IJCRR - 5(21), November, 2013
Pages: 78-82
Date of Publication: 21-Nov-2013
Print Article
Download XML Download PDF
COMMUNICATIONS IN ORAL BIOFILM
Author: Snophia Suresh, Satya Narayana
Category: Healthcare
Abstract:Molecular analysis of oral microbial communities has indicated that the human mouth provides a habitat for approximately 700 species of bacteria.The oral cavity is somewhat unique, since it provides hard, nonshedding surfaces (teeth) that are accessible for microbial colonisation. Oral microorganisms live in biofilm. Biofilms are composed of microbial cells encased within a matrix of extracellular polymeric substance such as polysaccharides, proteins and nucleic acids. In a biofilm, bacteria have the capacity to communicate with each other by the process called quorum sensing.
Keywords: oral biofilm, quorum sensing, bacterial communication
Full Text:
INTRODUCTION
Oral diseases like dental caries and periodontal diseases are usually associated with ecologic imbalance in oral microbial biofilm.[1] Periodontal disease is caused by complex microbial community which consists of several hundred different bacterial species. The dental biofilm is a microbial community that forms at high density on tooth and tissue surfaces of oral cavity. Biofilms are composed of microbial cells encased within a matrix of extracellular polymeric substances such as polysaccharides, proteins and nucleic acids. This microbial community is one of the most complex and pathogenic biofilm that exist in humans. In nature the bacteria rarely find life very easy. The major struggle faced by bacteria lies in obtaining sufficient nutrients to support growth. Human oral cavity consists of physiologically diverse organisms which coexist and thrive by adapting and responding to other organism in their local environment. The oral organisms express intraspecies and interspecies communication which favors the coordinated response by microbial community to environmental flux.
Communication of Bacteria
The bacteria within the biofilm have the capacity to communicate with each other and to other species by the process called quorum sensing. Quorum sensing allows the bacteria to sense one another and to regulate variety of physiological activities like symbiosis, virulence, motility, antibiotic production, and biofilm formation. Autoinducers are chemical signaling molecules which help in the process of bacterial communication. Quorum sensing plays a role in expressing genes for antibiotic resistance and in encouraging the growth of beneficial species to the biofilm and discouraging the growth of competitors.
Quorum sensing relies upon the interaction of a small diffusible signal molecule with a sensor or transcriptional activator to initiate gene expression for coordinated activities. The bacterial communication through signaling molecule was first demonstrated from studies of marine organism vibrio fischeri. Quorum sensing systems in bacteria have been generally divided into three classes like LuxI/LuxR–type quorum sensing in Gram-negative bacteria, oligopeptide-two-component-type quorum sensing in Gram-positive bacteria and luxS-encoded autoinducer 2 (AI-2) quorum sensing in both Gram-negative and Gram-positive bacteria.
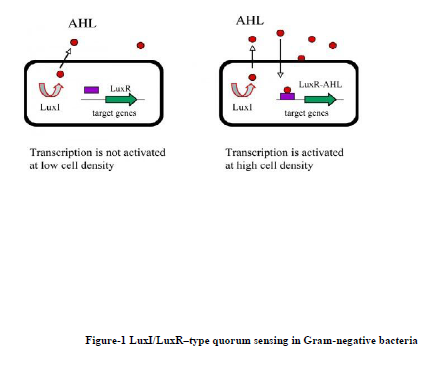
The signal molecules for Gram-negative bacteria are acyl-homoserine lactones (AHL) and the sensory mechanism that responds to signal consists of two proteins designated as LuxI and LuxR. LuxI protein is responsible for the synthesis of acyl homoserine lactones. AHL activates the transcription regulator LuxR and induces expression of luciferase operon and other quorum sensing related genes. AHLs freely diffuse across the cell membrane and increase in concentration in proportion to cell density. A cognate LuxR-like protein is responsible for recognition of the AHL and when bound to the AHL, LuxR-like protein binds to specific promoter DNA elements and activates transcription of target genes. The LuxI-like enzymes produce a specific AHL by coupling the acyl-side chain of a specific acyl-acyl carrier protein (acyl-ACP) from the fatty acid biosynthetic machinery to the homocysteine moiety of S-adenosylmethionine (SAM). This intermediate lactonizes to form acyl-HSL, releasing methylthioadenosine. (Figure-1)
Two types of quorum-sensing systems identified in gram – positive bacteria. In the first type, quorum-sensing systems generally consist of three components, a signalling peptide known as autoinducing peptide (AIP) and a two-component signal transduction system (TCSTS) that specifically detects and responds to AIP.[2] Gram-positive bacteria normally produce a signal peptide precursor, which is cleaved from the double-glycine consensus sequence and the active AIP is then exported through a peptide-specific ABC transporter into their environments. Detection of signalling peptides in Gram-positive bacteria is mediated by a two-component signal transduction system, which consists of a membrane-associated, histidine kinase protein sensing the AIP, and a cytoplasmic response regulator protein enabling the cell to respond to the peptide via regulation of gene expression.[3](Figure-2)
Bassler laboratory reported that the luciferase operon of vibrio harveyi was regulated by another quorum sensing system encoded by luxPQ genes which responded to an autoinducer designated as autoinducer 2 (AI-2).[4] AI-2 allows for inter-species communication, so it is called a “universal language”. This system is highly distributed among gram positive and negative bacteria. Autoinducer 2 produced by LuxS enzyme that functions in the activated methyl cycle and contributes to the turnover of S-adenosyl homocysteine into homocysteine and 4,5-dihydroxy-2,3-pentanedione (DPD) which undergoes intramolecular cyclization and hydration to generate R and S stereoisomers of 2,33,4- tetrahydroxymethyltetrahydrafuoran, the precursor of AI-2.Autoinducer 2 bound by perplasmic protein LuxP which interacts with sensor kinase LuxQ initiates a signal transduction cascade that flows through phosphorelay protein LuxU, a F54 dependent response regulator LuxO and a series of small regulatory RNAs in concert with the chaperone Hfq, modulate the expression of LuxR, the master regulator of the quorum sensing regulon. (Figure-3)
Quorum Sensing Inhibitors
Quorum sensing system of bacteria relies on signaling activities for their activity. So, any compounds that block these signaling molecules might inhibit bacterial quorum sensing. The use of quorum-sensing inhibitors block bacterial communication and control biofilm formation by increasing biofilm susceptibility to antibiotics and the susceptibility of the pathogens to host defenses and these compounds named as quorum quenchers.[5] Blocking of quorum sensing can be achieved through enzymatic degradation of the signal molecule, blocking the signal generation and blocking the signal reception. The acyl homoserine lactones which are the signaling molecules released by gram negative bacteria may be inactivated at alkaline PH and by the enzyme lactonases which hydrolyzes the ester bond of homoserine lactone ring.
Halogenated furanones produced by red seaweed are found to be powerful signal antagonists for prevention against bacterial colonization by interfering with acyl-homoserine lactone signalling system.[6] . Furanones inhibit bacterial colonization and biofilm formation through interference with acyl-HSL quorum-sensing pathway in Gram-negative bacteria.[7] They also interfere with AI-2 signalling systems in both Gram-negative and Gram-positive bacteria.
Numerous studies have described the application of AHL analogs or signal peptide analogs to achieve inhibition of quorum-sensing circuits in some bacteria. Zhu and Lau have recently reported a competence-stimulating peptide analog, CSP1-E1A, which inhibits competence development and reduces expression of pneumococcal virulence factors, such as choline binding protein D and autolysin A in vitro.[8]
DISCUSSION
Dental biofilm is a dynamic microbial community which forms high cell density on the tooth and tissue surfaces. This microbial community adhere to the acquired dental pellicle which favours the successive bacterial colonization. Most periodontal pathogens produce autoinducer-2 which is responsible for interspecies communication of bacteria. The widespread distribution of LuxS in both gramnegative and gram-positive bacteria has led to the suggestion that autoinducer-2 represents a universal quorum sensing system that mediates interspecies communication. In periodontal pathogens the important physiologic processes, such as the stress response, the acquisition of iron or hemin, and the development of structured biofilms, have been shown to be linked to autoinducer-2 signaling. Current data suggest that N-acyl homoserine lactone-dependent quorum sensing systems may not be common among organisms that populate the dental biofilm and are not present in several of the well-characterized pathogens associated with periodontitis. So the autoinducer-2 quorum sensing system may represent targets for the development of new therapies to control periodontal pathogen population and biofilm growth.
CONCLUSION
The biofilm has major impact on human health. Community behaviour of bacteria in oral biofilm imparts pathogenicity and antibiotic resistance. Quorum Sensing Inhibitors (QSI) have been found to increase the susceptibility of bacterial biofilms to existing antibiotics both in vitro and in vivo, thereby, increasing the success of antibiotic treatment of biofilm infections. The challenge of targeting autoinducer-2 quorum sensing arises from
the widespread distribution of LuxS and autoinducer- 2 among bacteria and the possibility that autoinducer- 2 may also be beneficial for establishment of the commensal community in the oral cavity. Better understanding of autoinducer-2 mechanism in oral bacteria and identifying the cellular components may be viable targets for therapeutic intervention of biofilm-associated oral diseases.
ACKNOWLEDGEMENT
Authors acknowlege the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.

References:
- Marsh PD. Dental plaque as a biofilm and a microbial community - implications for health and disease. BMC Oral Health 2006: 6 (Suppl.1): S14.
- Miller ST, Xavier KB, Campagna SR, Taga ME, Semmelhack MF, Bassler BL, Hughson FM. Salmonella typhimurium recognizes a chemically distinct form of the bacterial quorum-sensing signal AI-2. Mol Cell 2004: 15: 677–687.
- Dunny GM, Leonard BA (1997). Cell-cell communication in Grampositive bacteria. Annu Rev Microbiol 51:527-564.
- Bassler BL, Wright M, Showalter RE, Silverman MR. Intercellular signalling in Vibrio harveyi: sequence and function of genes regulating expression of luminescence. Mol Microbiol 1993: 9: 773–786.
- Costerton, W.; Veeh, R.; Shirtliff, M.; Pasmore, M.; Post, C.; Ehrlich G. The application of biofilm science to the study and control of chronic bacterial infections. J. Clin. Invest. 2003, 112, 1466–1477.
- Hentzer, M.; Givskov, M. Pharmacological inhibition of quorum sensing for the treatment of chronic bacterial infections. J. Clin. Investig. 2003, 112, 1300–1307.
- Zhang, L.H.; Dong, Y.-H. Quorum sensing and signal interference: Diverse implications. Mol. Microbiol. 2004, 53, 1563–1571.
- Zhu, L.; Lau, G.W. Inhibition of competence development, horizontal gene transfer and virulence in Streptococcus pneumoniae by a modified competence stimulating peptide. PLoS Pathog. 2011, 7, e1002241.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License