IJCRR - 5(22), November, 2013
Pages: 51-57
Date of Publication: 04-Dec-2013
Print Article
Download XML Download PDF
PREVALENCE AND RISK FACTORS OF MALARIA PARASITAEMIA IN FEBRILE CHILDREN WITH SICKLE CELL DISEASE IN NORTH CENTRAL NIGERIA
Author: James G. Damen, Obinwa C. Udoka, Dapus Damulak, Okpe, S.E.
Category: Healthcare
Abstract:Background: Malaria is endemic in Nigeria and sickle cell disease is very common in Nigeria and it is believed that malaria is the common cause of morbidity and mortality in children with sickle cell disease (SCD) in our environment as a result malaria prophylaxis is always recommended for them. The study was meant to determine the prevalence, risk factors and effect of malaria in sickle cell disease children. Methods: We recruited 200 febrile children with sickle cell disease and another 200 febrile non sickle cell disease children as control into the study. Two miles of their blood were collected and both thin and thick blood films were made on the same slide, stained by Giemsa methods and examined microscopically. Parasite densities were also determined and packed cell volume (PCV) were enumerated. Results: The study showed that the prevalence of malaria in febrile SCD was 49.0% and 61.0% in non SCD. The PCV of ?20% recorded the highest prevalence of 66.7% while the least prevalent of 41.1% malaria was recorded PCV ?40. In the non SCD the highest prevalent of 63.0% and the least prevalent 56.4%. SCD below 5yrs recorded the highest prevalent of 47.7% malaria while the least 20% malaria ?15yrs. In the non SCD ?5years recorded the highest prevalent while the least was 63.9% malaria. There 108 SCD males examined but 56(51.9%) were positive, while of the 112 non SCD males examined, 69(61.6%) were positive. Of the 98 SCD females examined, 42(42.9%) were positive and 88 non SCD female examined, 53(60.2%) were positive. Of the 73 SCD that uses LLINs, 21(28.8%) were positive while 127 do not used LLINs and had 77(60.0%) malaria, non SCD 51 uses LLINs, 23(45.1%) were positive for malaria, 149 without LLINs had 66.4% malaria. There was no malaria in SCD and non SCD that use insect repellants while 198 SCD that do not use repellants had 98(49.5%) malaria. SCD using door screen had 21.3% and without had 71.2% malaria, while non SCD had 32.6% and those without had 86.7%. SCD on malaria prophylaxis had 33.6% malaria, while without had 60.5% positive. Non SCD using prophylaxis had 23.5% malaria and without 64.5%malaria. SCD that take indigenous herbs at home had 56.9% malaria while without herbs had 40.8%malaria, non SCD that take indigenous herbs at home had 22.8% malaria and without had 93.5% malaria. Conclusion: We conclude that non sickle cell disease febrile children are more susceptible to malaria and have higher parasite densities than sickle cell disease febrile children studied. Therefore we recommend that malaria prophylaxis should not be limited to sickle cell disease children but to all children regardless of their genotype.
Keywords: Febrile, malaria, children, sickle cell disease.
Full Text:
INTRODUCTION
Malaria is a parasitic disease affecting red blood cells and is transmitted to human by infected female anopheles mosquitoes, malaria still rank as the number one killer disease in Africa, children, pregnant women, sickle cell anaemia patients, people in emergency situations and people living with HIV/AIDS are particularly vulnerable to malaria infection.1,2 Malaria is believe to be a major cause of morbidity in sickle cell disease (SCD) patients, it is a precipitating factor for the frequent vaso oclusive crises experienced by these patients3. Globally, some 300,000 children are born with sickle cell disease every year4. More than three quarters of these children are born in sub-Saharan Africa, where in the absence of interventions, their mortality remains high5,6. Although the sickle cell trait (i.e. hemoglobin genotype AS (HbAS) is known to protect against clinical malaria, it is widely believed that malaria is a major risk factor for death among children with SCD who are born in malaria-endemic areas7–9.
MATERIAL AND METHODS
The study was carried out in Emergency pediatric unit, ward 8 and pediatric outpatient department (POPD) of Jos University Teaching hospital north central Nigeria. The study populations were febrile children with sickle cell disease (SCD) while the controls were febrile non SCD febrile children. The children were within the ages of 6 months to 18 years. Ethical clearance was obtained from the Institutional health research ethical committee of Jos University Teaching Hospital before commencement of the study. Informed consent of the parents or guardians were obtained from both the subjects and the controls. A questionnaire was administered to each child of the consented parent or guidance; information required were demographic data, genotype results were obtained from case record note, temperature, history of malaria attack, history of chemoprophylaxis, use of long lasting insecticidal nets LLINs, repellants and door/window screens were obtained and recorded appropriately.
The method of sample collection employed was vene puncture technique.10, 11, 12 Both thick and thin blood films were prepared according to the technique11 from each blood on the same clean grease-free slide using the WHO blood film template, they were allowed to air dried and stained using 3% Giemsa stain solution for 30 minutes as describe by 13 and examined under immersion oil objective of binocular microscope. Speciation of the malaria parasites were done on the thin blood films while the parasite density was carried out on the thick blood films.
RESULTS
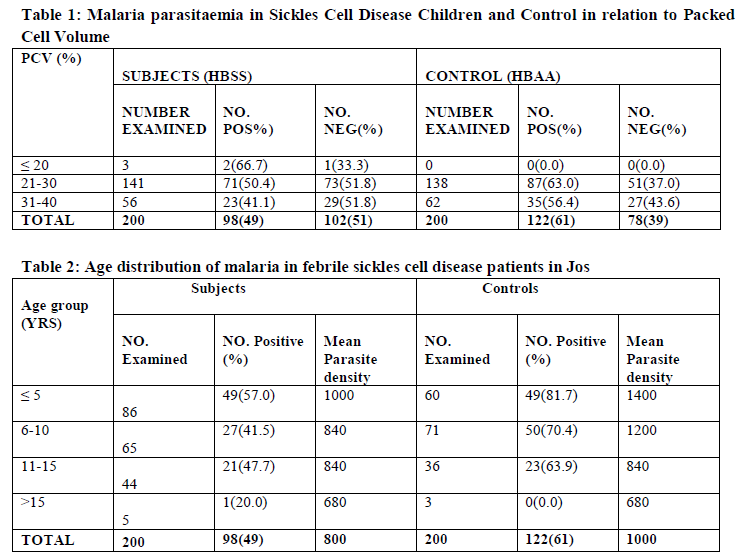
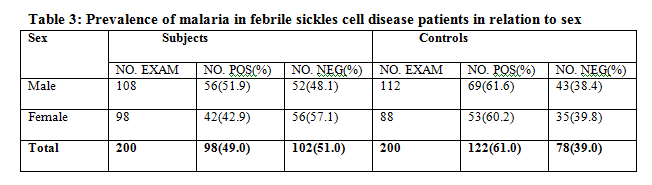
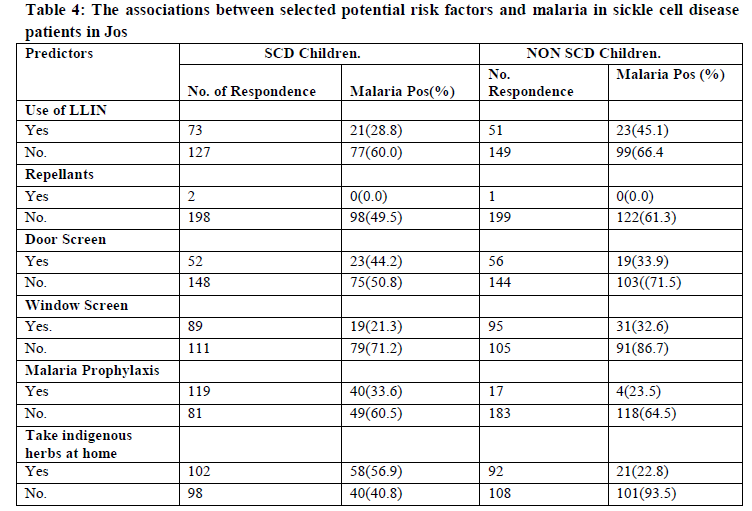
The results showed that prevalence of malaria parasitaemia reduces with increase in PCV of both subjects and the controls, this was indicated in the Three of the subjects with PCV of less than 20% had the highest prevalence of 66.7% malaria parasitaemia in the controls while there was no control with this same PCV, this was followed by 48.2% recorded by 144 with PCV of 21 to 30% and 63.0% in the corresponding controls and the least prevalence of malaria was recorded in both the subjects and controls among those that had the PCV of 31-40%, the subjects had 41.1% while the control had 56.4%. The finding of this study showed that malaria parasitaemia prevalence reduces with age in the controls while in the subjects the highest prevalence of 57.0% was recorded by those below the age of 5 years followed by 47.7% by 11 – 15 years then 41.5% by 6-10 years old while the least prevalence of 20% was recorded by those above 15years. The control had 81.7% with those within the age group of ≤ 5 years this was followed by 6-10years 70.4%, 11-15 years 63.9% those above 15 years recorded no infection. In the subjects 108 males were examined, 56(51.9%) were positive, while of the 112 males control examined, 69(61.6%) were positive. Of the 98 females subject examined, 42(42.9%) were positive and 88 female control examined, 53(60.2%) were positive. In this study of the 73 subjects that uses long lasting insecticidal net, 21(28.8%) were positive while 127 do not used LLINs and had 77(60.0%)for malaria, in the control only 51 uses LLINs, 23(45.1%) were positive for malaria, 149 without the use of LLINs had 66.4% prevalence. In the subjects only 2 that uses insect repellants and had no infection, while the 198 that do not use repellants had 98(49.5%) positive. There was only one control that used the repellant and had no infection. Of the 52 subjects that uses door screen, 23(44.2%) were positive, 148 do not used LLINs, 75(50.8%) were positive. In the control 56 uses LLINs 19(33.9%) were positive, 144 do not used LLINs 91(86.7%) were positive. The subjects had 89 that uses 19(21.3%) positive, 111 do not use window screen, 79(71.2%) were positive while of the 95 control that were not using window screen, 31(32.6%) were positive, the 105 that were not using LLINs, 91(86.7%) were positive for malaria. The subjects had 119 who were taken malaria prophylaxis, 40(33.6%) positive, 81 do not used prophylaxis, 49(60.5%) were positive while in the controls, of the 17 that uses prophylaxis, 4(23.5%) were positive and 183 that do not use prophylaxis, 118(64.5%) were positive . In the subjects 102 that always take drugs at home 58(56.9%) were positive, of the 98 that do not take drugs at home, 40 (40.8%) were positive, while 92 of the controls that takes drugs at home, 21 (22.8%) were positive and the 108 that do not take drugs at home, 101(93.5%) were positive.
DISCUSSIONS
The study gave a prevalence of 49% malaria parasitaemia in children with sickle cell disease and 61% in non sickle cell febrile paediatric patients attending Jos University Teaching Hospital. The results showed that children without sickle cell disease are more susceptible to malaria infection and have higher parasite densities than those with sickle cell disease; this finding is similar to previous study in South-South Nigeria14 who recorded 9% malaria parasitaemia in sicklers and 29% in non sicklers, also in South -West Nigeria15 24% and 43% in sicklers and non sicklers respectively. Malaria is widely considered a major cause of illness and death in patients living with SCA in sub-Saharan Africa8,9 but the finding of this study is revealing that patients with SCA have a level of immunity against malaria, this was demonstrated from the low prevalence of malaria infection and parasite densities in febrile sickle cell disease children as compared with the high prevalence of malaria and parasite densities
in febrile non sickle cell disease children. It was earlier known that there is partial protection against malaria by HbS particularly the heterozygous carriers (HbAS).16-18 However from our findings we are opined that the genotype of the homozygous (Hb SS) in which intracellular concentrations of HbS are considerably higher, might be associated with even greater protection. Our finding is also supported by studies that have investigated the mechanisms of malaria protection by HbS.19, 20 It was observed from the finding of this study that the prevalence of malaria parasitaemia decreases with increase in packed cell volume (PCV) of both the subjects and the control. Febrile sicklers patients with PCV of >20% had the highest prevalence of 66.7% malaria, while none of the non sicklers have malaria at this PCV. Sicklers with PCV of 21-30% had 48.2% malaria while their control counterpart had 63.0% malaria and for PCV of 30-40% they had 48.2% malaria while the non sicklers had 43.6% malaria, this finding might be associated with the fact that the pathogenesis of malaria infection is associated with haemolysis of the red blood cell leading to a decrease in the number of circulating red blood cells, which will result in anaemia. This finding is in agreement with 21 who reported that malaria is an important cause of anaemia. The study also showed that malaria was more prevalent on paediatric patient below the age of 5years in both sicklers and non sicklers, our findings are in concordance with those of 22. However febrile sickle cell disease children less than 5years old have a lower prevalent compared with their counterpart non sickle cell disease. This finding corresponds with the results of 23. This might be associated to their low immune status towards infections. We also observed from the results that the prevalence of malaria decreases with increase in age of the children studied for both sicklers and non sicklers, this could probably be base on the understanding that as we grow in life our immunity tends to increase thereby conferring some level of protection against malarial infections. The study showed that males had 51.9% and 61.6% prevalence of malaria for both sicklers and non sicklers respectively. The prevalence of 42.9% and 60.2% malaria were recorded by sicklers and non sicklers female counterpart. This might be associated with the fact that males are more engage in outdoor activities thereby exposing them to frequent contact with the bites of the infected female anopheles mosquitoes this finding corresponds with the previous report.24 The study showed that the prevalence of malaria is higher in children that do not uses long lasting insecticidal nets than those that uses them in both the subjects and the control, this was in agreement reports of.25 also seen in children who uses repellants, door screens, window screens, prophylaxis and those that take herbs at home before going to the health care facility. It was generally concluded that there a reduction in the prevalent of malaria in both the sicklers and non sicklers children who uses some forms of protection against malaria. However, the best method for the control of malaria is by the use of multi dimensional approaches. We therefore make our conclusion that febrile non sicklers children are more susceptible to malaria infection and have higher parasites densities than febrile sicklers children in the study area.
ACKNOWLEDGEMENT
The authors wish to thank the parents and guardians of the children who have given consent on behalf of their children to be part of the studies. We are also grateful to the nurses in the various sites of the study for their cooperation. We are also thankful to the management of Jos University Teaching hospital for the ethical approval given to us for the study, We appreciates the staff of Parasitology laboratory of Jos University Teaching Hospital and Faith medical laboratories for the technical assistance rendered for this study.
References:
- National Center for infectious diseases, Division of parasitic Diseases Anopheles mosquitoes. April,23 2004. http://www.cdc.gov/malaria/biology/mosquito/
- World Health Organization, 2007.WHO Fact sheet NO. 94. http//www.who.int/mediacentre/factsheets/fs094/en.
- Kotila TR, Management of acute painful crises in sickle cell disease. Clin Lab Haem 2005; 27:221-3.
- Modell B, Darlison M. Global epidemiology of haemoglobin disorders and derived service indicators. Bull. World Health Organ 2008; 86:480–7.
- Fleming, AF, (1979). Abnormal haemoglobins in the Sudan savanna of Nigeria. Prevalence of haemoglobins and relationships between sickle cell trait, malaria and survival. Ann Trop Med Parasitol 1979; 73:161–72.
- Molineaux L, Gramiccia G. (1980). The Garki Project: research on the epidemiology and control of malaria in the Sudan Savanna ofWest Africa. Geneva: World Health Organization, 1980.
- Serjeant GR. Mortality from sickle cell disease in Africa. BMJ 2005; 330:432–3.
- Diallo D, Tchernia G. Sickle cell disease in Africa. Curr Opin Hematol 2002; 9:111–6.19.
- Fleming AF. The presentation, management and prevention of crisis in sickle cell disease in Africa. Blood Rev 1989; 3(1):18–28.
- Okocha CEC, Ibeh CC, Ele PU, Ibeh NC (2005). Prevalence of malaria parasitaemia in blood donors in a Nigerian Teaching hospital. J. Vector-borne Dis. (142):21-24.
- Cheesbrough M (2004) Distric laboratory practice in tropical countries Part 2. Cambridge University Press. Pp 357.
- Amali, O.,Agada, BE., Awodi, N.O. and Etem, S. (2009). Malaria parasites among blood donors in Makurdi, Benue state, Nigeria. Nigerian Journal of Parasitology Vol. 30(20) 102-106.
- Federal Ministry of Health (2007) A manual for laboratory diagnosis of malaria in Nigeria National Malaria and Vector Control Division Nigeria 47-59.
- 0kuonghae HO, Nwankwo MU. Offor E. Malarial parasitaemia in febrile children with sickle cell anaemia. J Trop. Paediatric. 1992; 38(2):83-85.
- Rachel Kotila, Abiola Okesola and Olufunmilola Makanjuola (2007). Asymptomatic malaria parasitaemia in sickle cell disease patients: how effective is chemoprophylaxis? Journa of Vector Borne Disease 44: 52-55.
- Aidoo M, Terlouw DJ, Kolczak M, et al. Protective effects of the sickle cell gene against malaria morbidity and mortality. Lancet. 2002;359(9314):1311-1312.
- Williams TN, Mwangi TW, Wambua S, et al. Sickle cell trait and the risk of Plasmodium falciparum malaria and other childhood diseases. J Infect Dis. 2005;192(1):178-186.
- May J, Evans JA, Timmann C, et al. Hemoglobin variants and disease manifestations in severe falciparum malaria. JAMA. 2007; 297 (20):2220-2226.
- Cholera R, Brittain NJ, Gillie MR, et al. Impaired cytoadherence of Plasmodium falciparum-infected erythrocytes containing sickle hemoglobin. Proc Nat Acad Sci U S A 2008; 105:991–6.
- Pasvol G, Weatherall DJ, Wilson RJ. Cellular mechanism for the protective effect of haemoglobin S against P. falciparum malaria. Nature 1978; 274:701–3.
- Robert, V., MaCintyre, K., Keating, J., Trape, J-F., Duchemin, J-B., Warren, M and Beier, JC. 2003. Malaria transmission in urban Sub-Saharan Africa. American Journal of Tropical Medicine and Hygiene 68, 169-176.
- WHO (World Health Organization) and UNICEF (United Nations Children’s Fund). 2003a. Africa Malaria Report 2003. Geneva.
- Albert N. Komba, Julie Makani, Manish Sadarangani, Tolu Ajala-Agbo, James A. Berkley, Charles R. J. C. Newton, Kevin Marsh and Thomas N. Williams Malaria as a Cause of Morbidity and Mortality in Children with Homozygous Sickle Cell Disease on the Coast of Kenya Clinical Infectious Diseases 2009; 49:216–22
- Pam SH, Hawkay PM (1993). Medical parasitology, a practical approach, 2nd edition. 13:421-444.
- Jennifer C Davis, Tamara D Clark, Sarah K Kemble, Nalugwa Talemwa, Denise Njama-Meya, Sarah G Staedke1 and Grant Dorse. Longitudinal study of urban malaria in a cohort of Ugandan children: description of study site, census and recruitment Malaria Journal 2006, 5:1.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License