IJCRR - 5(8), April, 2013
Pages: 69-73
Date of Publication: 25-Apr-2013
Print Article
Download XML Download PDF
EVALUATION OF GENERIC AND BRANDED DRUG UTILIZATION PATTERN IN A TRIBAL DISTRICT TEACHING HOSPITAL OF SOUTH INDIA
Author: Mohammed Shakeel Mohammed Bashir, Kishor A Bansod, Ajay Khade
Category: Healthcare
Abstract:Background: Generic drugs are relatively cheap and equally effective as branded drugs. Government of Andhra Pradesh is promoting the use of generic drugs in all the districts and planning to open generic outlets in each district. We conducted the study to assess the status of use of generic drugs in a rural cum tribal district of Andhra Pradesh India. Materials and Methods: Case records of 200 patients analyzed retrospectively for use of generic and branded drugs. Equal cases were selected randomly from medicine, surgery, and gynecology and paediatrics departments. Result: A total of 952 drugs were used. 37.86% were prescribed by generic name. In general medicine, general surgery, obstetric and genecology and paediatrics 39.39%, 25.5%, 46.65% and 39.91% over all generic drugs and 52.70%, 28.86, 37.71% and 25.5% generic antimicrobials were used respectively. Ranitidine, Metoclopramide, Pheniramine, Frusemide, Dicyclomine, Nifidepine, Cefixime and Ceftriaxone were 100% prescribed by all the departments with brand names. All the departments prescribed Ampicillin as generic drug only. Conclusion: Quality controlled generic drugs are equally effective as branded drugs. We suggest that generic drugs are substitution for branded drugs which reduces over all treatment cost.
Keywords: Branded drugs, generic drugs, irrational medication
Full Text:
INTRODUCTION
In this century, increasing cost of drug therapy is a major problem for health care providers and patients in both developed and developing countries 1. Governments, insurance companies and health care providers are doing lot of efforts to control increased cost but health expenditures are not only increasing in developing countries but also in developed world every year. Apart from many reasons, ageing of the population, growing expectations regarding health by the society as well as the continuous improvement in health care facilities are the important reasons behind it in developed countries 2 while in developing nations there are different reasons.
Generic drugs are relatively cheap and equally effective as branded drugs. Because these drugs are launched after expiration of patent of the original innovative drug and companies producing such types of drugs after presenting data which indicates that their launched generic has 80%–125% bioavailability of the original drug. In most of the situations and for the many patients, variations in this range are probably having very less clinical consequences 3.
Thus generic pharmaceutical industry has the strong ability as a major force for shaping the economics of medication use. It is because generic drugs have huge potential to play an important role in containing costs of the drugs in disease management, although we cannot exactly and always easily measure the amount which we can save through the use of generic medications 4. Use of generic drugs has increased dramatically during last few decades and it is widely accepted practice in health care system 5. The economic impacts of generic drug use are much more on both the direction; in favor of consumers and against the RandD companies.
In India, pharmaceutical production is grossly diverse in nature. Lot of pharmaceuticals companies are manufacturing number of generic drugs 6. These drugs are cheap and easily available in India. Government of Andhra Pradesh is promoting use of generic drugs in all the districts and planning to open generic outlets in each district.
Prescription pattern can be evaluated retrospectively from clinical records of the institute or health care facility 7. Usually the main aim of such type of studies is to facilitate rational use of drugs in populations 8. It is one type of medical audit which review prescription status and, if needed prescription pattern can be modified for cost effective and rational use of medicine 9. Such type of studies also assesses the status of use of generic and branded drugs. In this background we planned the study to assess the actual status of use of generic drugs in this region so that suitable suggestion can be given.
MATERIALS AND METHODS
In this retrospective study case record of 200 patients belonging to general medicine, general surgery, obstetrics and gynecology and paediatrics department of Rajiv Gandhi Institute of Medical Science (RIMS) Adilabad were included. From each department case record of 50 hospitalized patients was randomly selected form medical record section of the institute. All the out patients record was excluded from the study. Case sheets were examined and findings related to use of generic and branded drugs recorded and analyzed using Microsoft Office Excel 2007.
Necessary permission was obtained from the institutional authorities for the study.
RESULT
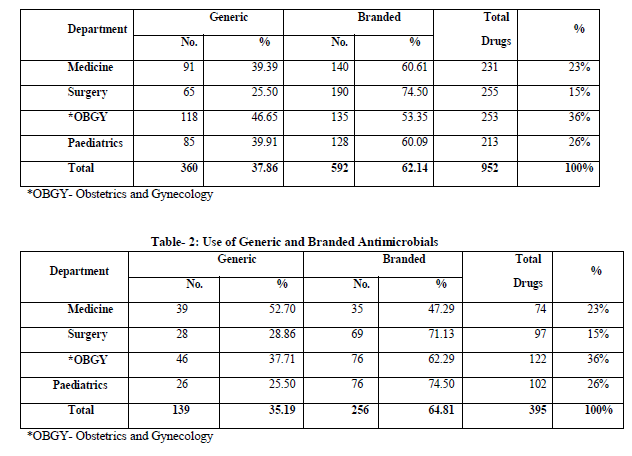
A total of 952 drugs were used by all the departments for the management of 200 different types of hospitalized patients. Out of 952 drugs 37.86% were prescribed by generic names while rest of the drugs by brand names. Maximum number of generic drugs were prescribed by obstetric and genecology (46.65%). Paediatrics and general medicine department used almost equal number of generic drugs; 39.91% and 39.39% respectively. General surgery department prescribed mainly branded drugs (74.50%) table-1.
Branded drugs were used most commonly in antimicrobial category in which all the departments used more than 50% branded antimicrobials except general medicine department in which they used 47.3% branded antimicrobials. Paediatric department used 74.5% branded drugs in antimicrobial category and general surgery department used 71.14% branded antimicrobials while obstetrics and gynecology department used 62.29% branded drugs (Table-2).
Ranitidine, Metoclopramide, Pheniramine, Frusemide, Dicyclomine, Nifidepine, Cefixime and Ceftriaxone were 100% prescribed by all the departments with brand names only. Ampicillin was the only drug which was prescribed by all the departments as generic drug only.
DISCUSSION
Uses of generic drugs become common in United States in the decade of 1970s. But in those days many of those generic drugs caused bioavailability problems 10. So it is well debated since many years that whether the generic drugs are equal to branded drugs or whether these drugs are of good quality and have fully investigated. There are also some questions that the bioequivalence studies can give the sufficient guarantee of efficacy and safety of generic drugs 11.
Generic drugs which are launched only after bioequivalence studies should be sufficient guarantee for clinicians to routinely substitute generic drugs for branded drugs. But narrow therapeutic drugs should be substituted cautiously since the safety and efficacy issue exist with these drugs. FDA firmly believes that generic drug should always be prescribed with the full expectation that the recipient will receive the same clinical benefit as can be with innovator drug 17.
In our study we observed use of almost 38% generic drugs by the four major departments of the institute. It is much higher in comparison to the observations of Irshaid et al 12 who observed use of 15% generic drugs in Saudi Arabia. But Guyon AB et al 13 in Bangladesh at primary health care centre level observed much higher number (78%) of use of generic drugs and Massele AY et al 14 in Dar es Salaam, Tanzania also found prescription of very high number of generic drugs in all health care facilities which were almost 80% of all prescriptions. In India, Ravi Shankar P et al 15 in South region found 67.4% of the drugs were prescribed by brand name which is higher in comparison to our findings. But generic drug utilization pattern was much less (7.4%) in Pune region of India as observed by Anuja et al 16. Bapna et al 17 also observed use of less number of generic drugs in comparison to branded drugs at primary health care level in southern India.
Above observations indicates that there is no uniform report regarding use of generic drugs as different patterns of generic drug utilizations are observed in different countries and even the different regions of India. There might be many reasons for such type of pattern. It may be health insurance related matter like in western world where insurance providers insist for generic substitution leading to high number of generic drug utilization. In some countries clinicians prescribe less generic drugs due to their belief of under quality of generic drugs. In some region pharmaceutical inducements to clinicians promote more utilization of branded drugs. Some governments promote generic drugs vigorously. Thus there are lots of factors which affect prescription pattern of generic drugs.
Use of Ampicillin only in generic form in our study indicates that generic drugs are qualitatively equal to branded drugs since it is most common antimicrobial agent which is used in most of the government institutions due to its cheap cost for the management of various conditions. As far as use of mostly branded cephalosporin in our region is concerned, it increases treatment cost which will not be helpful for the region since the district is tribal in nature with majority of population living in rural cum tribal areas 18.
CONCLUSION
Quality controlled generic drugs are equal in term of safety and efficacy. We suggest that generic drugs can be used since these drugs are cost effective which will have greater positive socioeconomic impact in this tribal region. Costly 3rd generation cephalosporins can be switched over with cheap generics or alternative antimicrobials to reduce health care costs.
ACKNOWLEDGMENT
We are thankful to Mr. M. Salimuddin record section incharge (Junior Assistant), RIMS Adilabad for his invaluable help during data collection. Authors also acknowledge the great help received from the scholars whose articles cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. Authors are grateful to IJCRR editorial board members and IJCRR team of reviewers who have helped to bring quality to this manuscript.
Table- 1: Use of Generic and Branded Drugs
References:
- Al Shimemeri A, Al Ghadeer H, Memish Z. Antibiotic utilization pattern in a general medical ward of a tertiary medical center in Saudi Arabia. Avicenna Journal of Medicine 2011; 1(1): 8-11.
- Ess SM, Schneeweiss S, Szucs TD: European healthcare policies for controlling drug expenditure. Pharmacoeconomics 2003; 21(2): 89-103.
- Pierre Blier. Brand versus generic medications: the money, the patient and the research. J Psychiatry Neurosci 2003; 28(3): 167–168.
- Kirking DM, Ascione FJ, Gaither CA, Welage LS. Economics and structure of the generic pharmaceutical industry. J Am Pharm Assoc (Wash). 2001; 41(4): 578-84.
- Ascione FJ, Kirking DM, Gaither CA, Welage LS. Historical overview of generic medication policy. J Am Pharm Assoc (Wash) 2001; 41(4): 567-77.
- Mohanty BK, Aswini M, Hasamnis AA, Patil SS, Murty KSN, Jena SK. Prescription Pattern in Rajahmundry. India Journal of Clinical and Diagnostic Research 2010; (4): 2047-2051.
- Hogerzeil HV. Promoting rational prescribing: An international perspective. Br J Clin Pharmacol 1995;39:1-6.
- Khan FA, Sheikh N, Salman MT. Patterns of prescription of antimicrobial agents in the department of otorhinolaryngology in a tertiary care teaching hospital. Int Res J Pharm Pharmacol August 2011; 1(5): 79-85. Available from: http://www.interesjournals.org/IRJPP {accessed on October 18, 2011}.
- Marr JJ, Moffet HL, Kunin CM. Guidelines for improving the use of antimicrobial agents in hospitals: A statement by the Infectious Diseases Society of America. J Infect Dis 1988; 157: 869-76.
- Al-Jazairi AS, Bhareth S, Eqtefan IS, Al-Suwayeh SA. Brand and generic medications: are they interchangeable? Ann Saudi Med 2008; 28(1): 33-41.
- Tschabitscher D, Platzer P, Baumgärtel C, Müllner M. Generic drugs: quality, efficacy, safety and interchangeability. Wien Klin Wochenschr 2008; 120(3-4): 63-9.
- Irshaid YM, Al Homrany M, Hamdi AA, Adjepon- Yamoah KK, Mahfouz AA. Compliance with good practice in prescription writing at outpatient clinics in Saudi Arabia. Eastern Mediterranean Health Journal 2005; 11(5/6): 922-28.
- Guyon AB, Barman A, Ahmed JU, Ahmed AU, Alam MS. A baseline survey at the primary health care level in Bangladesh. Bull World Health Organ 1994; 72: 265- 71.
- Massele AY, Mwaluko GM. A study of prescribing patterns at different health care facilities in Dar es Salaam, Tanzania. East Afr Med J 1994; 71: 314-6.
- Ravi Shankar P, Partha P, Nagesh S. Prescribing patterns in medical outpatients. Int J Clin Pract 2002; 56: 549-51.
- Pandey AA, Thakre SB, Bhatkule PR. Prescription Analysis of Pediatric Outpatient Practice in Nagpur City. Indian J Community Med. 2010; 35(1): 70-73.
- Bapna JS, Tekur U, Gitanjali B, Shashindran CH, Pradhan SC, Thulasimani M, et al. Drug utilization at primary health care level in southern India. Eur J Clin Pharmacol 1992; 43: 413-5.
- National informatics centre Adilabad, Official website of Adilabad collectorate.mht, {accessed on June 28, 2012}. Available from: http://www.adilabad.ap.gov.in
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License