IJCRR - 3(9), September, 2011
Pages: 05-10
Print Article
Download XML Download PDF
EVALUATION OF SOME QUALITY CONTROL ASSESSMENTS OF FIVE BRANDS OF AMOXICILLIN
SODIUM CAPSULES
Author: P.O. Osadebe, K.M. Ezealisiji, M.O. Agbo, I.C. Ezeani
Category: Healthcare
Abstract:The weight uniformity, disintegration time, active drug content and bactericidal assays of five brands (A-E) of amoxicillin sodium capsules in Nsukka, Nigeria were evaluated using official methods (BP, 2001). All the five brands passed the disintegration time test with disintegration time range of 240.0-360.0 sec; all the brands passed the weight uniformity test with percentage deviation value range of 1.51-4.27 %. However, only brand D passed the active drug content test with percentage absolute drug content of 103.33%. Brand B has percentage active drug content of 107.39 % which is above the BP and USP specifications. Brands A, C and E failed the active drug content with percentage active drug content of 80.00, 72.00 and 66.67 % respectively. All the brands exhibited comparable bactericidal activity (p< 0.05) against Gram positive Staphylococcus aureus and may be used interchangeably.
Full Text:
INTRODUCTION
Humankind has been subject to infection by microorganisms since before the dawn of recorded history. Mankind has been searching for suitable therapy for nearly as long. This was a desperately difficult enterprise given the acute nature of most infections and the nearly total lack of understanding of their origins prevalent until last century. Infectious diseases caused by bacteria accounts for over 25% cases of ailment recorded in various hospitals. Therefore there is need for a search for broad spectrum antibiotic. Nigeria is a developing country and really solely on importation for most of the drugs used in the country. This has given room for influx of fake and adulterated products in our markets.
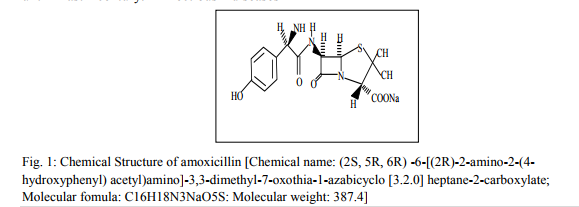
Amoxicillin is a semi-synthetic derivative of penicillins. It is indicated chemically as (2S, 5R, 6R) -6-[(2R)-2-amino-2-(4- hydroxyphenyl) acetyl)amino]-3,3- dimethyl-7-oxothia-1-azabicyclo [3.2.0] heptane-2-carboxylate (Fig. 1). The chemical formula is C16H18N3NaO5S molecular weight of 387.4 (Fig. 1). It is a white hygroscopic powder with an intense bitter taste. It is soluble in water, but sparingly soluble in ethanol and acetone. Amoxicillin is antibacterial agent with a para-phenolic hydroxyl group in the side chain phenyl moiety.amoxicilin is used in the treatment of penicillinase producing Staphylococcal and Meningococcal infection in man (Dax, 1997). The present study is aimed at determining some quantity control parameters and bacterial status of five bands of Amoxicillin sodium capsules (coded A – E) uses in Nigeria with a view to ascertaining their interchangeability.
MATERIALS AND METHODS
The following drug materials were procured; pure amoxicillin sodium powder (Juhel Pharmaceutical, Enugu), five different brands of amoxicillin code as: A – E, were randomly purchase at Ogige Market, Nsukka in June 2008. The brands under study were chosen based on frequency of prescription, use and availability in hospitals and pharmacy stores in Nsukka. Other material includes Hydrochloric acid (BDH, England), Distilled water used was freshly prepared by Science Training Centre, University of Nigeria Nsukka. The equipment used are: Electric balance (Thomas Wiley, USA) and UV-visible Spectrophometer (Model UNICO 2100).
Absolute drug content Determination
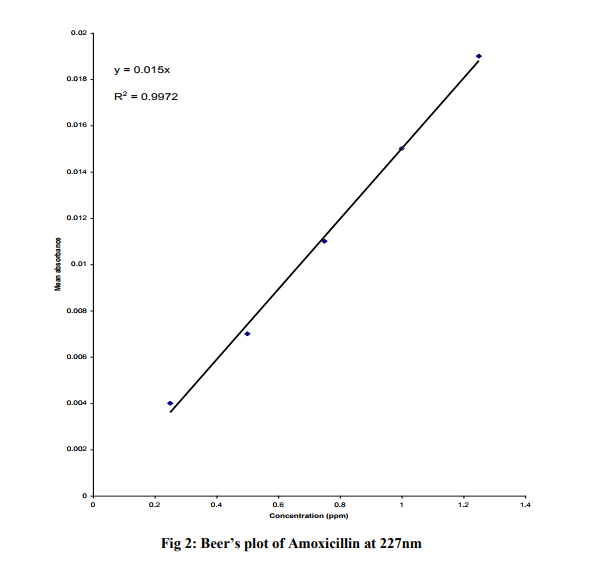
One hundred milligram (100 mg) of pure amoxicillin sodium powder was accurately weighed and dissolved in 1000 ml of distilled water. This is to obtain an equivalent active ingredient concentration of 100 ppm. Then 0.25, 0.5, 0.75, 1.0 and 1.25 mg were prepared accordingly from the stock by dissolving in 100 ml of distilled water. Exactly 5ml of the 0.75ppm solution of the stock was scanned into the UV-visible spectrophotometer to establish the wavelength of maximum absorption ( ). Then 5ml of the different dilutions were collected separately and their absorbance recorded at the established and the average absorbance determined. From the results obtained, Beer Lambert‘s plot was generated. The content of active ingredient of each of the five commercial brands of Amoxicillin sodium was extrapolated from the Beer‘s plot.
Disintegration time test
The disintegration time of randomly chosen five capsules from each sampled brands was determined in 900 ml of 0.10N HCl maintained at 37 2oC using Erweka disintegration apparatus set at 50rpm. The time taken for the capsule‘s shell to dissolve into solution was taken as the disintegration time (The British Pharmacopoeia, 2001).
Weight uniformity test
Twenty capsules were randomly selected and weighed singly using an analytical balance. The capsules were opened singly without loss of shell-material and the content removed. The shell was weighed again and the difference in weights represents the weight of the content. Mean weight, standard deviation and percentage deviation of the five brands of Amoxicillin sodium was calculated. Permitted percentage deviation of 15% for capsule weight 500mg was taken as the acceptable limit (Ofoefule, 2002).
Bactericidal Assay
The bactericidal assay of the sampled brands of amoxicillin sodium capsules were performed using gram positive bacteria Staphylococcus aureus (at 7.8125mg/ml). Agar diffusion method was used for the one-point assay (Lanani et al., 1995). The bactericidal activity of the brand and the pure sample were measured as inhibition zone diameter (IZD).
RESULTS
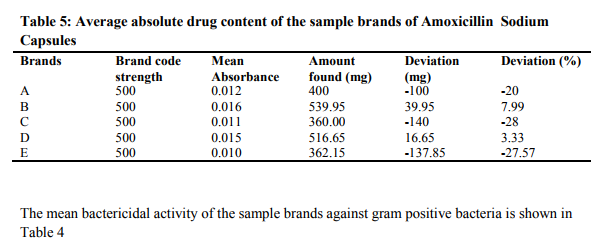
The features of the five branch of amoxicillin sodium used in this study are shown in Table 1. All the quality control parameters and the antibiotic assay of the five brands were done before their expiry dates. The table shows that most of the amoxicillin sodium used in Nigeria are imported from India and are all registered by the National Agency for Food Drug Administration and Control. (NAFDAC). The regression equation for the BeerLambert plot of pure amoxicillin was found to be Y = 0.015x and the correlation coefficient (R2) of 0.9972. The Beer‘s plot was obeyed in concentration range of 025 – 1.25ppm (Fig. 2). The weight uniformity tests results is shown in Table 2. All the five sampled brands passed the USP and IBP specifications. Table 3 shows the disintegration-time test results for the five sample brands. All the brands were complaint with the BP and USP specifications. The extrapolation from the Beer‘s Lambert plot gave the absolute drug concentrations for the different brands as shown in Table 5.
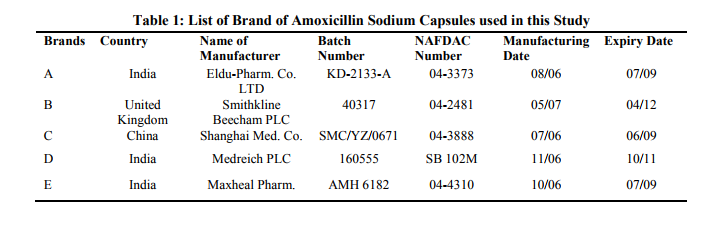
Table 2: Weight Uniformity of the Sample Brands of Amoxicillin Sodium Capsules
DISCUSSION
The weight uniformity test of the brands showed that the five brands fell within the allowed percentage deviation of 15% for capsules (Table 2). The disintegration time test in Table 3, showed that all the five sampled brands have a disintegration time ranging from 240.00 – 600.00 seconds. This is considered as satisfactory as compared to the acceptable upper limit of 900 seconds for capsules. Out of the five brands of amoxicillin assayed, three brands namely: A, C, and D showed a very wide and unacceptable quantity of active contents against the label claims – A: 400.00mg versus label claim of 500mg; C; 360.00mg versus 500mg label claim; and D: 362.15mg versus 500mg lable claim (Table 5). The results of the content of absolute drug content revealed that only brand D falls with the USP and BP (2001) acceptable limit of 90 – 110% active amoxicillin sodium. Brand B showed percentage active drug content of 107.39% and as result is considered as an over dose. The mean activities of the five brands against Staphylococcus aureus at 7.8125mg/ml shown in Table 4 since both brands have same activity against the bacteria. Also, brands C and E with similar bactericidal activity can be used interchangeably in the treatment of infections caused by Staphylococcus aureus.
CONCLUSION
This present study has shown that all the brands sampled showed similar variations with respect to the quality control parameters determined. Only brand D has active drug content within the acceptable limit. This poses a serious concern to the Health care providers in this country. The excess weight of active ingredient of brand B could cause poisoning.
References:
1. Ofoefule, S. I. (2002). A textbook of Pharmaceutical Technology and Industrial Pharmacy. Samakin, Nigeria. Pp. 57 – 66.
2. The British Pharmacopoeia (2001) Vol. 1. The stationery office Ltd, London. pp. 123 – 127.
3. Dax SL (1997). Antibacterial Chemotherapeutic Agents. Blackie Academic and Professional, New York. pp 165 – 182.
4. Lancini G, Parenti F, Gallo GG (1995). Antibiotics, A multidisciplinary approach. Plenum Press, New York. pp 1105 – 1131.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License