IJCRR - 5(21), November, 2013
Pages: 23-29
Date of Publication: 21-Nov-2013
Print Article
Download XML Download PDF
EFFECTS OF EXTREMELY LOW-FREQUENCY ELECTROMAGNETIC FIELD EXPOSURE DURING THE PRENATAL PERIOD ON BIOMARKERS OF OXIDATIVE STRESS AND PATHOLOGY OF OVARIAN TISSUE IN F1 GENERATION
Author: Hamideh Gharamaleki, Kazem Parivar, Jafar Soleimani Rad, Leila Roshangar Mehrdad Shariati
Category: Healthcare
Abstract:Objective: In recent years, numerous reports indicate of the negative effects of electromagnetic fields on biological system. To evaluate the effects of 50Hz electromagnetic fields on parameters of oxidative stress in pregnant rats and its effects on ovarian tissue in F1 generation rats during puberty. Methods: In treatment group pregnant Wistar rats were exposed 3mT EMF for 21 days, 4 hours per day. Pregnant rats under same condition of treatment group, but off the field as a Sham group intended and pregnant rats were used as control in the room. After delivery the blood sample of mothers for biochemical analyze of MDA and SOD provided. For investigation of ovarian tissue pups, they were kept until maturity. Then adult female ovary of F1 generation were removed, fixed and prepared for light microscopy studies. Results: Biochemical analysis showed that MDA was significantly increased in the treated group in comparing with the controls, but no significant differences in SOD levels were observed between the groups. Microscopic results of the follicles F1 generation of treated group, in comparison with the control and Sham, showed that granulosa cells have interspaced from the basement membrane and in this group narrow and irregular in zona pellucida, vacuolization in ooplasm, detachment of granulosa cells was observed. Conclusion: The results suggest that pregnant maternal exposure in magnetic field cause of increases some of parameters of oxidative stress and also adverse effect on ovarian follicles in F1 generation during maturation and may impact on fertility.
Keywords: Electromagnetic field, Oxidative stress, Pathology, Ovary, prenatal
Full Text:
INTRODUCTION
The presence of electromagnetic fields (EMFs), and extremely Low-frequency electromagnetic field (ELF-EMF) are part of today life due to the increasing usage of electricity. (1) The EMF is produced by different home appliances devices such as televisions, computers, mobile phones and other life devices.(2, 3)
Byus et al (4) have reported a decrease in the activity of c-AMP independent protein kinas in
response to Radio Frequency (RF) fields amplitude-modulated at extremely low frequencies (ELF). In biological systems, undesired effects, started or supported by EMF, trigger the cascade of events that end with adverse results.(5) Three mechanisms have been suggested to explain the effects of EMF on biological systems: magnetic induction, magneto mechanical effects and electronic interaction.(6,7)
Some epidemiological studies have showed that low frequency EMFs produced by 50 and 60 Hz electricity could increase malfunction of circulatory system, Central Nervous System (CNS) and even could increase the prevalence of neoplastic disorders in peoples living near the electric poles.(8) Interesting researches are performed in related with effects of magnetic fields on cellular stress, reactive oxygen species (ROS) and free radicals, reproductive and fertility.(9)
Recent studies have shown that ELF-EMF by affecting biochemical or biophysical processes in the cell could alter cellular behavior. EMF could affect chemical bonds between neighboring atom and could also change the direction of electron spin and by this way it could affect the reactions between biological molecules. This mechanism would result in concentration and life span free radicals.(10) EMF result in excessive formation of ROS which would result in irreversible tissue damage.(11,12) ROS can easily damage DNA, Lipid and proteins of the membrane.(12,13)
Living organisms can defense against free radicals by producing antioxidant enzymes such as: Catalase (CAT), Glutathione peroxides (GPX) and Superoxide dismutase (SOD).(10) SOD is the most important antioxidant enzymes which is involved in removal of superoxide and H2O2 Since lipid peroxidation is increased by increasing of ROS, Malondialdehyde (MDA) that indicate lipid peroxidation is used as a marker oxidative stress. (9,14)
EMF also affect reproductive system and could lead to sub fertility, implantation disorder and congenital malformations.(15)
It has been reported that EMF and Radio Frequency Radiation (RFR) have detrimental effects fertility and reproductive in females.(16,17) Most of In-Vitro studies suggest that low-frequency magnetic field EMF could affect cellular metabolism, proliferation such effects many result in abnormalities in embryonic development.(18) Previous studies of female newborn rats to EMF of 10 kV/m for 24- hours/ day during developmental and neonatal period resulted in delayed puberty and some histopathological changes on reproductive organs.(19)
Since Stem cells are fully active in embryos, every physico-chemical factors could affect embryonic development including their reproductive organs. The evidence suggests that special attention must be taken in preventing embryos and neonates from EMFs. (20)
The aim of the present study is to investigate the effect of EMF on biomarkers of stress oxidative such as MDA and SOD in pregnant rats exposed to EMF and on the ovary of the F1 generation during adulthood.
MATERIALS AND METHODS
Animals
In the present research, 18 female Wistar rats weighting 200-250g with 2-3 month age were studied. The rats were supplied from animal house of the histology Department Faculty of Medicine, Tabriz University of Medical Sciences. The rats were housed in plastic cages and kept under 12h light / dark condition under 20-220C, 50-60% humidity and free access to food and water. The rats were mated and pregnancy was determined by detection of vaginal plagues. The pregnant rats were divided into 3 groups of 6 rats in each group, including: experimental, sham and Control.
Study design
The rats in treatment group randomly were exposed to 3mT EMF produced by 50Hz and the rats were exposed for 4 hours/day during the pregnancy period. The sham groups were kept in a similar condition without exposure to EMF. The control group were kept in standard condition.
After delivery, all rats were bleeding from the eye angle and the blood and sera were kept in -800C freezer for biochemical analysis and detection of MDA and SOD. For study of ovarian tissue the neonates were kept up to adolescence and at that time anesthetized using chloroform and their ovaries were removed, fixed in 10% formalin, embedded in paraffin and 5µm sections were stained with H and E and studied with light microscope.
MDA was measured on the basis of reaction thiobarbituric acid (TBA) and measuring absorbance spectrophotometer and calculated as nmol/ml. (21)
SOD was determined using RANSOD kit (obtained from RANDOX company of England) as U/ml, according to Sun et al.(22)
Statistical analysis
The data were analyzed and compared with control and sham group with SPSS v.19 software by using T-test and P<0.05 is considered as significant.
RESULTS
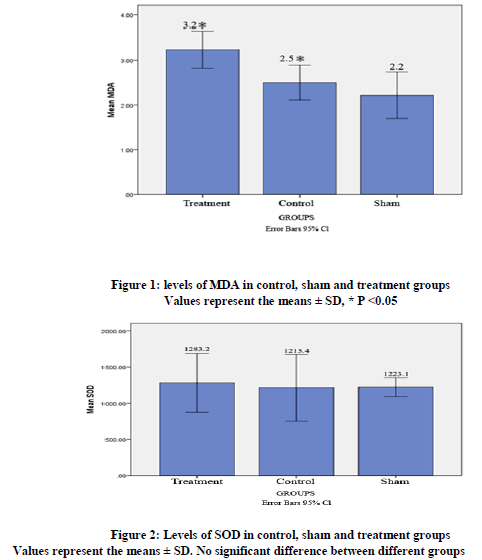
Biochemical analysis showed that in the treatment group MDA level in plasma compared with the control group was significantly higher (3.23 ± 0.39 vs.2.5 ± 0.36, P< 0.008). MDA level in Sham group was not significantly different from the control group (figure 1). SOD level were not significantly different between the groups (figure2).
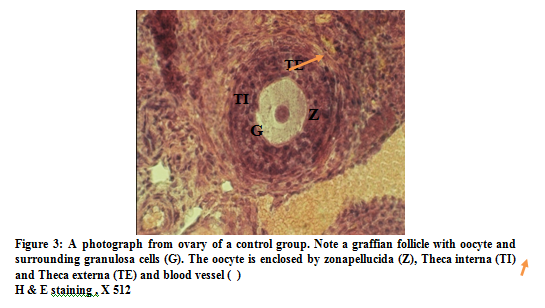
No pathological changes were observed in follicles of ovary in maturity F1 generation in control and Sham animals. In control group oocytes contain euchromatin nucleus and nucleolus, zona pellucid was obvious and a row of corona radiate cells, were obvious granulosa cells were regularly rest on the basement membrane. Internal and external theca layers were clearly seen and theca internal contained several blood vessels (figure 3). In sham group, the nucleus and nucleolus with zona pellucid and a row of corona radiate cells were similar to control group.
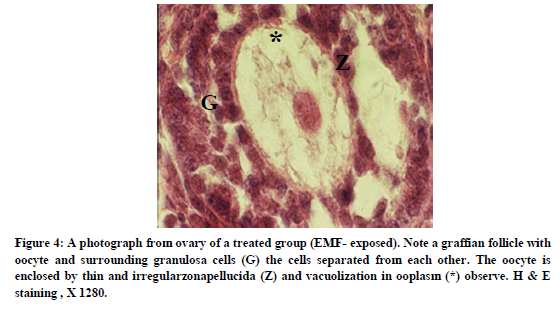
In treatment group, Granulosa cells were separated from each other and basement membrane was relatively large spaces. A thin and irregular zona pellucida was observable and ooplasm was vacuolated. Blood vessels appeared to be less extensive in the treatment group in comparison with control group (figure 4).
DISCUSSION
The results of present study that maternal exposure to ELF-EMF from 0 day of gestation lead to increasing of MDA level in treated groups in comparison with control group (figure 1). It has been reported that in rats exposed to 50 Hz electric field, MDA level increased. (23) Changes in activities of antioxidant enzymes were found following 10-day or 2-month exposures to MFs at 0.5- 50 µT.(24) Exposure of guinea pigs to 50 Hz, 1.5mT for four days increased MDA, Nitric oxide (NO) levels and myeloperoxidase activity but decreased GSH level.(25) It showed that long- term ELF-MF exposure increases lipid peroxides activity.(10) Increasing of MDA level in this study is consistent with previous studies and the results Guler G. et al that showed serum MDA level had increased in guinea pig in electric field.(26)
In the present study, the SOD level was not significantly different between groups (figure 2). Eraslan et al (2007) suggested that chronic (90 days) exposure to 50 Hz EMF does not cause oxidative stress. They showed that the concentration of antioxidant enzymes in the blood of mice did not increase. The studies showed that exposure to EMF for prolonged periods causes increasing of SOD activity in animal tissues. Levels of catalase, GPX and SOD were significantly increased in the liver and lungs of mice exposed to EMF for 8 weeks.(25) The discrepancy in SOD level in the present results, as compared to those mentioned above could be explained by the differences in the duration exposure time, intensity and frequency of EMF.
The present study shows that EMF exposure in prenatal period in ovary of F1 generation during maturity, caused separation of granolusa cells from each other and from basement membrane and thinning of zona pellucid and vacuolization of ooplasm (figure 4). It is reported that of exposure cultured follicles of mice to 33 Hz Super low frequency- electromagnetic field (SLF-EMF) for 5-day resulted in disturbed follicular growth. Also exposure to 33 Hz or 50 Hz SLF-EMF for 3 days inhibited the antrum formation of follicles cultured in-vitro.(2)
Exposure of ovarian follicles to EMF resulted in separation of Granulose cells from neighbor cells (27,28) which is similar to the result of the present study. Roshangar and colleagues have reported that EMF exposure increases degenerative changes within the follicles and narrowing of zona pellucid in treatment group.(29) Cecconi and colleagues found that exposure to 33 Hz EMF decreased significantly the growth rate of follicles and also at 33 and 50 Hz frequency reduced the proportion of follicles capable of further development. It was demonstrated that ELF-EMF exposure significantly affects the differentiation process of mouse follicles by diminishing the proportion of pre-antral follicles capable of complete growth and / or of developing antral cavities. It reported that ELF-EMF exposure has a detrimental effect on the physiological parameters of the majority of exposed follicles and that this detrimental effect on the Granulose cells, determines an impaired ability to sustain normal oocyte differentiation.(30)
CONCLUSION
The present study indicates that pregnant maternal exposure in magnetic field cause of increases some of parameters of oxidative stress and also adverse effect on ovarian follicles in F1 generation and may impact on fertility.
References:
- Sun j. Does exposure to Computers affect the routine parameters of semen quality. Asian Journal of Andrology 2005; 7 (3): 263-266.
- Chan Gye Myung and Jin Park Chan. Review Effect of electromagnetic field exposure on the reproductive system. Clin Exp Repord Med 2012; 39 (1): 1-9.
- Moussa S.A. Oxidative Stress in rats Exposed to microwave Radiation. J Biophysics 2009; 19 (2): 149-158.
- Byus CV, Lundak RL, Fletcher RM, Adey WR. Alterations in kinas' activity following exposure of cultured human lymphocytes to modulated microwave fields. Bioelectromagnetics 1984; 5: 341– 351.
- Repacholi MH, Greenebaum B. Interaction of static and extremely low frequency electromagnetic fields with living systems: health effects and research needs. Bioelectromagnetics1999; 20: 133–160.
- Adey WR. Biological effects of electromagnetic fields. J Cell Biochem 1993; 51:410–416.
- Cleary SF, Cau G, Liu LM. Effects of isothermal 45 GHz microwave on the mammalian cell cycle: comparison with the effects of isothermal 27 MHz radiofrequency radiation exposure radiation. Bioelectrochem Bioenerg 1996; 39: 167–173.
- Kula Boguslaw, Sobczak andrzej and kuska Rafal. Effects of electromagnetic field on field on free radical. Journal occup Health 2002; 44: 226-229.
- Saad El- Din Islam Mohammad. Effects of electromagnetic field on some physiological Parameters in Rat 2007; A Thesis submitted to Zagazig university for the degree of master of Veterinary Medical Science.
- Ciejka E, Kleniewska P, Skibska B, Goraca, A. Effects of Extremely Low frequency Magnetic field on Oxidative Balance in brain of Rats. Journal of physiology and pharmacology 2011; 62 (6) : 657-661.
- Canseven Gulnihal Ayse, Coskun Sule and Seyhan Nesrin. Effects of various extremely low frequency magnetic fields on the free radical processes, natural antioxidant system and respiratory burst system activites in the heart and liver tissues. Indian journal of biochemistry and Biophysics 2008; 45: 326-331.
- Ferniekim J and Reynoldajames. The Effects of electromagnetic fields from power lines Avian Reproductive Biology and Physiology. Journal of Toxicology and Enviromental Health. Part B 2005; 8: 127-140.
- Hardell Lennart and Sage Cindy. Biological effects from electromagnetic field exposure and public exposure standards. Biomedicine and pharmacotherapy 2008; 1-6.
- Somi, M.H, Hajipour B, Asi, N.A, Estakhri.R, Azar A.N, Zade M.N, Haghjou A.G and Vatankhah A.M. Pioglitazone Attenuates Ischemia/Reperfusion- Induced liver. Transplantation proceedings 2009; 41: 4105-4109.
- Chernoff N, Rogevs JM, Kavet R. A review other literature on potential reproductive and developmental toxicity of electric and magnetic fields. J Toxicology 1999; 14, PP: 91-125.
- Huuskonen H, juutilainen J, Komulainen H. J Effects of Low frequency magnetic fields on fetal development in rats. Bioelectro magnetic 1993; 14: 205-213.
- Behari jitendra and Rajamani Paulraj. Electromagnetic field exposure effects (ELF-EMF and RFR) on fertility and Reproduction. BioInitiative working Group 2012.
- Juutilainen Jukka J. Effects of Low-frequency magnetic fields on embryonic development and pregnancy. Environ Health 1991; 17:149-158.
- Exponent. EMF and Health. Health Science practice. Comprehensive Review and Update of the Scientific 2012.
- Bellieni Carlo V. Fetal and Neonatal Effects of EMF 2012; Section 19.
- Kaya H Sezik. Lipid peroxidation at various estradiol Concentrations in human circulation during ovarian stimulation with exogenous gonadotropins 2004; 36: 693-2004.
- Sun Y and Oberley Li Y. A Simple method for clinical assay of Superoxide dismutase 1988; 34: 497-500.
- Uzar Ertugral, Yilmaz H. Ramazan, Yilmaz Mustafa, Uz Efkan, Yurekli Vedat, Dundar Bumin, Koyuncuoglu Hasan Rifat, Comlekci Selcuk. Effects of 50Hz electric field on malondyaldehide and nitric oxide levels in Spinal cord of rats at prenatal plus postnatal period. Turk J Med Sci 2011; 1 (1): 65-72.
- Markkanen Ari. Effects of electromagnetic fields on Cellular Responses to Agents causing oxidative Stress and DNA Damage 2009.
- Martinez –Samano Jesus, Torres-Duran Patricia V, Juarez-Oropeza Marco A, Elias-Vinas David and Verdugo-Diaz Leticia. Effects Of Acute electromagnetic field exposure and movement restraint on antioxidant System in liver, heart, kidney and plasma of Wistar rats. Journal Radiat Biol 2010; 86: 1088-1094.
- Guler G, Turkozer Z, Seyhan N. Electric field effects on guinea pig serum. The role of free radicals. ElectromagenBiol Med 2007; 26: 207-222.
- Tilley JL, Kawalski KL, Schomberg DW and Hsueh AJ, Endocrinol. Apoptosis in atretic ovarian follicles is associated with selective decrease in messenger ribonucleic acid transcripts for gonadotropin receptors and cytochrome p450 aromatase1992; 131: 1670-1676.
- Kheradmand A, Roshangar L, Taati M, Sirotkia AV. Popho, etrical and intra cellular changes in rat ovaries following chronic administration of ghrelin. Tissue and cell 2009; 41: 511-517.
- Roshangar Leila and Soleimanirad Jafar. Ultrastructural Alteration and occurrence of Apoptosis in Developing follicles exposed to Low frequency Electromagnetic field in Rat ovary. Pakistan journal of Biological Sciences 2007; 1 (24): 4413- 4419.
- Cecconi Sandra, Gualtieri Giancaterino, Di Bartolomeo Angela, Troiani Giulia, Grazia Cifone Maria and Canipari Rita. Evolution of the effects of extremely low frequency electromagnetic fields on mammalian follicle development. Human Reproduction 2000; 15 (11): 2319-2325.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License