the results are shown in the table-2. The contents of carbon and hydrogen in percentage in the fuel are shown in the table-2. The oxygen content is the difference of the sum of the hydrogen and carbon contents from 100. The nitrogen content in the Segun tree dust is very small, so it is neglected. The sulphur amounts in both the samples are also found to be very negligible.
From the above results shown in the table-2, the empirical formula of the Segun tree saw dust is found as C1.74H2.78O and for the rice husk as CH1.865O1.08. The oxygen content is higher in rice husk than saw dust. As the oxygen content increases, the liquid will be oxygenated and being acidic and it creates corrosion.
TGA ANALYSIS
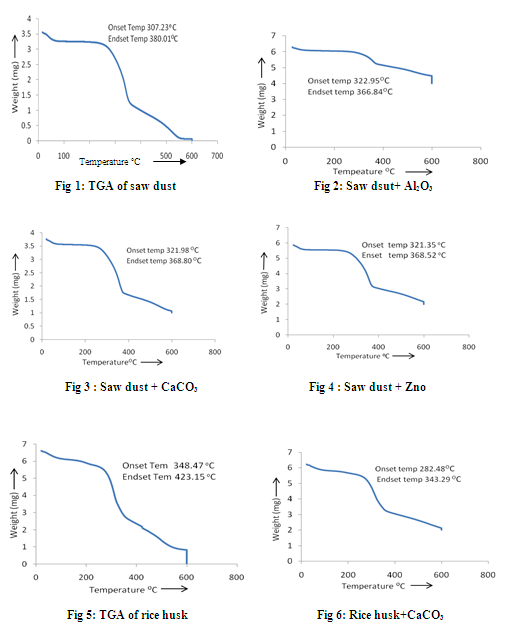
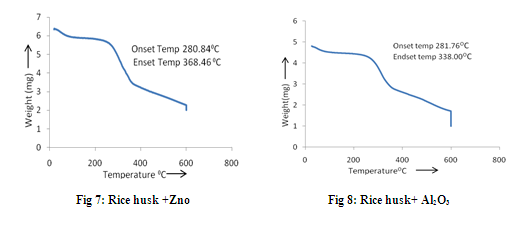
The TGA (Thermo gravimetric Analysis) is the technique, where thermal degradation of a sample can be observed. It shows the degradation of weight with the increase in temperature. The analysis is performed at first with the biomass alone, and then it is performed for the mixture in 1:1 ratio of the bio-mass and catalyst. The TGA experiments were carried out at the heating rate of 10oC/min in the nitrogen atmosphere in TGA analyzer (TGA-50, SHIMADZU). The TGA curves obtained from the analysis for the biomass samples with and without the mixing of catalysts are shown in figures 1 to 8. From the fig. 1 to 4, it is seen that the saw dust is stable upto the temperature of around 3070C, after which the abrupt volatilization starts and this continues upto a temperature of around 3800 C. But with the addition of catalyst, the saw dust mixture remains stable upto some higher temperature than 3070C but the abrupt loss of wight does not lust long unlike the single saw dust sample. In the mixture of saw dust and Al2O3, there is no sudden significant change has been observed. The volatilization is being gradual as the increase of temperature. It is also observed that in the mixture of saw dust and ZnO, the sample does not vaporize completely. In case of rice husk the sample remains stable up to the temperature of around 3480C and the sudden decrease in temperature starts from it and continues up to the temperature of around 4230C. But when it is mixed with catalysts its temperature of stability becomes lower. The vaporization of the sample starts from around 280 to 2820 C when it is mixed with the catalysts. Again in case of rice husk the complete volatilization occurs at around 6000C but when it is mixed with catalysts, it requires some higher temperature for complete vaporization. When the saw dust is mixed with the catalyst Al2O3 there is no such drastic change of weight has been observed as the increase in temperature, the loss of weight is very slow. So the rate of vaporization in saw dust with Al2O3 is slow up to the temperature 6000C. So the pyrolysis is not effective in this mixture up to 6000 C. Saw dust with CaCO3 and ZnO shows a very drastic loss of weight within a very short variation of temperature i.e. within 470 C. Within this temperature difference the catalyst CaCO3 reduces almost 45% weight and the catalyst ZnO reduces around 50% weight where as in case of bare saw dust around 86% weight is lost within a temperature difference of 870 C. In case of bare rice husk the maximum weight loss i.e. around 54% weight loss occurs within the temperature difference 750C. Rice husk with Al2O3 get its 50% weight loss within the temperature difference of 570C. Around 45% weight is lost during the temperature difference of 610 C when the rice husk is mixed with CaCO3 and 45% weight is lost in the temperature difference of 880C in case of rice husk and ZnO mixture.
CONCLUSION
From this analysis the thermal behavior or rate of volatilization of the biomass sample with some specific catalysts has been studied. It is found that the rate of decomposition and devolatilisation with the mix up of different catalysts are quite different. It has also been seen that with mixing of catalyst the total time required to make complete vaporization is more as compared to the bare biomass sample. So definitely it will have a significant effect on the pyrolysis process of the biomass which leads to the quality of the bio –oil produced from it.
References:
- Fatih Demirbus, M. and Balat, M., 2007, Biomass pyrolysis for liquid fuels and chemicals, A review Journal of Scientific & Industrial Research, 66, 797-804.
- Xi-feng, Z., Ji-lu, Z., Qing-xing, G. and Qing-shi, Z., 2006, Pyrolysis of rice husk and sawdust for liquid fuel. Environmental science, 18, 392-396.
- Ji-lu, Z., 2007, Bio-oil from fast pyrolysis of rice husk: Yields and related properties and improvement of the pyrolysis system. Journal ofAnalytical and Applied Pyrolysis, 80, 30-35.
- Vamvuka, D., 2011, Bio-oil, soild and gaseous biofuels from biomass pyrolysis processes- An overview, International Journal of Energy Researc, 35, 835-862.
- Su Heo, H., Ju Park,H., Kwon Park,Y., Ryu,C., Jin Suh,D., WoongSuh, Y., HeongYim, J. and Kim, S., 2010, Bio-oil Production from fast pyrolysis of waste furniture saw dust in a fluidized bed, Bioresource Technology,101, S91-S96.
- Elyounssi, K., Blin, J. and Halim, M., 2010, High yield Charcoal production by two-step pyrolysis. Journal of Analytical and Applied Pyrolysis, 87, 138-143.
- Chouchene, A., BesmaKhiari, M., Trouve, G. and Zagrouba, F., 2010, Study on the emission mechanism during devolatlization/ char oxidation and direct oxidation of olive solid waste in a fixed bed reactor, Journal of Analytical and Applied Pyrolysis, 87, 168-174.
- Tsai, W., Lee, M. and Chang, Y., 2007, Fast pyrolysis of rice husk: produst yield and compositions. Bioresource Technology, 98, 22-28.
- Torii, C., Reinikenen, M., Lindfors,C., Fabbri, D., Oasmaa, A. and Kuoppala, E., 2008, Investigation on catalytic pyrolysis of pine saw dust: Catalytic screening by Py-GC-MIP-AED., Journal of Analytical and Applied Pyrolysis, 88, 7-13.
- Mohanty, P., Pant, K., Naik, S, Das, L. and Vasudevan, P., 2011, Fuel production from Biomass: Indian perspective for pyrolysis oil, Journal of Scientific and Industrial Research, 70, 668-674.
- Sarma, R.K., Bakshi N.N., 1993, Catalytic upgrading of pyrolysis oil, Energy and Fuel, 7, 306-314.
- Jun, W., Mingxu, Z., Mingqiang, C., Manfei, N., Suping, Z., Zhengwei, R. and Yongjie, Y., 2006, Catalytic effects of six inorganic compounds on pyrolysis of 3 kinds of biomass, thermochimica Acta, 444, 110-114.
- French, R. and Czernik, S., 2010, Catalytic pyrolysis of biomass for biofuel production, Fuel Processing Technology, 91, 25-32.
- Wildschut, J., Mahfud, F. H., Venderbosch, R. H. and Heeres, H.J., 2009, Hydrotreatment of fast pyrolysis oil using heterogeneous noble- metal catalysts, Ind. Eng. Chem. Res., 48, 10324-10334.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License