IJCRR - 6(19), October, 2014
Pages: 47-53
Date of Publication: 10-Oct-2014
Print Article
Download XML Download PDF
IS CD38 EXPRESSION STILL RELEVANT PROGNOSTIC FACTOR IN CHRONIC LYMPHOCYTIC LEUKEMIA?
Author: Trajkova S., Cevreska L., Ivanovski M., Simjanovska-Popova M., Pivkova-Veljanovska A, Dukovski D., Panovska-Stavridis I.
Category: Healthcare
Abstract:Introduction: There is a decade of investigations into the role of CD38 in B cell chronic lymphocytic leukemia (B-CLL). Significant percentage of CLL patients expressed transmembrane glycoprotein- CD38 on the surface of leukemic cells. Several published studies suggested that CD38 is accepted as a dependable marker of unfavorable prognosis and as an indicator of activation and proliferation of CLL cells. The aims of the present study were to establish the predictive value of the CD38 expression and to examine the relationship between CD38 positivity and other established prognostic markers in Macedonian CLL patients. Material and methods: Peripheral blood samples from 100 consecutive treatment naïve CLL patients were analyzed by flow cytometry for CD38 expression on CD5/19 leukemic cells. Various patients established prognostic characteristics and molecular markers were studied in correlation to time to treatment (TTT). The Kaplan-Meier method was used to construct survival curves, and the log-rank statistic was used to compare these curves.Results: CD38 was expressed in 61 % of the patients. Patients with high CD38 expression (30% or more) with high value of B2M and advance disease according to Binet had significantly shorter survival times (p= 0 .00001) and (p=0.00033) respectively. Multivariate analyses showed that CD38 expression is an important prognostic factor for shorter TTT associated high B2M level (P .000002), age(P.00000), gender(P.00000), lower hemoglobin level (P.00008 ),hepatomegaly (P.00086). Conclusion: CD38 expression identified a group of patients with aggressive disease that was considered by traditional staging to be early-stage disease (Rai stages 0-II or Binet A). Patients with CD38 samples have significantly aggressive disease regardless of their clinical stage. But today in era of molecular and genetics markers when CD38 is loosing it prognostic value in CLL patients prognosis, we propose serial analyses of the percentage of CD38+cells to be done, resembling indicators of leukemic cell proliferation and may signal clone evolution to a more aggressive state.
Keywords: CLL, CD38, prognosis
Full Text:
INTRODUCTION
Chronic lymphocytic leukemia (CLL) is defined as a proliferation of B lymphocytes that express surface CD19 and CD5 with expression of CD23, and low levels of immunoglobulin (Ig), CD79b and CD22(1). Some patients with CLL die within 2 years of diagnosis, yet others have a normal lifetime. A significant number of CLL patients show an aggressive form of the disease from the early stages, characterized by refractoriness to treatment, infectious and autoimmune complications, and a relatively rapid fatal outcome. However, unlike the situation in non-Hodgkin’s lymphoma, there is no standard Prognostic Index that can be used to group patients with CLL according to likely outcome or to guide treatment. The earliest staging systems, Rai and Binet systems for CLL relied on disease burden parameters. Histology patterns of bone marrow (BM) involvement and lymphocyte doubling time (LDT) have also been used as valid prognostic indicators. However, these classifications fail to distinguish patients who will eventually progress to an aggressive form of the disease from those who have a more stable form of the disease. During the past 20 years, several soluble molecules have been used as prognostic tools in CLL, including thymi dine kinase, CD23, and B2M and genetic markers of tumor cells, such as genomic aberrations, gene abnormalities (p53, ATM), the mutation status of the variable segments of the immunoglobulin heavy chain genes (IGVH) or surrogate markers for these factors, such as CD38 and ZAP-70. However, some of these markers do help in determining predicting the outcome. Fais et al. (2) were the first to report that the presence or absence of immunoglobulin gene mutation could classify patients with B-CLL into 2 groups. Hamblin et al( 3) and Damle et al( 4) independently confirmed this observation and demonstrated that patients without the immunoglobulin gene mutation had more aggressive disease with shorter survival times, whereas patients with the immunoglobulin gene mutation had less aggressive disease and prolonged survival times. Damle et al(4) studied CD38 expression in CLL patients and reported that cells from patients with unmutated immunoglobulin genes were positive for CD38 expression and this was associated with aggressive disease. In this study, we measured the levels of CD38 expression in 100 patients with CLL and correlated these values with various clinical, molecular characteristics in ordered to investigate prognostic value of CD38 in CLL population when mutational status of IG gene plays the most important prognostic role.
Materials and methods
Patient population
One hundred consecutive treatment naïve patients with B-CLL seen at University Clinic for Hematology between September 2011 and August 2013 were included in this study. At initial evaluation, date of CLL diagnosis was recorded, and the time-to- treatment end point was defined as time from first University Clinic for Hematology visit to first CLL treatment. There was no restriction for time from diagnosis to presentation to University Clinic for Hematology. All patients had more than 1 months of treatment-free follow-up from initial University Clinic for Hematology evaluation, and physicians were to conform to 1996 NCIWG guidelines for initiating treatment. The study was conducted in accordance with the Helsinki declaration. Clinical and laboratory evaluation at first University Clinic for Hematology visit included history and physical examination, standard clinical laboratory evaluation, evaluation for CD38 by flow cytometry was performed on peripheral blood at University Clinic for Hematology. Traditional prognostic factors and clinical and laboratory variables included sex, age, Rai stage, Eastern Cooperative Oncology Group performance status, physical examination with evaluation of number of involved lymph node sites (cervical, axillary, and inguinal), measurement of liver and spleen size, white blood cells count (WBC), absolute lymphocyte count(ALC), hemoglobin level, platelet count, Beta-2 microglobulin(B-2M), lactate dehydrogenase (LDH), creatinine, albumin, and quantitative immunoglobulin (Ig) levels (IgG, IgA, and IgM)(5). Peripheral blood samples were collected at the time of presentation. The samples were taken to confirm the diagnosis by flow cytometry and characterization of CD38 expression before therapy was initiated. IGHV mutation status was characterized by direct sequencing method, and patients were categorized as unmutated (IGHV ≥98% germline homology) or mutated (< 98% homology) (6). There were 100 patients who had IGHV mutation status performed by Center for biomolecular pharmaceutical analyses, Faculty of Pharmacy, Skopje Macedonia.
Measurement of CD38 expression by flow cytometry
CD38 measurements were performed at University Clinic for Hematology, as reported (6). Peripheral blood samples were obtained at the time of presentation and diagnosis and were used for flow cytometry analysis. Samples were prepared using a 3-color staining method (4). Directly labeled monoclonal antibodies (mAbs) against the lymphoid antigens CD5-phycoerythrin (PE), CD19- allophycocyanin (APC) and CD38- fluorescein isothiocyanate (FITC; Becton Dickinson Immunocytochemistry Systems, San Jose, CA) were used. Immunophenotyping expression was measured by FACS IICalibur (Becton Dickinson) using DIVA softer program. B-CLL cells (CD5/19) were gated. The degree of CD38 expression in this gated population was expressed as percentage positivity, using a threshold at 30% expression to define positive cases.
Statistical analyses
The univariate Cox proportional hazard model was used to evaluate the possible associations between time tofirst-treatment (TFT) and each risk factor singly. Variables identified as statistically significant (P, .05) in univariate analyses were subsequently included in the stepwise multivariate Cox proportional hazards model. Survival time was measured from the test date to either the last follow-up date (censored) or to the time of death by any cause. Variables examined were age, sex, Rai and Binet stages, splenomegaly, hepatomegaly, Hgb level, B2M level in the serum, WBC)count, platelet count, lymphocyte count in the peripheral blood, number of nodal sites involved with disease, IGHV mutation status. All data were collected from reviewing the patient’s record and were entered into the leukemia database. The Kaplan-Meier method was used to construct survival curves, and results were compared using the log-rank test.
Results
Patient characteristics
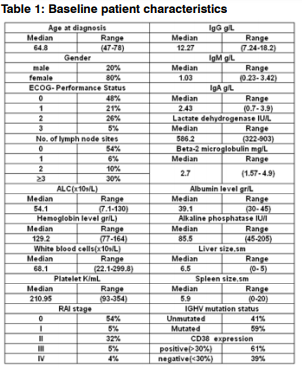
Patient characteristics are summarized in Table 1. An like other published data, there was female predomination in our population, and the median age was 64, 8 years. Ninety -one percent of patients had Rai stages 0 to II disease. CD38 expression varied in patients with CLL (Figure1). Sixty-one percent of the patients had CD38 expression in 30% or more of the cells, and 39% of the patients had CD38 expression in less than 30% of the cells. Sixteen percent of the patients had CD38 expression in more than 80% of the leukemic cells. Using 30% as a cut-off point in our analysis was arbitrary, and most of the analysis was repeated using CD38 as a continuous variable. Of the 100 patients we studied, 59% were previously untreated.
Comparison of CD38+ and CD38- patient subgroups
When the percentages of CD38+ cells were plotted for the entire patient population (data not shown), the patients could be segregated into 2 groups. Patients with 30% or more B cells expressing CD38 were considered positive, and those with less than 30% were considered negative. As shown in Table 2, there was no significant difference between the 2 groups in terms of Rai or Binet staging, splenomegaly, age, or sex. In addition, there was no significant difference between the 2 groups in WBC count, number of circulating lymphocytes. CD38+ patients were predominantly associated with unmutated IGHV (U-CLL) in addition CD38- patients were predominantly associated with mutated IGHV (M-CLL).
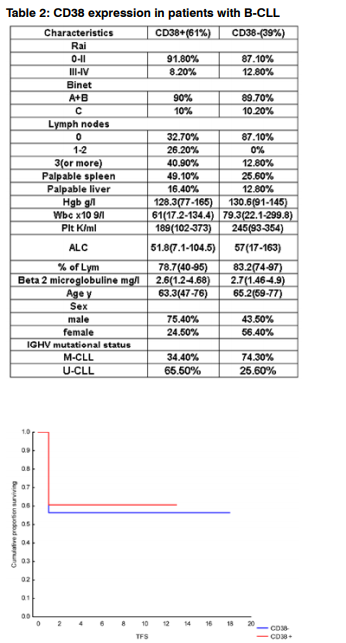
Figure 1: Time to first treatment by CD38 expression, KaplanMeier survival curves. Kaplan-Meier survival curve comparing patients with B-CLL whose samples were positive (30% or more) or negative (less than 30%) for CD38 expression; the difference is not significant at p= 0 .67.
Statistical analyses for CD38 expression
We used the univariate Cox proportional hazard model to assess associations between Treatment free survival time and various risk factors in this group of patients with CLL (Table 3).
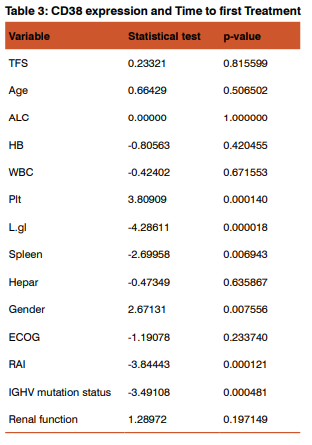
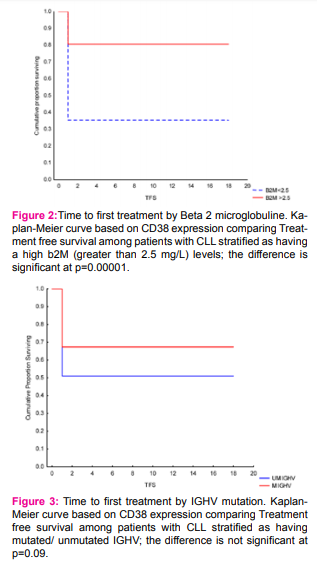
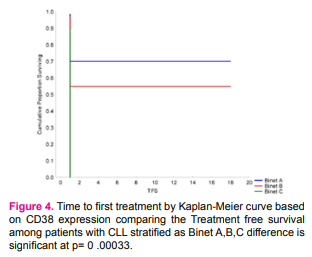
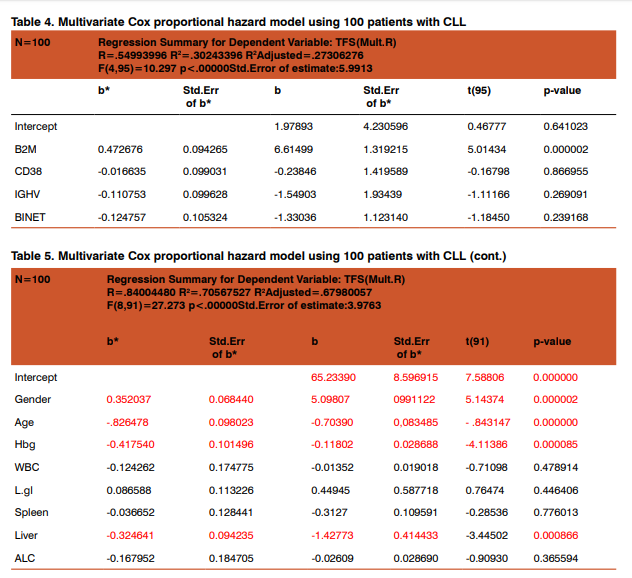
As expected patients with >30%CD38 expression have had shorter time to first treatment, associated with these prognostic factors: gender,platelet count, number of involved lymph node sites, increased spleen, advanced Rai and unmutated IGHVgene A multivariable model for time to first treatment was developed with 100 patients (100%), who had complete data available for the fitted covariates (table4). The following patient characteristics were independently associated with shorter time to first treatment: high b2M level (P .000002), age(P .00000), gender (P .00000 ),lower hemoglobin level (P .00008 ), hepatomegaly (P .00086 ).
Discussion
In this study, we found that patients with CLL could be divided into 2 prognostic groups one with worse prognoses had high CD38 expression on malignant cells (30% or more), and those with a better prognoses which had low CD38 expression (less than 30%). Multivariate analyze in our study did not confirmed that CD38 expression is an important prognostic factor in time when mulational status of IGHV gene plays the most important prognostic role. The prime subject behind the clinical observations is why expression of this molecule on the cell surface would correlate with a worse clinical outcome. One of the propose possibility is that CD38 expression reflects events occurring inside the cell. Indeed, the percentage of CD38+cells within a leukemic clone was originally described as one of two independent indicators along with IGHV gene mutations of clinical outcome in CLL (4). Because both indicators marked somewhat overlapping populations, a link with IGHV mutation status, signals mediated by B cell receptor (BCR), and CD38 was proposed(5). These two parameters are not obligatorily linked; there is one hypothesis that the presence of cell surface CD38 provides a more global molecular association to the setting, changing the balance between survival/proliferation and apoptosis in favor of the survival. CD38 is a transmembrane type II glycoprotein with a short amino-terminal cytoplasmic tail, a single mem-brane spanning region, and a long extracellular carboxyterminal domain with molecular weight of 45 kDa. It has ecto-enzymatic activity that serves in the conversion of nicotinamide adenine dinucleotide to cyclic adenosine diphosphate ribose, or cADP, an important regulator of intracellular Ca 2+ release. CD38 has no lineage restriction and mediates variable functions. In myeloid cells, CD38 is expressed on immature precursors and can be up-regulated using all-trans retinoic acid. Its presence has been associated with better prognosis in acute myeloid leukemia (AML), with the exception of AML-M3. CD38 has been reported to play a complex role in lymphocyte proliferation. Ligation of CD38 using an agonistic monoclonal antibody produced diverse responses manifested as growth or apoptosis. CD38 ligation on mature B cells protected against apoptosis and up-regulated the expression of the Bcl-2 proto-oncogene. In contrast, ligation of the CD38 molecule suppressed the growth of immature B cells in the bone marrow micro-environment. Recent studies divided patients with CLL into two prognostic groups based on their IgHV gene mutation status, one with mutation and good outcome and the other without mutation but with poor outcome. But they also identify two subgroups of CLL patients with different clinical outcomes according to CD38 expression. This distinction is based on the percentage of CD38+ leukemic cells within a CLL clone. In the majority of studies, the threshold is considered as +30% CD38+ clone members (7). The two patient subgroups that result from this cut-off point differ clinically in several ways, including overall survival, time to first treatment, (8) toward male gender, number of leukemic cells with atypical morphology(9), extent and level of adenopathy, lactate dehydrogenase and B2-microglobulin levels, (10) and absolute lymphocyte counts. These subgroups also differ in responsiveness to various therapies. In the past decade Damle et al (4) found that patients with B-CLL with high CD38 expression did not have IgH mutations and that this correlated with poor prognosis, whereas patients with low CD38 expression had IgH mutations that correlated with good prognosis. Based on this observation, they proposed that the B-CLL subset with high CD38 expression arises from pregerminal center B cells and that the subset with low CD38 expression arises from postgerminal center cells. Today it is accepted that more sensitive prognostic CLL marker is mutational status of IGHV, and where is the place of CD38 like prognostic CLL marker? IGHV mutation status is a more static marker, indicative of the cell of origin and the maturational events that occurred in the life of the B cell before its leukemic transformation. Antigen binding specificity correlates with outcome because it indicates the extent of antigenic epitopes that the BCR can engage, the affinity of these engagements, and therefore the likelihood that survival signals will be delivered to the CLL cell. It is for these reasons that CD38 expression, which is a reflection of the existing level of cellular activation within. leukemic clone often correlates with a lack of IGHV somatic mutations, which is more likely to lead to polyspecific binding, cell signaling, and eventual cellular proliferation. Thus, both prognostic indicators can be linked by the common thread of leukemic cell proliferation: IGHV mutations indicating the likelihood of binding multiple antigens and of cellular stimulation and CD38 expression representing the consequence of such binding and stimulation. But CD38 is dynamic marker, although CD38 expression was constant (negative or positive) over a variable period of time it increased. This increase in CD38 expression was associated with marked worsening of the patient’s clinical parameters. This was an unexpected finding and contradicted the concept that CD38 expression reflects the origin of B cells from pregerminal or postgerminal centers. Although we cannot rule out the possibility of an evolving new clone of pregerminal center origin, the fact that the patient was untreated suggests an evolution of the same disease. According to some other studies(11) this change is usually not large (10%) and more often, does not disregard the restrictions that mark “superior” or inferior” clinical outcome, it can occur. However, when it does, the suggested weakness can be actually viewed as a positive feature of this prognostic indicator as an rising trend in CD38-expressing cells may signal clone evolution to a more aggressive state (12). Therefore, we propose serial analyses of the percentage of CD38+cells to be done, like indicators of leukemic cell proliferation and prediction of worsening the clinical course of the disease. This laboratory setting can have an additional advantage of indicating a change in clone behavior. Some scoring systems are involving a multiplicity of markers: IGHV mutations, CD38 and/or ZAP-70 levels, chromosomal abnormalities, p53 mutations, and serum molecules such as B2-microglobulin, can be effective risk assessors and group stratifies. These approaches may require further prospective testing to refine and simplify the methods.
Conclusion
CD38 expression identified a group of patients with aggressive disease that was considered by traditional staging to be early-stage disease (Rai stages 0-II or Binet A). Patients with CD38 samples have significantly aggressive disease regardless of their clinical stage. But today in era of molecular and genetics markers when CD38 is loosing it prognostic value in CLL patients prognosis, we propose serial analyses of the percentage of CD38+cells to be done, resembling indicators of leukemic cell proliferation and may signal clone evolution to a more aggressive state.
Acknowledgement
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Rozman C, Montserrat E. Chronic lymphocytic leukemia. N Engl J Med. 1995;333:1052-1057
2. Fais F, Ghiotto F, Hashimoto S, et al. Chronic lymphocytic leukemia B cells express restricted sets of mutated and unmutated antigen receptors. J Clin Invest. 1998; 102:1515- 1525.
3. Hamblin TJ, Davis Z, Gardiner A, Oscier DG, Stevenson FK. Unmutated Ig VH genes are associated with a more aggressive form of chronic lymphocytic leukemia. Blood. 1999;94:1848- 1854.
4. Damle RN, Wasil T, Fais F, Ghiotto F, Valetto A, Allen SL, Buchbinder A, Budman D, Dittmar K, Kolitz J, Lichtman SM, Schulman P, Vinciguerra VP, Rai KR, Ferrarini M, Chiorazzi N: Ig V gene mutation status and CD38 expression as novel prognostic indicators in chronic lymphocytic leukemia. Blood 1999, 94(6):1840-1847.
5. Wierda GW, O‘Brien S.,Xuemei Wang X., et all. Multivariable model for time to first treatment in patients with chronic lymphocytic leukemia.J Clin Oncol.2011;vol. 29 (31): 4088-4095
6. Chiorazzi N, Ferrarini M. Cellular origin(s) of chronic lymphocytic leukemia: cautionary notes and additional considerations and possibilities.Blood.2011;117(6):1781-1791.
7. Matrai Z. CD38 as a prognostic marker in CLL. Hematology. 2005;10(1):39-46.
8. Morabito F, Mangiola M, Stelitano C, Deaglio S, Callea V, Malavasi F. Peripheral blood CD38 expression predicts time to progression in B-cell chronic lymphocytic leukemia after first-line therapy with high-dose chlorambucil. Haematologica. 2002;87(2):217-218.
9. Nowakowski GS, Hoyer JD, Shanafelt TD, et al. Percentage of smudge cells on routine blood smear predicts survival in chronic lymphocytic leukemia. J Clin Oncol. 2009;27(11):1844-1849.
10. Domingo-Domenech E, Domingo-Claros A, Gonzalez-Barca E, et al. CD38 expression in B-chronic lymphocytic leukemia: association with clinical presentation and outcome in 155 patients. Haematologica. 2002;87(10):1021-1027
11. Ghia P, Guida G, Scielzo C, Geuna M, Caligaris-Cappio F. CD38 modifications in chronic lymphocytic leukemia: are they relevant? Leukemia. 2004;18(10):1733-1735.
12. Chang CC, Cleveland RP. Conversion of CD38 and/or myeloid-associated marker expression status during the course of B-CLL: association with a change to an aggressive clinical course. Blood. 2002;100(3):1106.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License