IJCRR - 9(4), February, 2017
Pages: 01-07
Date of Publication: 20-Feb-2017
Print Article
Download XML Download PDF
SYNTHESIS, CHARACTERIZATION AND EVALUATION OF ANTIBACTERIAL ACTIVITY OF COPPER(II) CHELATES OF 3-NITRO-1,5-DIARYLFORMAZANS
Author: Manavjot Kaur1, Nitika2, Rajeev Sharma2
Category: General Sciences
Abstract:Copper(II) chelates of 3-nitro-1,5-diarylformazans have been synthesized by reacting aqueous solution of copper(II) acetate tetrahydrate with methanolic solution of the ligand. The so synthesized copper(II) chelates of 3-nitro-1,5-diarylformazans were purified, crystallized and characterized on the basis of elemental analysis, IR, Electronic and EPR spectral studies, and magnetic susceptibility measurements. The studies reveal that these copper(II) chelates of 3-nitro-1,5-diarylformazans characterized as bis-chelates having 1:2 stoichiometry, are paramagnetic in character and most probably have a symmetrical six membered chelate ring structure. The anti-bacterial activity of these bis(3-nitro-1,5-diarylformazanato)copper(II) chelates has been evaluated against Escherichia coli and Bacillus subtilisby disc diffusion method and compared with standard Amoxycillin and they are found to possess potent anti-bacterial activity against selected bacterial strains.
Keywords: 3-nitro-1, 5-diarylformazan, Copper(II), Chelates, Anti-microbial activity
Full Text:
INTRODUCTION
3-nitro-1,5-diarylformazan ligands react with copper(II) acetate tetrahydrate in 2:1 molar ratio in methanol to yield bis(3-nitro-1,5-diarylformazan) copper(II)chelates. These compounds have been characterised by elemental analysis, IR, EPR, electronic spectral data and magnetic measurements. The studies suggest that the copper(II) chelates are paramagnetic in character and probably contain planar symmetric six membered chelate rings. The copper(II) chelates of formazans are generally prepared by reacting copper(II) acetate[1-8] or copper(II) chloride[9] with hot alcoholic solution of formazan[2-9]. There are reports in literature that certain
3-acetyl-1,5-diarylformazans[10], 3-benzyl-1,5-diarylformazans[11] and 3-ethoxycarbonyl-1,5-diarylformazans[10] react with copper(II) acetate in ethanol to give copper(II) formazan complex having 1:1 stoicheiometry.
The copper(I) complex of 1,3,5-triphenylformazan has structure I, and thus the first stage in this reaction is the oxidation of formazan to tetrazolium salt by cupric ion. Subsequently, 1,3,5-triphenylformazan reacts with cuprous ion to give a copper(I) complex I, having 2:1stoicheiometry with a constitution comprising one molecule of1,3,5-triphenylformazan and one molecule of deprotonated 1,3,5-triphenylformazan, i.e. a formazylanion and one copper(I)ion.
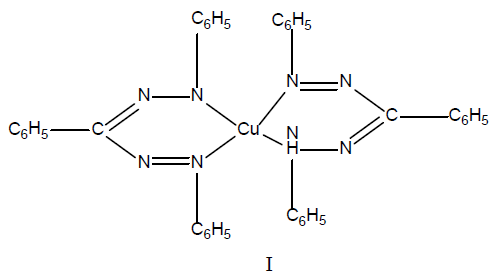
The behavior of 1,5-diarylformazans having acetyl-, benzyl- or an ethoxycarbonyl group in the3-position, with cupric ions contrasts with that of a1,3,5-triphenylformazans that the former are reported to give copper(II) complex II, having a 2:1 stoicheiometry under comparable conditions.
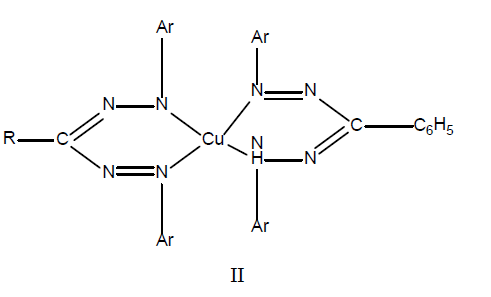
It is a well known fact that very often the reaction between copper(I) ions and 3-arylformazan results in the formation of a mixture of copper(II) and copper(I) formazan chelates, it was thought worthwhile to investigate whether 3-nitro-l,5-diarylformazans also behave in a similar manner or not. It was also aimed to identify the products of the reaction of copper(II) with 3-nitro-1,5-diarylformazans on the basis of elemental analysis, IR, EPR, electronic spectral data and magnetic measurements. Gilroy (2008)[12] prepared complexes of (Fe3+, Co3+, Ni2+ and Pd2+) metals and used 3-substituted diphenyl formazans (3-cyano and 3-nitro formazan) as ligands. Nitika et al [13] prepared 3-nitroformazans by reacting diazotized aromatic amine with nitromethane in cold in an alkaline medium and reported them to be intra-molecularly hydrogen bonded having strongly chelated and a symmetrical structure (III). Balt et al[14] reported kinetic study of complex formation between copper(II) and 1-(2-hydroxyphenyl)-3,5-diphenylformazan in an ammoniacal ethanol-water mixture has brought evidence for a stepwise coordination of the tridentate ligand to the metal ion.
Search for new metallic species with enhanced biological applications is ongoing. Among the metal ions, copper, nickel, cobalt and zinc complexes have proved to be excellent candidates. Copper complexes have shown remarkable efficiency in antioxidant[15], DNA-binding and anti-cancer studies[16-17].
In the present work an attempt has been made to synthesize copper(II) chelates of 3-nitro-1,5-diarylformazans. The so obtained chelates were characterized on the basis of elemental analysis, IR, electronic and EPR spectral studies and magnetic measurement studies. The anti-microbial activities of these copper(II) chelates have been evaluated by studying the zone of inhibition against test organisms E. coli and B. subtilis by Agar Well Diffusion method and compared with standard Amoxycillin.
MATERIAL AND METHODS
Synthesis of copper(II) complexes of 3-nitro-1,5-diarylformazans:
The method of preparation of one of the representative copper(II) complex of 3-nitro-1,5-diarylformazansis given below:
Copper(II)acetate tetrahydrate (200 mg) was dissolved in minimum quantity of distilled water. Solution of the ligand, 3-nitro-1,5-di-p-bromo-phenylformazan(700 mg in 50 ml of methanol) was added to this. The mixture was kept undisturbed for 3-4 hours and then refluxed on water bath for 2 hours. After that the solution was concentrated and cooled by adding a few ml of distilled water. The reddish brown precipitates formed were filtered, washed with distilled water and dried. The crude complex was then column chromatographed using benzene as eluent. The brown fraction which eluted out first was collected, concentrated and re-crystallized from chloroform-hexane. The gummy impurities remained adhered to the column and could not be moved by using methanol as eluent. It was found difficult to crystallize these gummy products but these showed a negative test for copper.
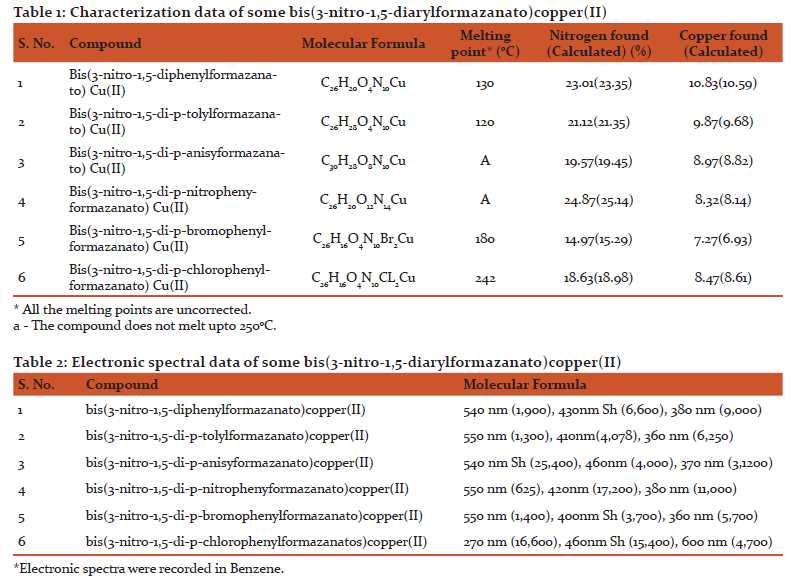
Nitrogen in these copper(II)chelates was estimated by Kjeldahl's method[18] and copper was estimated iodometrically[19].The results are given in Table 1.
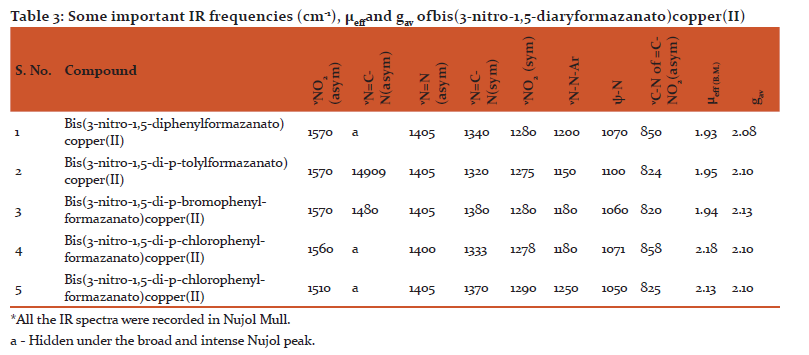
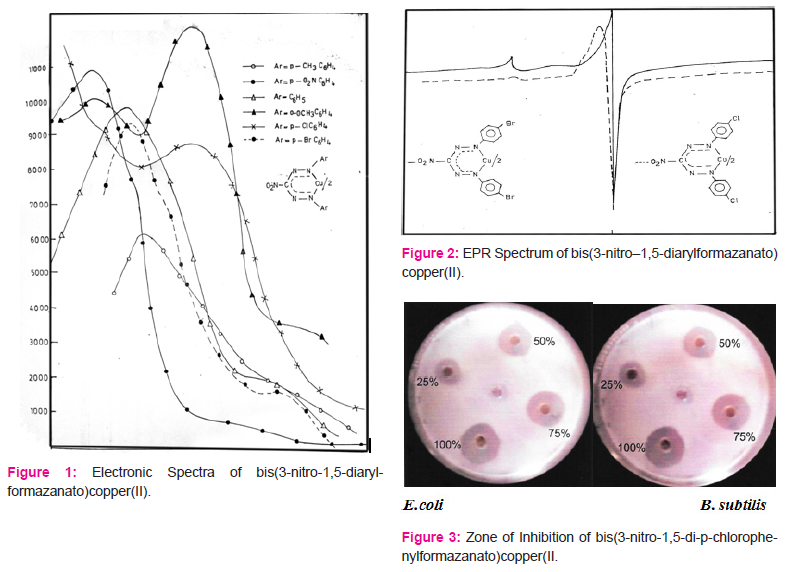
The IR spectra of these copper(II) chelates were recorded and the results are as given in Table2. EPR spectra of powdered samples of the complexes were recorded on JES-FE 3XG EPR spectrometer operating at X-band frequency (9.44 GHz), at room temperature. The magnetic susceptibility measurements for the bis(3-nitro-1,5-diarylformazanato)copper(II) complexes were made on finely powdered samples using magnetic susceptibility equipment based on Evans method and manufactured by Johnson Matthey Catalytic systems division standard procedure. µeff values calculated are reported in Table 2.
EVALUATION OF ANTI-MICROBIAL ACTIVITY OF COPPER(II)CHELATES
Anti-microbial activity was studied by disc diffusion method against Escherichia coli and Bacillus subtilis, and was compared with the standard Amoxycillin.
RESULTSAND DISCUSSION
Elemental Analysis and Spectral Studies
The elemental analysis of copper(II) chelatesof 3-nitroformazans shows that they have 1:2 stoichiometry. The lack of NH absorption band (2900-3200 cm-1) in their IR spectra as compared to those of ligands reveal that these complexes are formed by the replacement of imine hydrogen atom of formazan ring by copper(II). The bis(3-nitro-1,5-diarylformazan)copper(II) complexes are neutral and aresoluble in organic solvents. Therefore, they are not Copper(I) chelates of 3-nitro-1,5-diarylformazans. Thus, it appears that in case of 3-nitro-1,5-diarylformazans only copper(II) chelates and not a mixture of copper(II) and copper(I) chelates are formed.
Having established the composition of reaction between copper(II) and 3-nitro-1,5-diarylformazans, effort was made to establish the structure of these chelates. TheνNO2(asym) and νNO2(sym) appear approximately in the samerange in the IR of these copper(II) chelates as compared to νNO2(asym) and νNO2(sym) of the corresponding ligands. Had the-NO2 group been involved in coordination with copper(II), it would lead to the linkage of the type, III or IV.
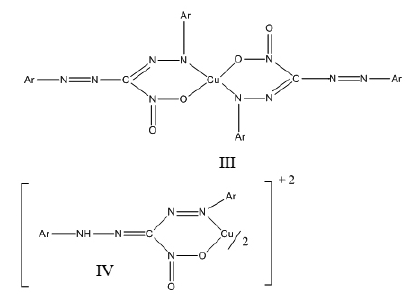
In which event, the νNO2(asym) and νNO2(sym) which appear in the IR of the ligands in the range 1570-1520 cm-1 and1290-1250 cm-1 respectively would disappear and two new absorption bands in the range 1390-1360 cm-1 and 1110-1050cm-1characteristic of metal-nitrito type of linkage would have appeared, as very strong bands. No such change is observed in the IR of bis(3-nitro-1,5-diarylformazanato)copper(II) chelates. There is only a slight decrease in νN=N(sym),νN=N-C-N(sym) and νN-N=Ar of the free ligands as compared to the complexes as expected because the coordination of copper(II) with formazyl group is bound to decrease these frequencies slightly. On the basis of above IR data, the following two structures V and VI are likely.
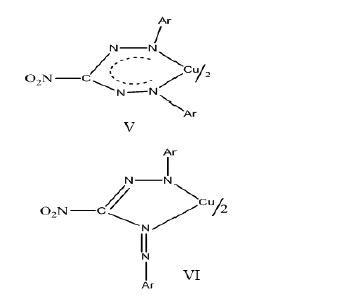
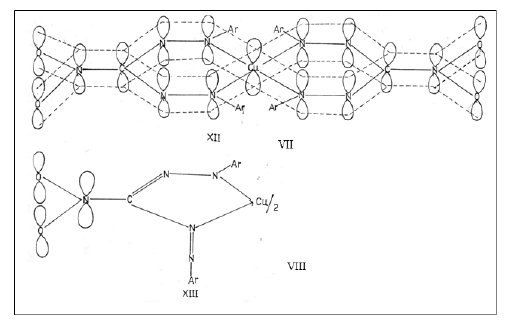
It is obvious from above structures VII and VIII that the stabilisation through π-electron delocalisation will be more predominant in a planar symmetric six membered ring structure, VII as compared to unsymmetric five membered ring structure VIII. This extensive π-electron delocalization in a symmetric six membered structure VII tends to favour aplanar configuration as in case of bis(1,3,5-triphenylformazan)Copper(II), bis(1,3-diphenyl-5-p-tolylformazan)Copper(II),bis(1,3-diphenyl-5-p-chlorophenylformazan)Copper(II) and bis-(1,3-diphenyl-5-o-carboxyphenylformazan)Copper(II)[28]. Also, the ring strain in five membered structure VIII, is more than in the six membered structure VII. This fact also favours a six membered structure VII in these bis(3-nitro-1,5-diarylformazan)Copper(II).
Consideration of both of these factors as mentioned above led Ermakova et al[29] to believe that bis(formazan)Copper(II) complexes have a planar symmetric sixmembered structure II,(where Ar'=p-tolyl, R=Ar=C6H5; Ar'=p-chlorophenyl, R=Ar=C6H5; Ar'=o-carboxyphenyl, R=Ar-C6H5).This was later on confirmed by X-ray studies[30].
Similar very extensive π-electron delocalisation exists in case of bis(3-nitro-1, 5-diarylformazan)Copper(II). The µeff. in case of bis(3-nitro-1,5-diarylformazan)Copper(II) complexes also fall in the range 1.8-2.23 BMas in case of bis(3-arylformazan)Copper(II) complexes (TABLE 3). Hathaway[28] has reported that in practice moments of magnetically dilute compounds of Copper(II) are in the range1.8-2.23 BM with compounds whose geometry approaches octahedral having moment at the lower end and those with geometries approaching tetrahedral having moments at the higher end, but their measurements cannot be used diagnostically with safety unless supported by other evidences. The ‘gavvalues calculated for bis(3-nitro-1,5-diarylformazanato)Copper(II) complexes reported in Table 2 are consistent with the planar structure of these Copper(II) complexes. Therefore, a planar six membered symmetric ring structure V is indicated to be the structure of bis(3-nitro-1,5-diarylformazanato)copper(II) complexes. However, this is merely a conjecture, unless X-ray studies are carried out.
Results of study of antimicrobial activity of copper(II) chelates of 3-nitro-1,5-diarylformazans
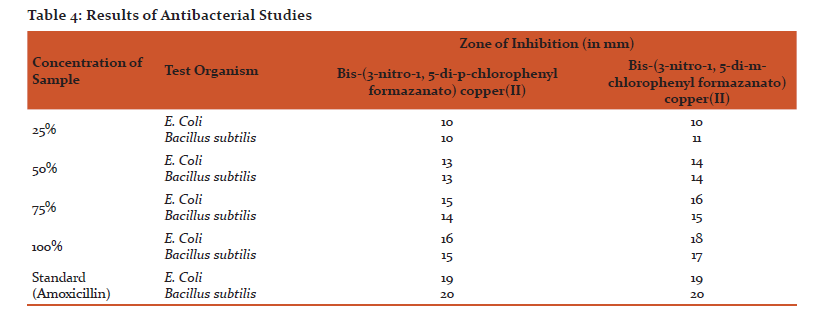
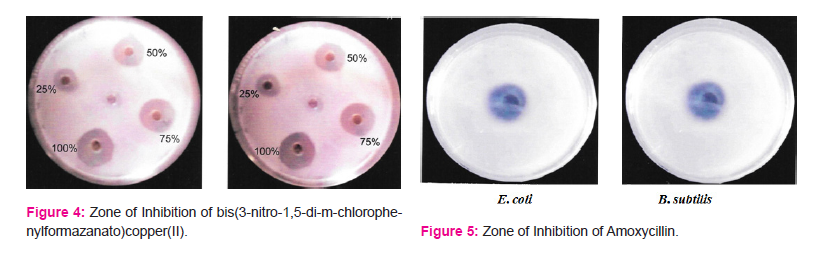
Different compounds were studied for zone of inhibition against the test organism E.coli and Bacillus subtilisusing Well diffusion method. The maximum activity was observed at 100% concentration of different compounds and standard Amoxycillin. The zone of inhibition of compounds is Table 4.
The study reveals that bis(3-nitro-1,5-diarylformazan)copper(II) chelates possess promising anti-microbial activity against E. coli and B. subtilis.
CONCLUSIONS
From the above results it is concluded that
An attempt has been made to synthesize bis(3-nitro-1,5-diarylformazanato)copper(II) complexes by reacting methanolic solution of 3-nitro-1,5-diarylformazan with aqueous solution of copper(II)acetate tetrahydrate. These chelates were found to have stoichiometry 1:2 and paramagnetic in character. These bis(3-nitro-1,5-diarylformazanato)copper(II) chelates have µeff. between 1.8-2.23 BM. All the bis(3-nitro-1,5-diarylformazanato)copper(II) complexes reported here probably have a planar six membered symmetric ring structure, as supported by their µeff.and 'gav' values. The antimicrobial activity of these copper chelates was evaluated against E. coli and Bacillus subtilis by Agar well diffusion method. The observations show that the zones of inhibition for bis-(3-nitro-1,5-di-p-chlorophenylformazanato)copper(II) against E. coli and Bacillus subtilis are good and are quite comparable to standard Amoxycillin.
References:
- Hunter, L.; Roberts, C.B.; J.Am.Chem.Soc., 823(1941).
- Wizinger R.; Bird V.;Helv. Chim. Acta, 32, 901(1949).
- Seyhan, M.; Rev. Fac. Sci. Univ. Istanbul, 17A, 182 (1952).
- Ibid. 299 (1952).
- Wizinger R.; Herzog, H.; Helv. Chim. Acta, 36, 531(1953).
- Seyhan M.; Fernlius, W.C.; Chem. Bet., 89, 2482(1956).
- Hirsch, B.; Ann., 637, 137(1960).
- Petrovanu, M.; Borcea, A.; AnaleleStiint Univ.A. I. Cuza" Jasi, Sect I[N.S], 4, 157(1958); C.A. 55,452i.
- Lipunova, G.N.; Krylov E.I.; Bednyagina, N.P.; Sharov, V.A.;Zh. Obsch. Khim,39(6), 1293(1964).
- Hirsch, B.; Ann., 637, 137(1960).
- Hirsch,B.; Jauer, E.A.; Ann., 682, 99(1965).
- Gilroy JP; The design, synthesis and Chemistry of stable verdazyl radicals and their precursors; Thesis for Ph. D, University of Victoria; 2008
- Nitika, Kumar S and Sharma R; Chem Sci Trans; 2014; 3(2); 670-675
- Balt S; Meuldijk J; and Renkema WE; Inorg Chim Acta. 1977; 22:161-168
- Ho, Y.P.; Au-Yeung, S.C.; Med Res Rev. 23,633–655(2003).
- Marzano, C.; Pellei, M.; Tisato, F.; Santini, C.; Agents Med. Chem., 9,185 211(2014).
- Ruiz-Azuara, L.; Bravo-Gomez, M.E.; Curr. Med. Chem. 17, 3606–3615(2010).
- Steyermark,A.I.;Quantitative Organic Micro Analysis, Academic Press, New York and London, Edition 11, p.209(1961).
- Jeffery, G.H.; Bassett, J.; Mendham J.; Denney, R.C.;Vogel's Text Book of Quantitative Chemical Analysis,5th Edition,p.393(1989).
- Koyama, H.; Saito Y.; Kuroya, H.; J. Inst. Polytechn. Osaka City Univ., 4, 43(1953).
- Bevan, J.A.;Graddon,D.P.; McConnell, J.F. Nature, 199, 373(1963).
- Figgis, B.N.; Harris, C.M.;J. Chem. Soc., 85(1959).
- Adams, S.G.;Tyson, G.N.;J. Am. Chem. Soc., 62L 1228(1940).
- Harris, C.M.;Kokot, E.; Lenzer, S.L.; Nature, 196, A71(1962).
- Gill, N.S.; Nyholm, R.S.; J.Chem. Soc., 3977(1959).
- Lyons, F.; Martin,K.V.;J.Am. Chem.Soc., 79, 1273(1959).
- Ermakova, M.I.; Krylov,E.E.; Postovskii, I.Ya. Zh;Obshchei.Khim, 30, 849(1960).
- Hathaway, B.J.;Stereochemistry and Electronic Propertiesof the Copper(II) Ion, Essays in Chem., 2, Press London, p.61-92(1971).
- Ibid. 299 (1952).
- Budesinsky, B.W.; Svec.J.; Inorg. Chem., 2, 10(1971).
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License