IJCRR - 2(7), July, 2010
Pages: 39-55
Print Article
Download XML Download PDF
DESIGN AND EVALUATION OF KETOROLAC TROMETHAMINE SUSTAINED RELEASE MATRIX TABLETS
Author: Ch.Rajveer, B.Stephen Rathinaraj, Mohd. Fareedullah, Ganesh Sheshrao Bangale, Gajanan V.Shinde
Category: Healthcare
Abstract:Ketorolac is an NSAID. It is indicated for short term management of moderate to moderately severe acute pain, including post surgical pain , acute musculoskeletal trauma pain and post partum uterine cramping pain. The biological half life of ketorolac is 3-6 hrs, hence lower doses of ketorolac are required as loading dose and higher doses as maintenance dose. Therefore, it is considered as a suitable drug for the formulation of sustained release matrix tablets to prolong its therapeutic action. In the Present work, studies were carried
on the preparation and evaluation of matrix tablets of ketorolac using hydrophilic swellable polymers (HPMC
K4M & K15M, Guar gum) and Eudragit (RSPO) with a view to obtain sustain release characteristic to achieve
prolonged therapeutic effect by continuously releasing medication over a extended period of time after
administration of single dose. The dissolution result shows that an increased amount of polymer resulted in
retarded drug release. A concentration dependent drug release is evident in case of the polymer i.e., lower concentration of polymers, release is marginally retarded at higher concentration is considerable.
Our prepared matrix formulation containing Eudragit (RSPO) 10 % is probably showing better release based
on 80 –90 % drug release within 8 -9 hours, which is the average G.I. residence time.
Keywords: Ketorolac., Matrix tablets
Full Text:
Introduction:
Sustained release depot and repository dosage forms are terms used to identify drug delivery systems that are designed to achieve a prolonged therapeutic effect by continuously releasing medication over an extended period of time after administration of single dose. In the case of orally administered dosage forms, the period is measured in hours and critically depends on the residence time of the dosage from in the gastrointestinal tract.1-5 The system attempts to control drug concentrations in the target tissues or cells. Ketorolac tromethamine (Pka – 3.46) is an off white to White crystalline Powder6 . Ketorolac tromethamine is a non steroidal anti inflammatory drug that exhibits analgesic activity mediated by peripheral effects. Ketorolac inhibits the synthesis of prostaglandins through inhibition of the cyclo-oxygenase enzyme system. In the present study, the objective was to prepare sustained release matrix tablets of Ketorolac Keeping this in view, the present investigation has been aimed at designing suitable sustained release matrix tablets using polymers like HPMC K4M 7 , K15M, Guar Gum8 , and Eudragit (RSPO)9 . The matrix tablets were evaluated by weight uniformity, thickness, hardness10 , and In Vitro drug release studies.
Materials and Methods:
Ketorolac Tromethamine was gift samples from Dr Reddy?s Lab Hyderabad, India. Hydroxy Propyl Methyl Cellulose K15M 11-13 and Hydroxy Propyl Methyl Cellulose K4M 14 (SDFineChemicals,Mumbai,India) Guar Gum15 (Warkem Industries, Mumbai,India) Eudragit RSPO16-20 (Degussa India Pvt Ltd,India) Starch(Loba Chemie Pvt Ltd. Mumbai,India) Dicalcium Phosphate (ACTO Lab, Warangal ,India) Talc & Magnesium Stearate (LR) (SD Fine Chemicals, Boisar, India) Sodium Hydroxide (LR) and Potassium Dihydrogen Orthophosphate (Ranbaxy Lab SAS Nagar,India) Methanol (LR)(SD Fine Chemicals, Boisar, India).All other reagents and chemicals used were of analytical reagent grade.
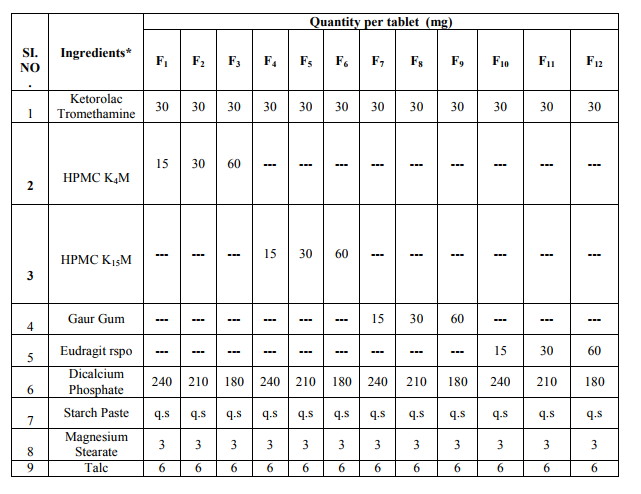
Formulation of Matrix Tablets of Ketorolac: Preparation of Sustained Release Matrix Tablets of Ketorolac with HPMC K4M as Retarding Material:
Accurately weighed quantity of Ketorolac, HPMC K4M and Dicalcium phosphate were placed in mortar and mixed. Starch paste 6 % was added to the dry blend gradually with constant kneading to ensure a homogenous mass. The dough mass was passed through a # 14 mesh sieve. Then granules were dried at 500C and dried granules were lubricated with talc (4 %) and magnesium stearate (2 %) and compressed into tablets on a 10-station punching machine using concave punches. Each tablet contains 30 mg of ketorolac. The drug matrix ratio was varied to obtain the matrix tablets of vary polymer concentration
Preparation of Sustained Release Matrix Tablets of Ketorolac with HPMC K15M as Retarding Material: Accurately weighed quantity of Ketorolac, HPMC K15M and Dicalcium phosphate were placed in mortar and mixed. Starch paste 6 % was added to the dry blend gradually with constant kneading to ensure a homogenous mass. The dough mass was passed through a # 14 mesh sieve. Then granules were dried at 500C and dried granules were lubricated with talc (4 %) and magnesium stearate (2 %) and compressed into tablets on a 10-station punching machine using concave punches. Each tablet contains 30 mg of ketorolac. The drug matrix ratio was varied to obtain the matrix tablets of vary polymer concentration.
Preparation of Sustained Release Matrix Tablets of Ketorolac with Guar Gum as Retarding Material: Accurately weighed quantity of Ketorolac, Guar Gum and Dicalcium phosphate were placed in mortar and mixed. Starch paste 6 % was added to the dry blend gradually with constant kneading to ensure a homogenous mass. The dough mass was passed through a # 14 mesh sieve. Then granules were dried at 500C and dried granules were lubricated with talc (4 %) and magnesium stearate (2 %) and compressed into tablets on a 10-station punching machine using concave punches. Each tablet contains 30 mg of ketorolac. The drug matrix ratio was varied to obtain the matrix tablets of vary polymer concentration.
Preparation of Sustained Release Matrix Tablets of Ketorolac with Eudragit (RSPO) as Retarding Material: Accurately weighed quantity of Ketorolac, Eudragit rspo and Dicalcium phosphate were placed in mortar and mixed. Starch paste 6 % was added to the dry blend gradually with constant kneading to ensure a homogenous mass. The dough mass was passed through a # 14 mesh sieve. Then granules were dried at 500C and dried granules were lubricated with talc (4 %) and magnesium stearate (2 %) and compressed into tablets on a 10-station punching machine using concave punches. Each tablet contains 30 mg of ketorolac. The drug matrix ratio was varied to obtain the matrix tablets of vary polymer concentration. Evaluation of Pre-Compression Parameter49: I.R. Studies: Method: The pure drug and its formulation were subjected to IR studies. In the present study, the potassium bromide disc (pellet) method was employed. Absorption peaks of Ketorolac tromethamine were obtained at wave numbers 1588.37/cm, and 3351.71/cm. The peaks obtained in the spectras of each formulation correlates with the peaks of drug spectrum. This indicates that the drug is compatible with the formulation components. The spectras are attached. Evaluation of the Prepared Formulation for Physico-Chemical Characteristics: In-Vitro Drug Release Studies using 0.1N HCL and Phosphate Buffer pH 7.4 Theoretically, anIn- Vitrotest for drug availability should measure in reality the physical phenomenon controlling availability in-vivo. However,InVitrotest can be carried out which will indicate the effects of these variables on the mechanism and kinetics of drug release from a dosage form. This will give an idea of how the dosage form will behave when subjected to in-vivo studies. For the present workInVitrodissolution studies were carried out in simulated G.I fluid (0.1N HCL & phosphate buffer of pH 7.4) using dissolution test apparatus USP XXIV rotating basket assembly,
Determination of Dissolution Pattern:
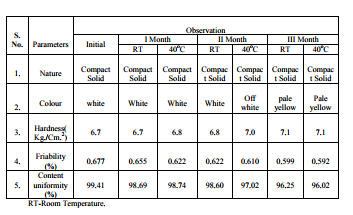
Freshly prepared test media of 900 ml was placed in dissolution vessels of dissolution test apparatus USP XXIV model. Samples of the matrix tablet of ketorolac (after weighing) was placed in basket by holding it above the solution layer immediately, basket was immersed in dissolution media and maintained at 37.5 �10C and was rotated at the speed of 100 rpm. Five ml of samples were withdrawn at fixed time intervals, and this was immediately replaced with same volume of test media. The samples withdrawn were filtered and estimated spectrophotometrically at 322 nm. Stability Studies: The selected formulation (F11) was tested for 3 months at the storage conditions of room temperature and 400 C at 75 % RH, were analyzed for their drug content including physical parameters. The residual drug contents of formulations were found to be within the permissible limits as shown in the Table – 17. No appreciable changes were found in their physical parameters. The tablets showed satisfactory physical stability at room temperature and 400 C at 75 % RH. The physical appearance did not change considerably
Discussion
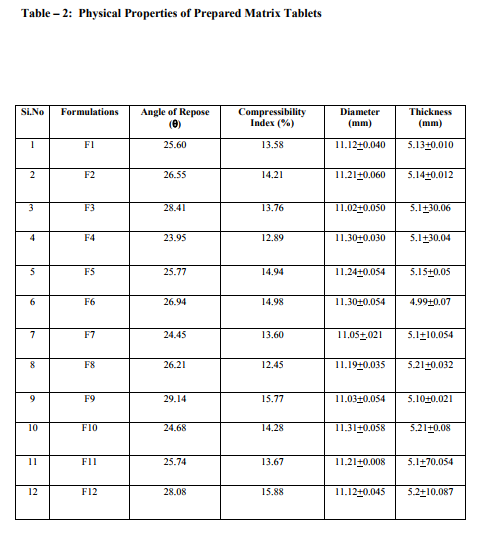
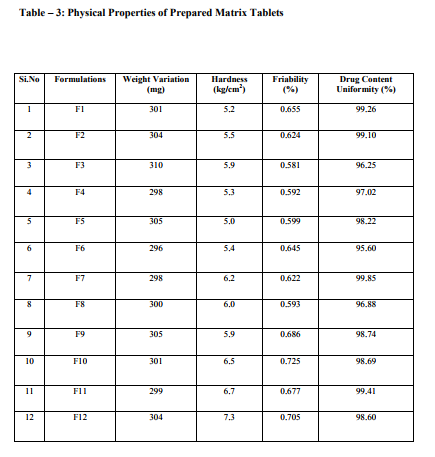
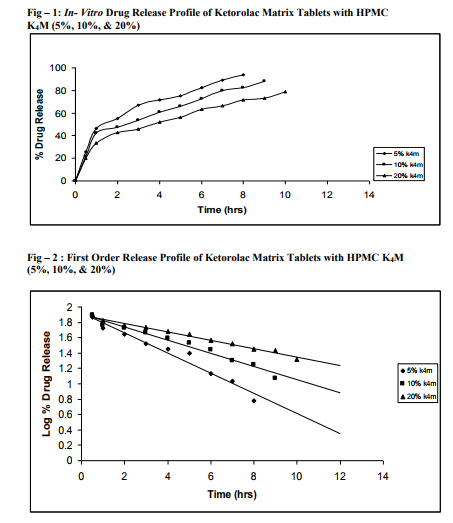
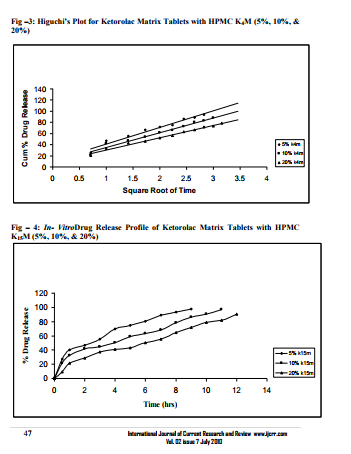
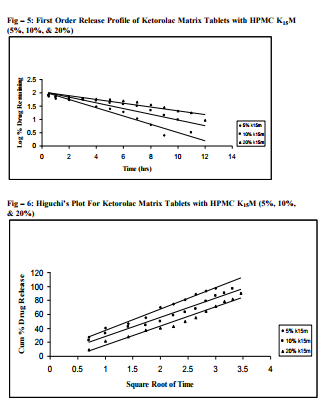
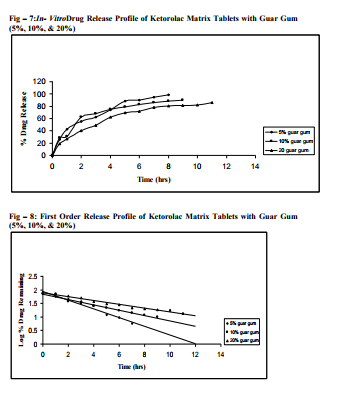
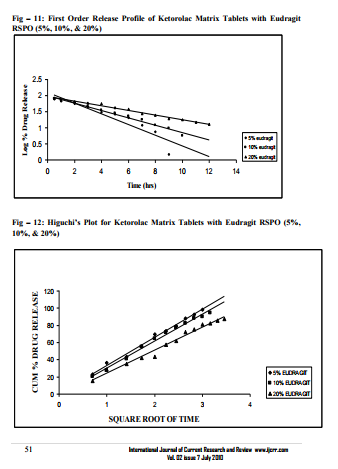
Sustained release matrix tablets of Ketorolac were prepared using hydrophilic and hydrophobic polymers. Polymers used were HPMC K4M, HPMC K15M, Guar Gum and Eudragit RSPO. Hardness, friability and content uniformity test were performed in triplicate and the results are shown in Table-3 & 4. The dissolution rate studies were performed by using USP XXIV tablet dissolution test apparatus employing rotating basket at 100 rpm (Apparatus –1). The dissolution media is simulated G.I. fluids and the study was continued upto 12 hours. HPMC K4M & HPMC K15M is semi-synthetic, nonionic cellulose ether which is widely used in sustained release dosage forms because of its non-toxic nature, its capacity to accommodate high levels of drug loading and its non pH – dependence. The drug release data for HPMC K4M formulation drug release profiles in Fig: 1-3. The drug release data for HPMC K15M formulation drug release profiles in Fig: 4-6.Guar Gum is a polymer used for the preparation of hydrophillic matrix tablets because of its high water swellability, non –toxicity and low cost. The drug release data for the Guar Gum used formulation drug release profiles in Fig: 7-9 respectively. The drug release data for the Eudragit RSPO used formulations drug release profiles in Fig: 10- 12 respectively. The formulation of HPMC K4M 5% showed release profile with Dt 50% in about 2 hrs and Dt. 90% in about 7hrs. The formulation with HPMC K4M 10% and 20% showed release profile with Dt 50% about 3 and 4 hrs and Dt: 90% in about 10 and 12 hrs respectively. Among all HPMC K4M formulations, HPMC 10% showed good release profile. Dosage form which can release 80 – 100% of drug in about 8 - 12 hours is considered to be a better formulation because the transit time in GIT is around 8-12 hours in the absence of any special gastro retentive methods. The matrix tablets cannot reside in small intestine beyond 12 hours. Both the formulation with HPMC K4M 5% and 10% showed quick release about 50% of the drug released in 2 hrs and 3 hrs respectively, and about 70% of drug released in 4 & 6 hrs respectively. The reason for quick release may be burst effect of matrix tablet. The formulation of HPMC K15M 5% showed release profile with Dt 50% in about 2.5 hrs and Dt 90% in about 8 hrs. The formulation with HPMC K15M 10% and 20% showed release profile with Dt 50% in about 4 & 6 hrs and Dt 90% in about 10 & 12 hrs. Among all HPMC K15M formulations, HPMC 10% showed good release profile. Both the formulations with HPMC K15M 5% and 10% showed quick release about 50% of drug released in 2.5 & 4 hrs and above 70% drug released in 5 & 7.5 hrs. The reason for quick release may be burst effect of matrix tablets. The formulation with Guar Gum 5%, 10% and 20% showed release profile with Dt 50% values about less than 1.5, 2, & 3 hrs and Dt 90% values were about 6, 9 & 12 hrs respectively. The formulations containing Eudragit RSPO with 5%, 10% and 20% showed Dt 50% values of about 2.5, 3 & 4.5 hrs and Dt 90% values were about 8, 9 & >12 hrs. Amongst Eudragit Rspo matrix tablets, 10% Formulations (F11) showed better release profile releasing 80 – 90% of drug in 8 hours.. In comparison, Eudragit RSPO 10% formulation is probably the better formulation because 30%, drug release in 1 hrs, 50% drug release in 3.0 hrs and 90% drug released in 9.0 hrs.
Conclusion:
Approximately all the matrix tablets prepared with different polymers exhibit concentration dependent release retardation effect. However, the required release was better with 10% HPMC K15M and 10% Eudragit RSPO, 10% Eudragit RSPO formulation showed zero order release from 1 – 8 hrs. Matrix tablets are easy to prepare and have sound technology. They are cost effective and exhibit predictable release behaviour. The matrix formulation containing Eudragit rspo 10% is probably showing better release based on the 80 – 90% drug release within 8 – 12 hrs, which is the average G.I residence time.
References:
References:
1. Lachman Leon, Liberaman HA. and Kanig JL, The Theory and practice of Industrial Pharmacy (3rd Edn), Varghese Publishing House Bombay, 430.
2. Swain Kalpan ad Gennaro AR (Ed.) Remington, The Science and Practice of Pharmacy, 19th Edition, Vol. II, 1995; 1662.
3. Telikapalli Prasanna, Patel MM., Sheth MN., Gohel MC and Chauhan GM, “Sustained Release Formulation of Verapamil Hydrochloride using Hydrophilic Matrices”, The Eastern Pharmacist, 1995, 185 – 187.
4. Brahmankar DM, Karwa RM, Jaiswal SB, “Cellulose matrix for controlled release of Ketorolac tromethamine”, Indian Drugs, 1996; 33 (3): 120 – 123.
5. Nath BS, Venkatesh, Hiremath D, “Formulation and Evaluation of Sustained Release Dosage from of Therophylline using a Combined Hydrophobic and Hydrophilic matrix”, Ind. J. Pharm. Sci., 2000; 62 (1); 33 – 36.
6. Goldstein A.M, Alter EN, Seaman JK, Guar gum. In: Whistler RL, editor. “Industrial gums, polysaccharides and their derivatives”. New York: Academic Press: 1973. P. 303 – 21.
7. Egakey AE, Mohammed, Speiser, Peter, Pharm, Acts, Helv, 1982, 57 (8), 236 – 40.
8. Nish Dhiman, S.S Poddar, A. Shajahan “Development of Matrix and coated units for pH – Independent Release”. 2003.45.
9. Hosseinali Tabandeh*, Seyed Alireza Mortazavi, Tina Bassir Guilani “Preparation of SustainedRelease Matrix Tablets of Aspirin with Ethylcellulose, Eudragit RS100 and Eudragit S100 and Studying the Release Profiles and their Sensitivity to Tablet Hardness” Iranian Journal of Pharmaceutical Research 2003, 201-206.
10. Egakey AE, Mohammed, Speiser, Peter, Pharm, Acts, Helv, 1982, 57 (8), 236 – 40.
11. Rangaiah, K.V., Madhusudahn, S and Verma P.R.P., “sustained release of Theophylline from HPMC and Eudragit tablets”, indian drugs, 1995, 32, 543-7.
12. Ju, R.T.C ,”Drug release from hydrophilic matrices . A mathematical model based on the polymer disentanglement concentration and the diffusion layer”, J. pharm. Sci., 1995;84, 1464-77
13. Salomen, J.L., Doelkar, E. and Buri, P., “Sustained release of water soluble drug from hydrophillic compressed dosage forms”, pharm. Ind., 1979,41(8), 799-802.
14. Haririan I, Ghaffari A, Mohammed Pour M, “Formulation of controlled Release Matrix Tablets of Isosorbide Dinitrate”, Indian J. Pharm. Sci., 2001, 63 (1); 24-29.
15. Bhalla, H.L. and Sanzgere, Y.D., “Improvised controlled release tablet of Salbutamol sulphate”, Indian J. Pharma, Sci, 1989,49, 22 – 25.
16. Khatua M.K., Dutta J., Prasad R., and Dutta P.K., “ Study on chitosanamine oxide bulk matrices for controlled release of cefaclor” Indian Drugs 2003,40,1.
17. V.P.Pandey., R.Manavalan., T.Sundar rajan and K.S.Ganesh “Formulation and release characteristic of sustained release diltiazem hydrochloride tablet”, Ind. J. Pharm. Sci., 2003, 65(1): 44-48.
18. Abraham MA and Shirwaikar, “Formulation of Multilayered Sustained Release Tablets using Insoluble Matrix System”, Indian J. Pharm. Sci., 1997, 59 (6), 312 – 315.
19. Ruckmani K, Agul B, Jaykar B and Ghosal SK, “Sustained Release of Nifedipine from Eudragit Matrix Tablets”, The Eastern Pharmacist, Nov, 1995, 137 – 139.
20. Farid DJ, Sadeg Shoberi N, Hassani M and Nokhodehi , “Sustained Release Formulations of Nifedipine”, Indian J. Pharm. Set., 1998, 60 (6); 375 – 378.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License