IJCRR - 5(1), January, 2013
Pages: 49-53
Print Article
Download XML Download PDF
HEART RATE VARIABILITY DURING DEEP BREATHING AS AN INDEX OF AUTONOMIC DYSFUNCTION IN OBESE MEDICAL STUDENTS- A CROSS SECTIONAL STUDY
Author: Rohini H.N., Padmashri S. Kudachi, Shivaprasad S. Goudar
Category: Healthcare
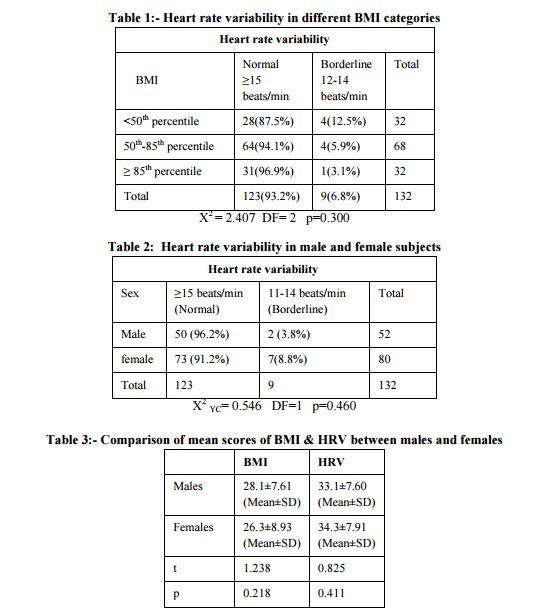
Abstract:Background: Students getting admitted in private medical colleges belong to rich affluent family background. They tend to consume relatively high calories compared to physical output, leading to significant weight gain overtime. Objective:-Study objective was to find the association between heart rate variability and obesity in obese medical students. Materials and Methods:-A cross sectional study of one forty eight medical and dental students was conducted. They were screened for body mass index. The students were divided into three groups according to WHO technical series 854 recommended cut off values. An ECG is recorded continuously throughout the period of deep breathing. The result is then expressed as the mean of the difference between maximum and minimum heart rates for the six measured cycles in beats per minute. A difference of ? 15 beats/min (normal), 11 -14 beats/min (borderline), and values of ?10 beats/min (abnormal). Results:-Among students with BMI cut off value < 50th percentile, 28(87.5%) and 4(12.5%) had normal and mild HRV values respectively. Students with BMI cut off value 50th-85th percentile, 64(94.1%) had normal and 4(5.9%) had mild HRV values. The students with BMI cut off value >85th percentile, 31(96.9%) and 1(3.1%) had normal and mild values respectively. Majority of male students [96.2 %( n= 50)] and female [91.2 %(n=73) ]showed normal HRV values. Conclusion:- In this study, there was no association between obesity and heart rate variability. The gender did not appear to be related to heart rate variability during deep breathing.
Keywords: Heart rate variability, Deep breathing High calories, Medical students, Obesity.
Full Text:
INTRODUCTION
Overweight and obesity are major public health problems and the most common nutritional disorder. The prevalence of obesity is expected to rise with urbanization and as lifestyle shift towards reduced physical activity [1] Assessing the risk for the presence of major CVS risk factors in young adults is of particular importance, since it would allow us to promptly identify the persons at high risk of development of clinical cardiovascular disease later in life and the need to implement health promotion programs and perform large scale epidemiological studies within the young adults. The BMI is the simplest and most widely accepted measure of obesity. HRV db is a reliable and sensitive and is one of the most promising clinical test for early detection of cardiovagal dysfunction in a wide range of autonomic disorders. [2,3,4] The study objective was to assess heart rate variability during deep breathing (HRVdb) in obese medical students.
MATERIALS AND METHODS The present study was conducted among the medical and dental students enrolled in the academic year 2010-2011. Study design- A cross sectional study. Selection of participants- The total number of 150 medical and 100 dental students within the age group of 18-20 years, including males and females were recruited as study participants. All these students were belonged to Asian continent. Ethical clearance- The study protocol was submitted to the ‘Institutional Ethics Committee for Human Subjects Research’ for approval. The study was undertaken after the ethical clearance was obtained by the institutional ethical committee.
Selection criteria- A proforma containing a set of questions about name, age, sex, nativity, demographic, food habits, family history of obesity, hypertension and diabetes or both, medication was given to the participants. Baseline clinical examination including heart rate and blood pressure was done. Based on this data healthy students were taken for the study. The participants who were on long term drug therapy (steroids), taking anticholinergics (including antidepressants,antihistamines),Sympathomimeti cs (α and β agonist), Parasympathomimetics and with history of endocrinal disorders were excluded from the study. Method of data collection- The total number of 250 students were screened for height and weight. Body weight was measured by the Digital scale (seca) with an accuracy of + 100gm. Subjects were made to stand on the scale platform and weighed without shoes in light clothings. Standing height was measured without shoes by using commercial stadiometer. The heels of the feet were placed together and touching the base of the vertical board. BMI was calculated using an formula Weight in Kg/ Height in m2 . Based on BMI these students were classified based on WHO Technical series 584 recommended cut off values for the adolescents [5] .
1) 85th percentile- At risk of Overweight Out of total 250 students, 52 male students and 80 female students’ total of 148 students were recruited in the study. 16 students who did not consent to participate in the study were excluded. Finally 132 underwent the test procedure. Written informed consent was taken from each subject The group II category with the BMI 50th - 85th percentile was taken as normal control group. The subjects were informed to avoid food preceding two hours of the testing and to avoid coffee, nicotine or alcohol 24 hrs and have a sound sleep prior to the test. All the subjects arrived to the laboratory in the early morning between 8.30-9.30AM. The students were 15 minutes prior to the commencement of testing procedure. The participant was explained in detail about the procedure.
The subject was made to lie down in supine position comfortably in couch. Electrocardiogram was recorded by using a instrument BPL Cardiart 108T- DIGI. Lead II was placed by applying gel and was asked to inspire for the first 5 seconds from the count of 1 to 5 and expire the next 5 seconds from the count of 5 to 1. ECG was recorded in lead II. The subjects were instructed not to talk, move hands, legs and body, sleep and cough. Short term HRV analysis was done by recording 5 minutes ECG in lead II during deep breathing. An electro-cardiogram was recorded continuously throughout the period of deep breathing and the onset of each inspiration and expiration were marked. The maximum and minimum R-R intervals during each breathing cycle were measured and converted to beats per minute. The result was then expressed as the mean of the difference between maximum and minimum heart rates for the six measured cycles in beats per minute. A difference of 15 beats or more was considered to be normal, mean values between 11 to 14 beats per minute as borderline, and values of 10 beats or less per minute as abnormal. [6]
Statistical analysis The obesity and heart rate variability scores were analyzed by chi square test, gender differences and anxiety scores were compared with X2 With Yates Correction. p< 0.05 was considered as significant.
RESULTS There was no significant heart rate variability changes as 93.2% had normal HRV values. (Table 1). Spearman’s correlation coefficient value between BMI and HRV was zero and p values was 0.996, between BMI and HRV in males p value was 0.800 and in females p value was 0.679. Hence there was no gender differences.
DISCUSSION Heart rate variability (HRV) is beat to beat variation in heart rate (i.e. in R-R intervals) under resting conditions. These beat to beat variations occur due to continuous changes in the sympathetic and parasympathetic outflow to the heart. HRV has been shown to be a good tool to quantify the tone of autonomic nervous system to the myocardium. It has been associated with high predictive value in many diseases. Heart rate variability (HRV) and respiration over the past 150 years has led to the insight that HRV with deep breathing (HRVdb) is a highly sensitive measure of cardiovagal or parasympathetic cardiac function. This sensitivity makes HRVdb an important part of the battery of cardiovascular autonomic function tests. The present study was intended to measure the obesity in young medical and dental students by standard parameters and also intended to detect the subclinical autonomic dysfunction by HRV test. In our study no change in HRV was observed. The possible reason for this findings may be for not including the tests that detect sympathetic predominance like hand grip test and cold pressor test, for not recording 24 hours long term recording. Another possibility could be the study participants being in younger age group with no morbid obesity changes. Further research including morbid obesity group may show autonomic dysfunction. The findings of the current study indicated that there were no predictable changes in cardiovascular autonomic activity as measured by HRV.
The significance of our study was early screening of autonomic dysfunction in young obese individuals and warns for specific therapeutic strategies that may reduce the risk of developing various metabolic syndromes and cardiovascular diseases involving autonomic disturbances. In our study there was no gender differences in autonomic function in the obese population. Our study included 80 female and 52 male students. Clearly more work is need to be done with the battery of autonomic function tests when they reach the adulthood. Heart rate variability measures the effect of autonomic function on heart alone so it the most useful method to investigate the effect of obesity on cardiovascular disease and it is important to emphasize the effect of obesity on heart rate variability. Decrease in heart rate variability significantly increase cardiovascular mortality [7] Higher measures of both overall and central adiposity confer greater risk of subsequent CVD in both men and women [8,9] . A previous studies concluded that there was decreased sympathetic activation and no change in parasympathetic nervous system functioning in obese children as compared to controls[10-12] An increase in sympathetic tone shows reduction in vagal tone. This allows to hypothesize that autonomic nervous system changes depend on the time course of obesity development.[13] . In Cardiac autonomic neuropathy, the disruption of the parasympathetic nervous system is usually detected earlier than that of the sympathetic nervous system Decrease in heart rate variability (HRV) is noticed as first indicator of cardiac autonomic neuropathy and decrease in E/I ratio is considered to be a sign of parasympathetic dysfunction. [14] Another study observed that the parameters reflecting parasympathetic tone (HF band, root mean square successive differences, proportion of successive normal to normal intervals and scatter plot width) were significantly and persistently decreased in obese groups with lean controls. In this study power spectral analysis was done to assess heart rate variability. [15] A study conducted by Nargai provided evidence of autonomic depression in obese children which was associated with duration of obesity [16] . In a study boys had higher LF/ HF ratio reflecting increased sympathetic modulation but counterpart females had less sympathetic modulation explaining the cardio protective effect. In this study sample size was 1,724 adolescents where our study sample size was very less which is one of the limitation of our study [17] .
CONCLUSION In this study, there was no association between obesity and hear rate variability. The gender did not appear to be related to heart rate variability during deep breathing.
ACKNOWLEDGEMENT We extend our sincere gratitude towards Department of Psychiatry, JNMC, Belgaum and thank the statistician Mr. M.D.Mallapur for analyzing the data. We are thankful to technical staff, Department of Physiology, Mr. Prem kumar Yadav and all the Ist year medical students/
References:
1. Yanowski,S.Z., Yanowski,J.A . obesity.N Eng J MED 2005; 346: 591-602.
2. ROBERT W. SHIELDS, JR, MD. Cleveland Clinic Journal of Medicine. April 2009;2:37- 40.
3. Schwartz P.J., Priori S.G. Sympathetic nervous system and cardiac arrhythmias. In: Zipes D.P., Jalife J., eds. Cardiac Electrophysiology. From Cell to Bedside. Philadelphia: W.B.Saunders.1990;330-343
4. Wheeler T, Watkins PJ. Cardiac denervation in diabetes. Br Med J1973; 4:584–586.
5. WHO technical report series 854. Physical status: The use and interpretation of Anthropometry. World health organization Geneva ; 1995: p271
6. Ewing, D. J., Clarke, B. F. Diagnosis and management of diabetic autonomic neuropathy. Br. Med. J. 1982; 285: 916-20.
7. Min HK. NIDDM in Korea. Diabet Med 1996;13:S13-5.
8. Sheldon.E.Litwin,MD,FACC. Which Measures Of Obesity Predict Cardiovascular risk?. J Am coll Cardiol 2008; 52:616-619.
9. Gelber RP, Gaziano JM, Orav EJ,Manson JE, Buring JE, Kurth T. Measures Of Obesity and Cardiovascular Risk Among men And women. J Am coll Cardoil 2008 Aug 19;52(8):605-615.
10. Mona Bedi, Shilpa Khullar and V.P. Varshney. Vascular Disease Prevention 2009; 6: 139-141
11. Piccirillo G, Vetta F, Viola E, et al. To asses autonomic modulation of cardiovascular activity in massively obese subjects. Int J Obes Relat Metab Disord 1998; 22(8): 741- 50.
12. Hofmann KL, Mussgsy L, Ruddel H. Autonomic cardiovascular regulation in obesity. J Endocrinol 2000; 164: 59-66.
13. Rabbia et al. Autonomic Modulation in Adolescent Obesity.Obesity research April 2003(4);11: 541
14. Vinik AI, Maser RE, Mitchell BD & Freeman R. Diabetic autonomic neuropathy. Diabetes Care 2003; 26: 1553–1579.
15. Riva P, et al. Obesity and autonomic function in adolescence.Clin EXP Hypertens 2001 Jan-Feb;23(1-2):57-67.
16. Nagai N. Autonomic nervous system activity and the state and development of obesity in Japanese school children. Obes Res 2003; 11: 25-32.
17. Chen-Chung Fu. Heart Rate Variability in Taiwanese Obese Children.Tzu Chi Med J 2006;18:199-204.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License